Running Head: Efficacy of IND/GLY in Dyspneic Patients with COPD
Funding support: Medical writing support was funded by Novartis Pharma AG (Basel, Switzerland).
Date of Acceptance: July 14, 2016
Abbreviations: Global initiative for chronic Obstructive Lung Disease, GOLD; chronic obstructive pulmonary disease, COPD; long-acting beta2-agonist, LABA; long-acting muscarinic antagonist, LAMA; indacaterol/glycopyrronium, IND/GLY; salmeterol/fluticasone propionate, SFC; Baseline Dyspnea Index, BDI; Transition Dyspnea Index, TDI; odds ratio, OR; forced expiratory volume in 1 second, FEV1; modified Medical Research Council, mMRC; inhaled corticosteroid, ICS; area under the curve from 0 to 12 hours, AUC0–12h; St. George’s Respiratory Questionnaire, SGRQ; forced vital capacity, FVC; electronic diary, eDiary; confidence interval, CI; least squares, LS; standard error, SE; standard deviation, SD; least-squares mean, LSM
Citation: Mahler DA, Keininger DL, Mezzi K, Fogel R, Banerji D. Efficacy of indacaterol/glycopyrronium in patients with COPD who have increased dyspnea with daily activities. Chronic Obstr Pulm Dis. 2016; 3(4): 758-768. doi: http://doi.org/10.15326/jcopdf.3.4.2016.0138
Introduction
Chronic obstructive pulmonary disease (COPD) is a heterogeneous disease.1 Within individual categories of airflow limitation (Global initiative for chronic Obstructive Lung Disease [GOLD]2 grades), high variability is observed in symptoms, health status, exercise capacity, and exacerbation frequency.1 In a factor-loading analysis of patients with (predominantly severe) COPD, variability in symptoms contributed more than any other variable in explaining disease heterogeneity.3 Therefore, patients with severe airflow limitation may not necessarily have a poor health status or a high symptom burden, and vice versa.1 Since studies indicate that only a modest correlation exists between airflow limitation, symptoms, and health status,4,5 GOLD suggests that post-bronchodilator forced expiratory volume in 1 second (FEV1) alone is an incomplete descriptor of COPD disease status, and does not adequately capture disease heterogeneity.2 For this reason, GOLD recommends a combined assessment of symptom severity, airflow limitation, and/or exacerbation risk for the effective evaluation of disease status in patients with COPD.2
Dyspnea is a principal symptom of COPD and a major cause of disability.2 Dyspnea severity can be measured using a range of tools in addition to the modified Medical Research Council (mMRC) questionnaire recommended by GOLD for the assessment of symptoms. The Baseline Dyspnea Index (BDI) and the Transition Dyspnea Index (TDI) are widely used for the measurement of dyspnea in clinical trials.6 The BDI and TDI provide multidimensional, interview-based measurements of breathlessness based on activities of daily living through the assessment of 3 components that provoke breathing difficulty: functional impairment, magnitude of task, and magnitude of effort.7,8 To establish baseline dyspnea severity using the BDI, each of the 3 components is assessed and scored on a scale from 0 (severe) to 4 (unimpaired), with the total BDI score out of 12 obtained through adding together the scores for each component.7 A lower BDI score therefore indicates a higher severity of dyspnea at baseline.7
BDI score correlates well with mMRC grade and other measures of dyspnea in patients with COPD,8,9 as well as with exercise capacity,7,10 quality of life,11 and mortality.12 However, BDI scores correlate only modestly (but significantly) with lung function.7Several comparisons of different dyspnea scales have indicated that the BDI has the highest levels of reliability and validity.9,13
The recommended treatment for patients with mild-to-moderate COPD who are experiencing dyspnea is monotherapy with a long-acting beta2-agonist (LABA) or long-acting muscarinic antagonist (LAMA).2 If the patient continues to experience dyspnea, or if dyspnea is severe, GOLD recommends the use of LABAs and LAMAs in combination.2
The purpose of this post-hoc analysis was to investigate the efficacy of fixed-dose LABA/LAMA combination QVA149 (indacaterol/glycopyrronium [IND/GLY] 110/50 µg) in patients with COPD who experienced moderate-to-severe dyspnea at baseline. Dyspneic patients were identified based on a BDI score ≤7. We primarily studied the patient-reported outcomes dyspnea and health status, with supportive analysis of lung function and rescue medication use.
