Running Head: Interventional Options for Advanced COPD
Abbreviations: chronic obstructive pulmonary disease, COPD; Global initiative for chronic Obstructive Lung Disease, GOLD lung volume reduction surgery, LVRS; ventilation/perfusion, V/Q; forced expiratory volume in 1 second, FEV1; National Emphysema Treatment Trial, NETT; diffusing capacity of the lungs for carbon monoxide, DLCO; Food and Drug Administration, FDA; lung volume reduction, LVR; St. George’s Respiratory Questionnaire, SGRQ; residual volume, RV; lung mesenchymal stem cell, LMSC; International Society of Heart and Lung Transplantation, ISHLT; Body-mass index- airflow-Obstruction-Dyspnea and Exercise index, BODE; ex vivo lung perfusion, EVLP
Citation: Quezada W, Make B. Interventional options for COPD-lung volume reduction surgery, bronchoscopic therapies and the future. Chronic Obstr Pulm Dis. 2016; 3(1): 446-453. doi: http://doi.org/10.15326/jcopdf.3.1.2015.0171
Introduction
Pulmonary emphysema, the destruction of lung parenchyma, is a key feature of end stage chronic obstructive pulmonary disease (COPD) that contributes to lung hyperinflation, airflow limitation, and poor exercise capacity.1 Current available therapies act primarily to relax airway smooth muscle and improve airflow limitation, but do not address the fundamental problem of architectural/mechanical disruption and tissue damage. Surgical intervention is recommended for patients with very severe Global initiative for chronic Obstructive Lung Disease (GOLD) stage IV COPD,1 and refer to therapies that aim to ameliorate the defective lung mechanics and hyperinflation that result from lung damage in advanced COPD. Current interventional therapies include bullectomy, lung volume reduction procedures (surgical and bronchoscopic) and lung transplantation. In the future, advances in stem cell transplantation may reverse the tissue destruction that accompanies end stage COPD. The following is a review of these interventions.
Lung Volume Reduction Surgery
The goal of surgical intervention in advanced COPD is to restore the mechanical alterations that result from emphysema and hyperinflation. The heterogeneity in the distribution of emphysema observed in some patients with COPD can lead to hyperinflation of the upper lobes causing compression of more normal lung tissue in the lower lobes. There may be ventilation of damaged lung apically and decreased perfusion of more normal lung at the bases, altering ventilation/perfusion (V/Q) matching at rest with further deterioration during exercise as a result of dynamic hyperinflation. Furthermore, flattening of the diaphragms places the patient at a mechanical disadvantage and is an additional contributor to dyspnea.
The goal of lung volume reduction surgery (LVRS) is two-fold: to eliminate emphysematous regions of the lung and to reduce hyperinflation. By eliminating damaged emphysematous lung regions, dead space is decreased and ventilation is shifted away from poorly perfused lung (physiologic dead space regions) to areas that are better perfused, improving V/Q matching. Reducing hyperinflation results in expansion of compressed normal lung, improvement of diaphragmatic mechanics, and reduces dynamic hyperinflation during exercise.