Our aim was to confirm the efficacy of a LABA/LAMA in the treatment of dyspneic patients, as recommended in the GOLD treatment strategy, in comparison with commonly prescribed LAMA monotherapy tiotropium and LABA/inhaled corticosteroid (ICS) salmeterol/fluticasone propionate (SFC).
Methods
Study Design
This post-hoc analysis was conducted using data from 2 pivotal IND/GLY clinical studies, SHINE14 and ILLUMINATE.15 These studies were selected based on similarities in duration (26 weeks), endpoints studied (the primary endpoint in both studies was lung function), and patient population (both studies enrolled symptomatic patients with moderate-to-severe COPD; the majority of patients in both studies had moderate COPD). The studies were not combined for the analysis, but analyzed separately.
Full details of SHINE and ILLUMINATE have been published previously.14,15 Briefly, SHINE (NCT01202188) was a 26-week, randomized, controlled study comparing IND/GLY 110/50 µg with IND 150 µg, GLY 50 µg, open-label tiotropium 18 µg, and placebo, all administered once daily. ILLUMINATE (NCT01315249) was a 26-week, randomized study comparing once-daily IND/GLY 110/50 µg with twice-daily SFC 50/500 µg, both administered double-blind.
The primary endpoints in SHINE and ILLUMINATE were trough FEV1 for IND/GLY compared with IND and GLY, and FEV1 area under the curve from 0 to 12 hours (FEV1 AUC0–12h) for IND/GLY compared with SFC, respectively (both at 26 weeks). Secondary endpoints in both studies included TDI total score, daily rescue medication use, and St. George’s Respiratory Questionnaire (SGRQ) total score. Safety and tolerability were also assessed in both studies. In this post-hoc analysis, we studied the treatment differences with IND/GLY compared with placebo and established treatments tiotropium and SFC in patients with COPD who experience moderate-to-severe dyspnea at baseline. We focused on dyspnea and health status, with supportive analysis of lung function and rescue medication use. Exacerbations were not investigated as the studies analyzed were not optimized to determine meaningful changes in exacerbation rate.
Patients
SHINE and ILLUMINATE enrolled men and women aged ≥40 years, with a diagnosis of moderate-to-severe COPD (Stage II or III according to GOLD 2008 criteria), and with a smoking history of ≥10 pack years. At screening, patients were required to have a post-bronchodilator FEV1 ≥30% and <80% of the predicted normal value in SHINE, and a post-bronchodilator FEV1 ≥40% and <80% of the predicted normal value in ILLUMINATE. Patients enrolled in both studies had a post-bronchodilator FEV1/forced vital capacity (FVC) ratio <70%. Patients were required to be symptomatic at baseline in both studies, assessed using a patient-completed daily electronic diary (eDiary). Symptoms were recorded during a 14-day run-in period and were measured on a scale of 0 to 3 (where 0 refers to no symptoms). Patients with a total symptom score ≥1 on 4 or more of the last 7 days prior to Visit 3 (Day 1) were classified as symptomatic. Patients with a COPD exacerbation requiring treatment with antibiotics, systemic corticosteroids or hospitalization were excluded from SHINE if the exacerbation occurred during the 6 weeks prior to the study, and from ILLUMINATE if the exacerbation occurred within 1 year prior to the study. Patients with a respiratory tract infection within the 4 weeks prior to Visit 1 were excluded from both studies. Concomitant ICS use (in constant doses and dose regimens for at least 1 month) was permitted in SHINE, but not in ILLUMINATE. Patients were provided with a salbutamol inhaler for use as rescue medication during both studies.
Assessments and Variables
In SHINE and ILLUMINATE, dyspnea was measured using the BDI at baseline and the TDI at Weeks 12 and 26. Health status was measured using the SGRQ at baseline and at Weeks 12 and 26 in both studies. In SHINE, FEV1 was assessed at baseline, Days 1 and 2, and Weeks 2, 4, 8, 12, 16, 20, and 26. In ILLUMINATE, FEV1 was assessed at baseline, Day 1, and Weeks 6, 12, 18, and 26. Use of rescue medication in the 2 studies was measured over the 26-week treatment period using an eDiary.
BDI Stratification
Studies suggest that a BDI score ≤7 correlates with an mMRC grade ≥2, the GOLD cut-off for symptomatic patients.2,9,10,16 Patients with a BDI score ≤7 were therefore selected as a subgroup to represent patients with moderate-to-very severe dyspnea. Patients with a BDI score ≤7 have moderate-to-very severe functional impairment and experience shortness of breath when exerting moderate-to-no effort and when carrying out few tasks.7 Data from the wider stratifications of a BDI score ≤6 and ≤8 were also analyzed, to provide further information on the efficacy of IND/GLY in patients who had severe dyspnea at baseline.
Statistical Analysis
Statistical analyses were performed using SAS statistical software version 9.3 (SAS Institute, Cary, North Carolina). Main demographic parameters were analyzed by means of descriptive statistics. TDI, change in trough FEV1, mean rescue medication use (puffs/day), and SGRQ total score were assessed by means of a mixed model with treatment, baseline FEV1, FEV1 reversibility components, baseline smoking status, baseline ICS use, region, and center (region) as covariates. The differences in the proportion of patients with a 1-unit improvement in TDI total score17 and the proportion of patients with a 4-unit improvement in SGRQ total score18 were assessed via logistic regression, using the same covariates as for the other efficacy variables. For this post-hoc analysis, last observation carried forward values at 26 weeks were used.
Results
Patients
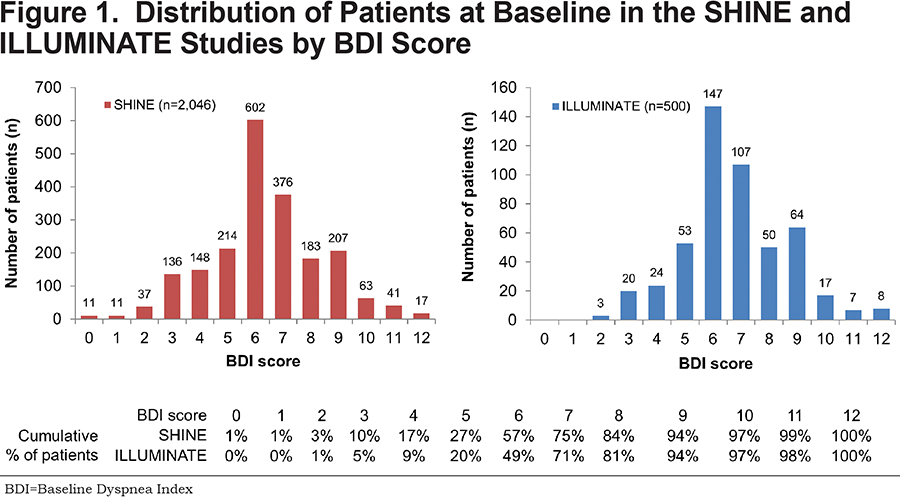
Distribution of BDI scores in SHINE and ILLUMINATE indicate that most patients were at least moderately symptomatic based on BDI score (Figure 1).
Patient baseline demographics and characteristics of the total study populations and BDI ≤7 subgroups are displayed in Table 1.