LVRS was initially proposed in the late 1950s by Brantigan and Mueller,2 who operated on patients with diffuse emphysema using staged bilateral thoracotomies, multiple lung resections and plications of the most diseased-appearing lung regions, and radical hilar stripping to denervate the lung (in an effort to reduce sputum production). Approximately 90% of patients had improvement in symptoms. However, the procedure failed to gain widespread acceptance as it was associated with an early mortality rate of 18% and lack of objectively documented benefit.2 Cooper and colleagues3 revisited the concept of LVRS in the 1990s, performing bilateral LVRS using median sternotomy in 20 patients with diffuse emphysema resulting in an 82% mean improvement in the forced expiratory volume in 1 second (FEV1) and no associated mortality. These results led to increased public demand for surgery, but the varying results and surgical techniques between centers raised the question of whether a randomized controlled trial was needed to evaluate LVRS.4 The National Institutes of Health subsequently sponsored the National Emphysema Treatment Trial (NETT), a large, prospective, randomized, multicenter trial comparing LVRS to optimal medical therapy for COPD (based on American Thoracic Society guidelines). The primary outcome measures were survival and change in exercise capacity. Overall, there was a survival benefit in the surgery group compared to the control group. An early publication identified a group of patients for whom LVRS was associated with significant morbidity and mortality: those with FEV1 < 20% predicted and diffusing capacity of the lungs for carbon monoxide (DLCO) < 20% predicted, or patients with FEV1 < 20% predicted and homogenous emphysema. In these patients, there was an 18% mortality rate in the first 30 days following surgery.5 The best outcomes were seen in 290 patients with upper lobe predominant emphysema and low baseline exercise tolerance; there was significant improvement in exercise and health status and more importantly, LVRS conferred a survival advantage (overall mortality 18.7% versus 33.8%, p=0.005).6,7 In summary, an algorithm (Figure 1) has been proposed in which pulmonary function testing (FEV1, DLCO) can be used to identify patients at increased risk for mortality, and computed tomography and cardiopulmonary exercise testing can identify those patients who will gain survival and quality of life benefit from LVRS (those with upper lobe predominant disease and low exercise capacity). A follow up study investigating long term outcomes following LVRS showed persistent survival benefit and improvement in exercise capacity at a median follow up of 4.3 years.7
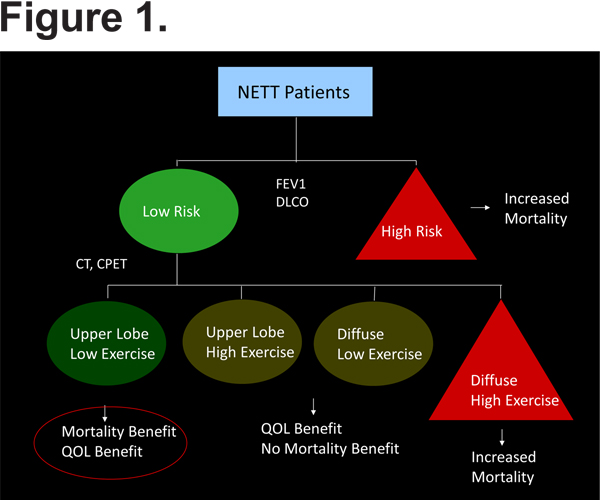
It was expected from the NETT results that LVRS would be more frequently employed as a treatment option to those patients found to have benefit from the procedure. A recent review by the American Thoracic Society, however, showed that in the 8-year period following the publication of the NETT results only 538 LVR surgeries had been reported.8 This was likely related to the initial perception of the high risk of death associated with the surgery in the high risk group. Even after exclusion of patients who were not deemed high risk, 90-day post-surgical mortality was 5.5%, and a significant number of patients had post-operative complications including air leak lasting 7 days or more (in > 46% of the patients). There was also the hope that the development of less invasive bronchoscopic procedures would spare patients the risks of a more invasive surgery.
Bronchoscopic Approaches to Lung Volume Reduction
COPD Foundation Guidelines call for lung volume reduction (LVR) as a therapeutic option for the emphysema phenotype of COPD.9 LVRS is still the only intervention whose mortality benefit has been proven in a large randomized controlled trial. The results of the NETT showed that in a specific phenotype of COPD patients (upper lobe dominant emphysema, poor exercisers), LVRS had an approximately 50% reduction in mortality rate. Patients with more diffuse distribution of emphysema and high capacity for exercise had poorer outcomes including increased mortality.6 This emphasized the importance of careful patient selection and represented the first use of subtyping and phenotyping in COPD.
Despite its proven mortality benefit, LVRS is still associated with significant morbidity (median pneumothorax 7 days, pneumonia 18%, re-intubation 22%, tracheotomy 8%) and mortality (4.3%).10 Given these data, the development of less invasive interventions with potentially less morbidity becomes an attractive idea. Currently, valves and surgical coils are the interventions that are best studied. Sealants and steam driven interventions have been studied also with less success.