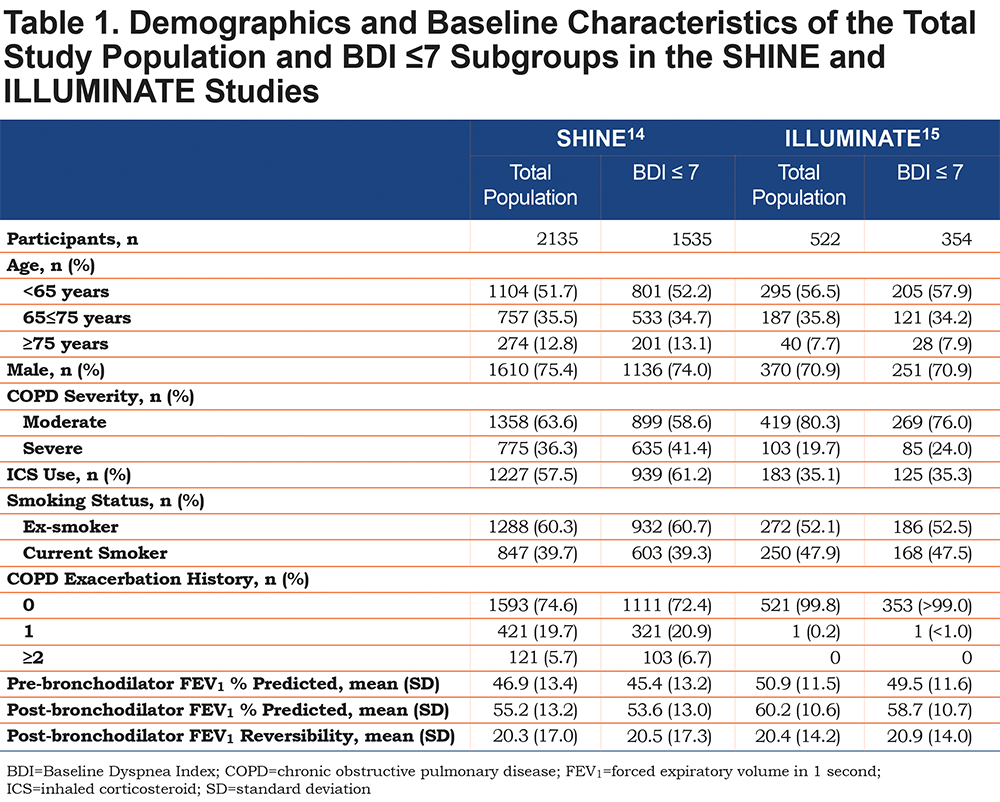
Seventy-five percent of patients in the total SHINE population and 71% of patients in the total ILLUMINATE population had a BDI score ≤7. The total study populations and BDI ≤7 subgroups were similar in terms of age, sex, and smoking status.A slightly higher proportion of patients in the BDI ≤7 subgroup had severe COPD compared with the total population for both SHINE (41.4% and 36.3%, respectively) and ILLUMINATE (24.0% and 19.7%, respectively). Mean post-bronchodilator FEV1 was similar between the total population and the BDI ≤7 subgroup for both SHINE (55.2% and 53.6%, respectively) and ILLUMINATE (60.2% and 58.7%, respectively).
Approximately half of the patients in both SHINE and ILLUMINATE had a BDI score ≤6, and 84% of patients in SHINE and 81% of patients in ILLUMINATE had a BDI score ≤8.
Efficacy in the BDI ≤7 Subgroups
Dyspnea
TDI Total Score
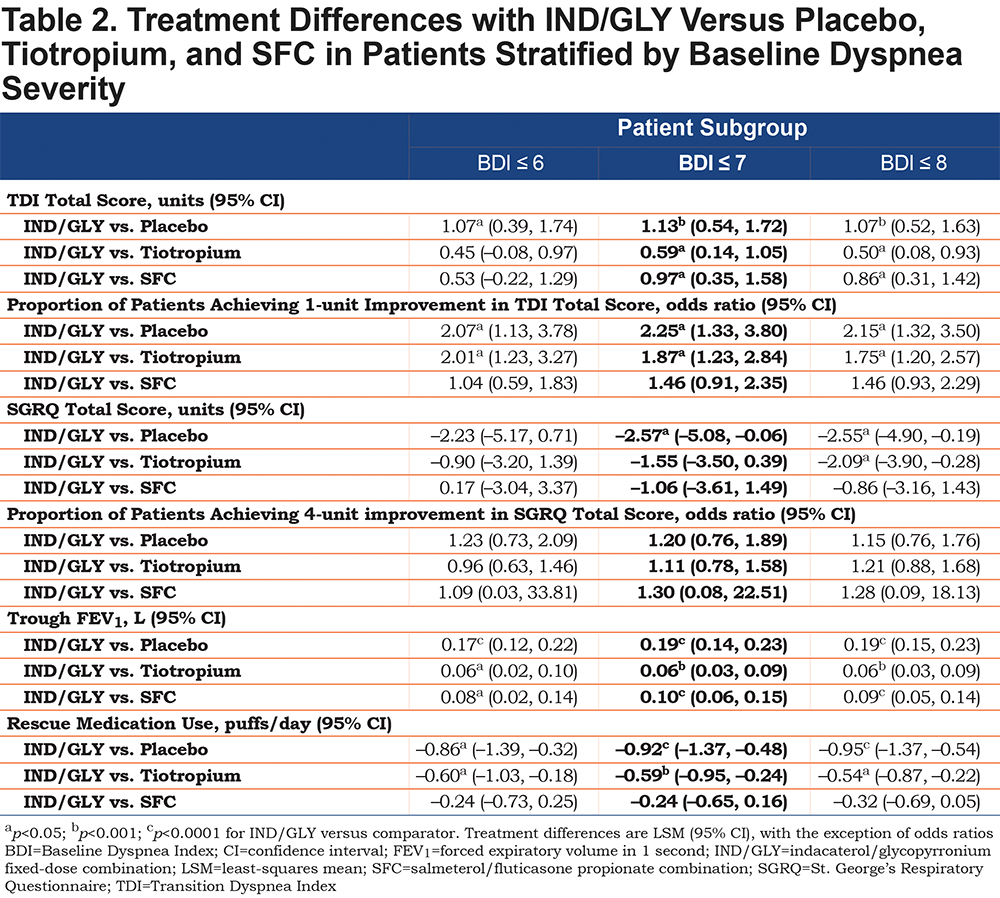
IND/GLY significantly improved dyspnea compared with placebo, tiotropium, and SFC, with treatment differences of 1.13 units (p<0.001), 0.59 units (p<0.05), and 0.97 units (p<0.05), respectively (Figure 2 and Table 2).
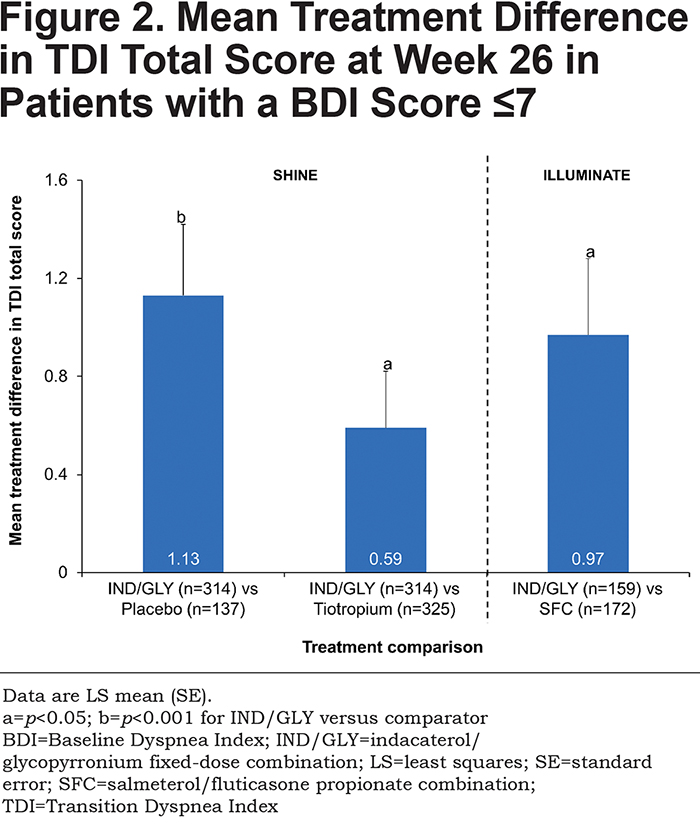
TDI Responders
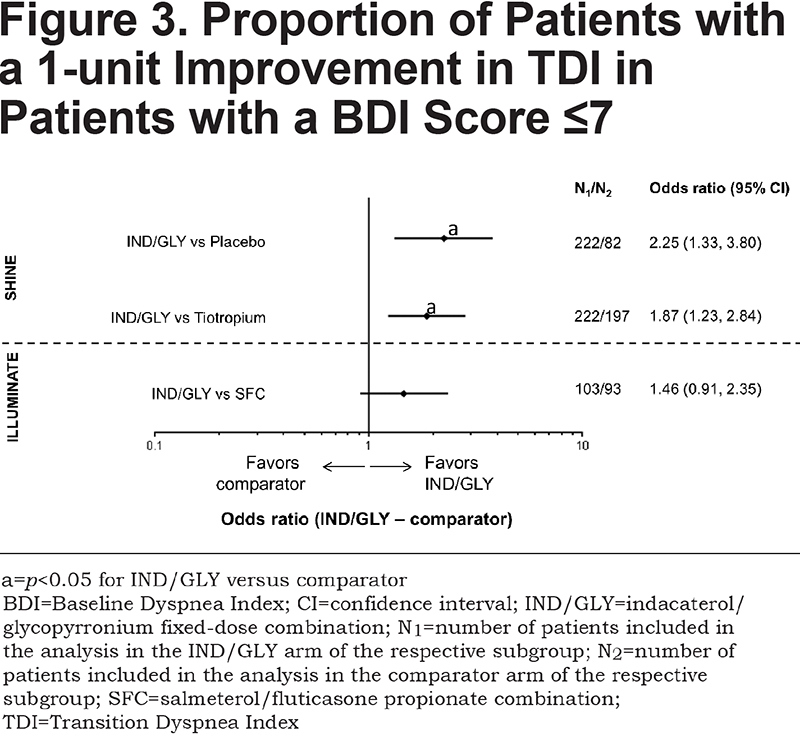
IND/GLY significantly increased the proportion of patients achieving a 1-unit improvement in TDI total score compared with placebo (odds ratio [OR] 2.25; 95% confidence interval [CI] 1.33, 3.80 [p<0.05]) and tiotropium (OR 1.87; 95% CI 1.23, 2.84 [p<0.05]) (Figure 3 and Table 2). There was a non-significant, numerical difference in the proportion of patients achieving a 1-unit improvement in TDI total score with IND/GLY compared with SFC (OR 1.46; 95% CI 0.91, 2.35 [p=0.117]).