Endobronchial Valves
The Endobronchial Valve for Emphysema Palliation Trial (VENT)11 is the largest pivotal trial examining the effectiveness of endobronchial valves that function by collapsing regions of the lung with heavy emphysema involvement (by means of unidirectional air flow). Patients with greater than 25% heterogeneity in terms of involvement of upper and lower lobes had clinically important responses in FEV1 and 6-minute walk distance. The majority of patients in the trial who had less than 25% heterogeneity had minimal to no response. One reason was that the technical placement of the valves was not closely monitored, and approximately 40% of the patients did not achieve adequate lobar occlusion. Furthermore, we learned that some patients have substantial collaterals that connect the targeted lobe from the non-targeted lobes. Air flow into the targeted lobe can persist via collaterals, making the mechanical intervention less effective. Using fissure integrity as a proxy for inter-lobar collaterals12 it was found that up to 50% of participants did not have adequate fissure integrity. Participants with complete fissures who received endobronchial valves had a greater reduction in volume in the targeted lobe and improved inflation in the adjacent lobe compared to all participants in the trial. These results highlight the importance of closely assessing fissure integrity in future trials of endobronchial interventions. An endobronchial catheter has been developed to allow the detection of collateral airflow into a targeted lobe prior to insertion of an endobronchial valve.13
Examining the complication rates at 1 year, there was no difference in death between the groups. There was a slight increase in the rate of pneumonia distal to the valve, generally treated with the valves intact. The Food and Drug Administration (FDA) recommended a repeat trial in patients with high heterogeneity and a re-designed pivotal trial (LIBERATE) has begun enrolling, featuring the enrollment of participants with >15% heterogeneity, using Chartis Bronchoscopic flow to assess for collateral airflow, repositioning valves that have been misplaced or have migrated, and more vigilantly monitoring for pneumothorax with a 5-day required admission.
Endobronchial Coils
Lung volume reduction coils were developed to address the subset of patients with less degree of heterogeneity and with inter-lobar collaterals. Currently, PneumRx owns the technology with nitinol devices that are inserted straight via a bronchoscope that then recoil to collapse the targeted lobe. The procedure calls for the placement of 10-15 coils of various sizes (100, 125, and 150 mm) sequentially, 2-4 months apart. An analysis showed that both heterogeneous and homogenous patients defined using visual and quantitative approaches can have significant improvement in clinical parameters including 6-minute walk distance and St. George’s Respiratory Questionnaire (SGRQ) scores.14 The RENEW trial is a pivotal trial that completed recruitment in October 2014, and involved the placement of 10-14 coils (n=315) with treatment of the contralateral lung 4 months later. The primary outcomes were 6-minute walk distance and safety. The initial residual volume (RV) inclusion criteria of 225% predicted was lowered to 175% predicted to improve recruitment. One-year follow up is expected in the Fall 2015, and complete results will be available in the Spring 2016.
The decision between endobronchial valve versus endobronchial coil placement in patients who do not desire or are poor surgical candidates for LVRS can be based on assessment of lung anatomy. The effectiveness of endobronchial coils relies on some degree of airway structure to allow the coils to grab and collapse the targeted lobe. In a patient with heterogenous disease (upper lobe predominant) fissure integrity can be used to determine which intervention is optimal: endobronchial valve (complete fissure) and endobronchial coil (incomplete fissure). Patients with homogenous disease and very severe hyperinflation may benefit from coil placement (and not valve placement). Hence, precision medicine principles can be applied to patients with advanced COPD based on their individual characteristics.
In summary, while LVRS is the only treatment proven to prolong life in patients with heterogeneous emphysema, the patient wishes for less invasive procedures must be respected. Endobronchial valves should be the first consideration in collateral-negative heterogeneous emphysema patients. Coils can be considered in collateral-positive heterogeneous emphysema patients and in severely hyperinflated homogenous emphysema patients.
Stem Cell Therapies for Advanced Emphysema
Cellular regenerative therapies have been used successfully in the treatment of hematological and orthopedic conditions, and partial successes have recently been reported for treatment of cardiac diseases.15,16 The technology exists to potentially advance our knowledge of stem cell research as a potential therapeutic modality in patients with advanced emphysema to regrow functional lung tissue at diseased sites using pharmacologic or cell-based approaches (i.e. lung regeneration).17 Over the last decade, there has been an increase in the number of studies looking at stem cell therapies; currently over 30 trials are focusing on pulmonary disease and 7 specifically on emphysema). Of the emphysema trials, very few are taking place in academic centers in the United States, and only 1 is a randomized controlled trial. All of them use the same cell type, and only one has presented acceptable data.18 Currently a PubMed search will yield approximately 5,000 references.