Health Status
SGRQ Total Score
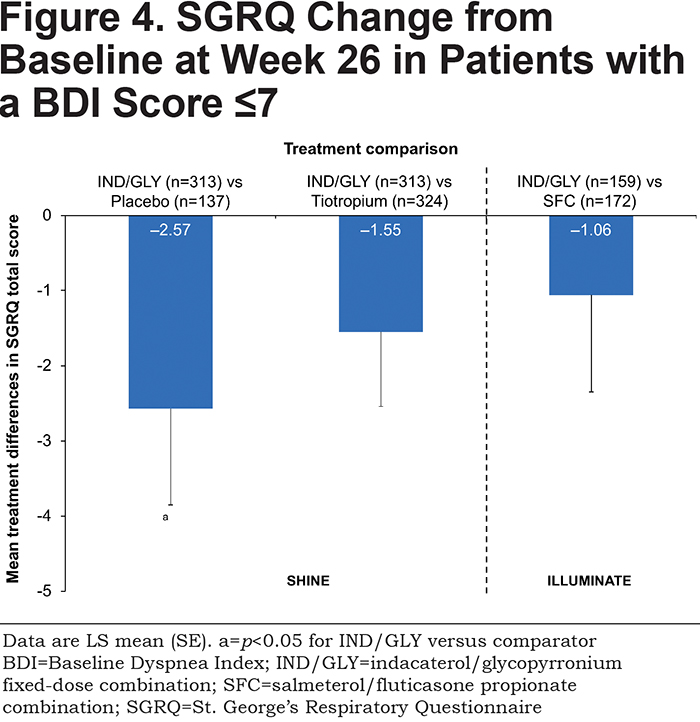
IND/GLY significantly improved SGRQ total score compared with placebo, with a treatment difference of –2.57 units (p<0.05; Figure 4 and Table 2).There was a non-significant, numerical difference in SGRQ total score between IND/GLY and tiotropium (treatment difference: –1.55 units [p=0.117]) and IND/GLY and SFC (treatment difference: –1.06 [p=0.414]).
SGRQ Responders
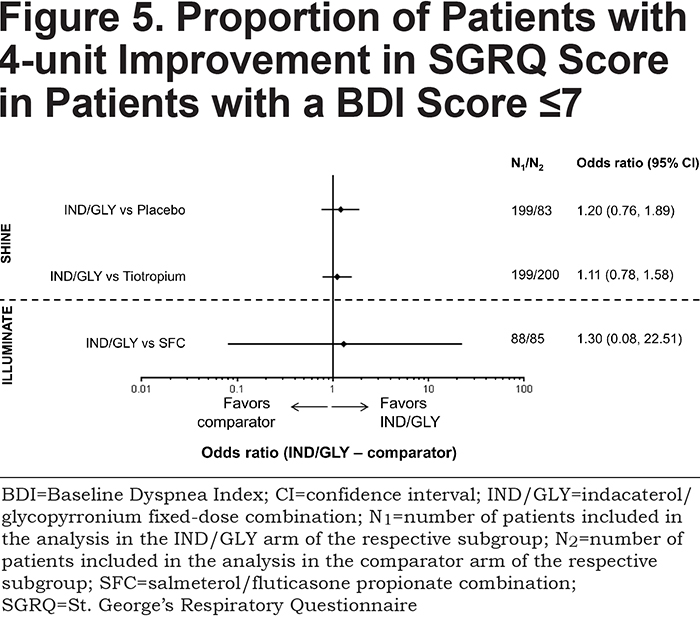
There was no significant difference in the proportion of patients achieving a 4-unit improvement in SGRQ total score with IND/GLY compared with placebo (OR 1.20; 95% CI 0.76, 1.89 [p=0.425]), tiotropium (OR 1.11; 95% CI 0.78, 1.58 [p=0.570]) or SFC (OR 1.30; 95% CI 0.08, 22.51 [p=0.451]) (Figure 5 and Table 2).