Current data suggests that stem cells are compartmentalized within the lung: in the upper and lower airways, the alveolar spaces, and in the vasculature and interstitium. The working hypothesis has traditionally been that in order for stem cell therapies to work there needs to be a pleuripotent stem cell that has the capacity to recapitulate ontogeny. A more modest approach is to focus on a disease where a compartment of the lung is particularly abnormal and try to design around the disease, within that compartment. The development of successful stem cell-based therapies will require addressing multiple challenges, including identification of an appropriate regenerative cell population and determination of the correct dose. Methods for isolating, manufacturing, and purifying uniform populations of functional cells for therapeutic application must be developed. Technologies to facilitate engraftment at the intended target site and cell migration from the airway and alveolar surface to the niche where they can exert regenerative effects are being developed. The development of strategies to prevent anoikis in the immediate post transplantation period and to address rejection associated with allogeneic tissues are also required.19
At Brigham and Women’s Hospital, a stem cell research program utilizes a particular stem cell, a lung mesenchymal stem cell (LMSC), which is much like an undifferentiated fibroblast. Brigham has developed a technique of biopsying the lung, isolating and growing these cells and subsequently placing them back into the patient. This explant approach preserves stem cell niche, allows for autologous treatment for the generation of >30 million P10 autologous cells from a single piece of tissue by serial tissue passage. This latter step preserves the pleuripotent nature of the first generation cells. Characterization of these cells reveal that they are highly clonogenic (can generate a large number of cells), are highly proliferative, and maintain multi-potency (can differentiate into different types of cells). However, LMSCs also have the ability to build lung tissue (collagen and laminin) and secrete growth factors that promote vascularization and epithelial cell proliferation. When placed on an integrin-activating bioscaffold, it is observed that LMSCs retain the capacity to engraft and migrate to the lung interstitium. The first study with LMSCs, a safety study in sheep with bullous emphysema, demonstrated that injection into emphysematous areas resulted in increased perfusion, suggesting a therapeutic impact. Computed tomography imaging performed 1 month after injection shows shrinkage of the bullae resulting from proliferation of healthy lung tissue.17,19
In summary, regenerative based therapies are advancing into trials for patients with end stage emphysema. Current trials are not using stem cells to regenerate epithelial lineage cells, but rather are employing stromal cells with regenerative paracrine effects to influence endogenous repair and regeneration. To date, bone marrow derived stem cells have not proven effective for this application, and alternate sources of pleuripotent stromal cells are being explored. Many challenges still plague stem cell research, as highlighted above, and regulatory hurdles are also substantial. The FDA has been supportive in trying to develop an approvable approach. It is highlighted that current data does not support the effectiveness of stem cell therapy in advanced COPD.
Lung Transplantation for COPD
Since its dawn of clinical reality in the mid 1980s, lung transplantation has been a therapeutic option for patients with end-stage lung disease from COPD, offering hope for improvement in quality of life and symptom burden. Worldwide, there are nearly 4000 lung transplants performed annually, with about 33% of the historical cohort being done for the indication of COPD.20 Unfortunately, there remains significant morbidity and risk of mortality, largely due to the adverse effects of chronic immunosuppression that is required for life. For COPD, the median survival after lung transplant remains 5.5 years.20 Due to some data suggesting improved survival in recipients who receive a bilateral transplant rather than a single lung transplant, current global trends demonstrate an increasing use of the bilateral operation.18
The Pulmonary Council of the International Society of Heart and Lung Transplantation (ISHLT) recently published an updated consensus document for the selection of lung transplant recipients.21 Several patient features that were once absolute contraindications to lung transplantation are now referred to as relative contraindications, including age > 65 years, class I obesity, and infections with human immunodeficiency virus, hepatitis B, and hepatitis C. More than 15% of patients undergoing lung transplantation worldwide are now over age 65, and the average age of recipients is approaching 60 years-old.22
One important feature of the updated consensus statement is careful consideration of the clinical features that determine timing of referral to a transplant center as being different from those that determine timing for that center to pursue listing of that same patient. The reason for this dichotomy is an attempt to allow for an opportunity for discussion and for amendment of various comorbidities (i.e. obesity or substance abuse) that are only possible with an early referral. There is a concept of a transplant window, representing the period where a patient is sufficiently advanced in his disease to benefit from transplant, but before he becomes so ill as to not have sufficient time to receive a donor offer or to have difficulty in recovery from the operation. (Figure 2)
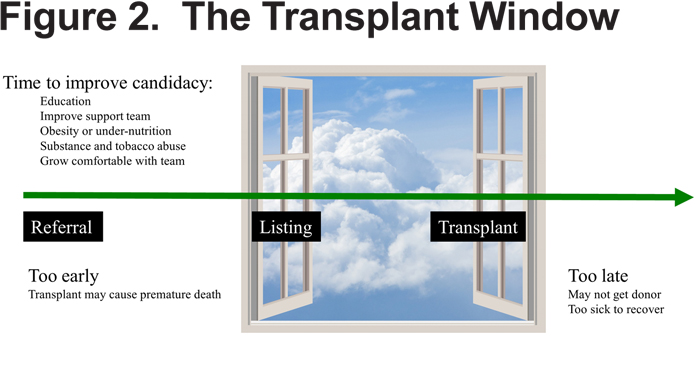
Features that should prompt correct timing for referral in COPD include:
- Disease is progressive, despite maximal treatment including medication, pulmonary rehabilitation, and oxygen therapy.
- Patient is not a candidate for endoscopic or surgical LVRS
- Simultaneous referral of patients with COPD for both lung transplant and LVRS evaluation is appropriate
- Body-mass index-airflow Obstruction-Dyspnea and Exercise index (BODE index) of 5 to 6
- PaCO2 >50 mmHg or 6.6kPa and/or PaO2 <60 mmHg or 8kPa
- FEV1 <25% predicted
Features that should prompt correct timing for listing in COPD include:
- BODE index >7
- FEV1 <15-20% predicted
- Three or more severe exacerbations during the preceding year
- One severe exacerbation with acute hypercapnic respiratory failure
- Moderate-to-severe pulmonary hypertension
A relatively new modality, ex-vivo lung perfusion (EVLP), has recently become a clinical reality, with pre-clinical and clinical data from the Toronto group promulgating the science and its translation to the clinic.23,24 The technique involves taking a marginal donor lung (that would probably otherwise be unused and discarded), and putting it into a chamber where it is hooked up to a ventilator and to a perfusion pump where a blood substitute is perfused through the organ. The technique allows for a period of hours where the organ can be monitored for health and function and where interventions such as antibiotics and fluid removal can be accomplished, making an unusable organ usable for transplant. A pivotal report showed equivalent survival outcomes for patients who received EVLP lungs as those who received transplants through traditional transplant operations.24 As a result of 2 subsequent multi-center clinical trials in the United States (unpublished to date), the FDA has recently approved 2 different machines for clinical use. An upcoming clinical trial will also test the concept of a third party performing EVLP for transplant programs, utilizing 2 different cold ischemia times to make it possible for donor lungs to be treated several states away in some circumstances. EVLP offers the potential promise of increasing the limited supply of useable donor organs (important, as patients often die before a donor can be identified) and improving quality of the organs transplanted (as poorly functioning lungs can be identified before a transplant, and treatments to improve them can be utilized ex-vivo). Estimations are that this technology could increase donor supply by about 20%.
It remains uncertain whether lung transplantation confers a survival benefit to patients with COPD,25 but several studies have demonstrated improvements in quality of life and function.20,26,27 Survivors of transplant usually do not require supplemental oxygen and can return to normal activities and work.20 And while a majority of patients with COPD under evaluation for transplant claimed in a questionnaire to be seeking improved quality of life, many also hold an unrealistic expectation about likelihood of survival and underestimate the likelihood of morbidities.28 These points highlight the importance of robust communication and education of patients and families during the preparation for transplant.
Declaration of Interest
Dr. Make has participated in research studies and/or served on medical advisory boards for AstraZeneca, Boehringer-Ingelheim, GlaxoSmithKline, Novartis, Spiration, and Sunovion. Dr. Scuirba has received grants and research support from PneumRx and Pulmonx. Dr. Igenito is a full time employee of Amgen, Inc.