Lung Function
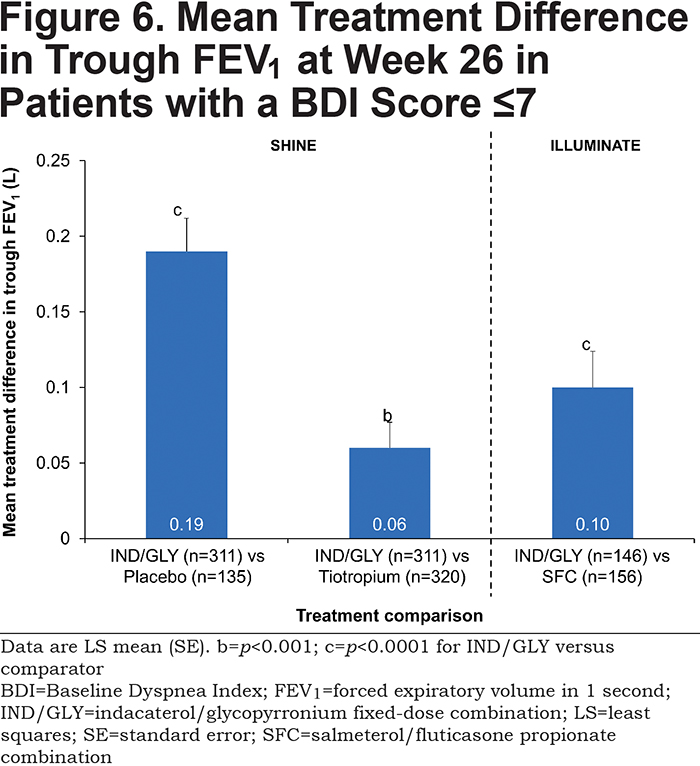
IND/GLY significantly improved trough FEV1 compared with placebo, tiotropium, and SFC, with treatment differences of 0.19 L (p<0.0001), 0.06 L (p<0.001), and 0.10 L (p<0.0001), respectively (Figure 6 and Table 2).
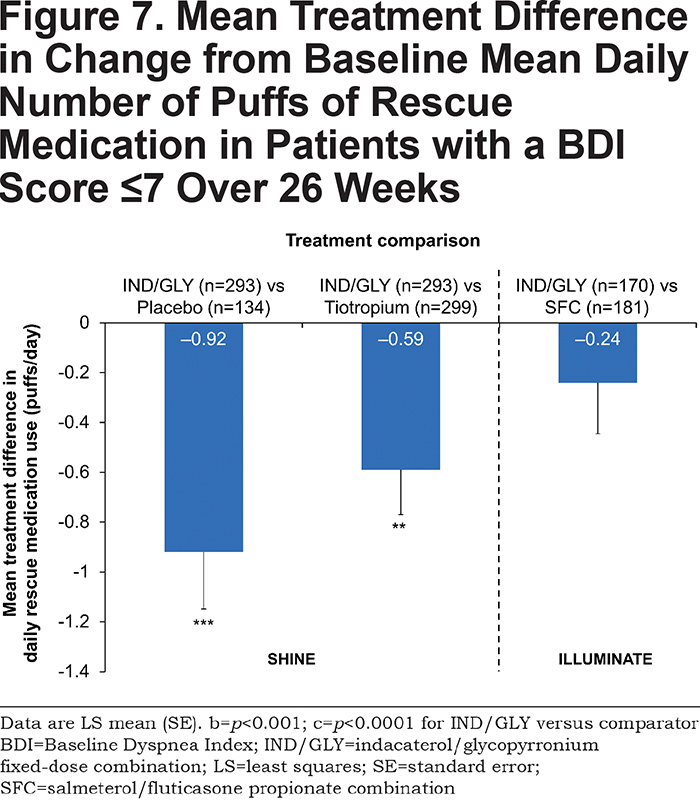
Rescue Medication Use
IND/GLY significantly improved rescue medication use compared with placebo and tiotropium, with treatment differences of –0.92 puffs/day (p<0.0001) and –0.59 puffs/day (p<0.001), respectively (Figure 7 and Table 2). There was a non-significant, numerical difference in rescue medication use between IND/GLY and SFC (treatment difference: –0.24 puffs/day [p=0.235]).
Efficacy in the BDI ≤6 and BDI ≤8 Subgroups
The results for the BDI ≤6 and BDI ≤8 stratifications were largely similar to those for the BDI ≤7 subgroups, for all endpoints studied (Table 2). However, the BDI ≤7 subgroup demonstrated the biggest effects in TDI score, proportional with TDI improvement and trough FEV1.
Discussion
This post-hoc analysis indicates the relative efficacy of IND/GLY compared with placebo, tiotropium, and SFC in patients with moderate-to-severe dyspnea at baseline, based on a BDI score ≤7.According to the BDI questionnaire, patients with a BDI score ≤7 have moderate-to-very severe functional impairment and experience shortness of breath with moderate-to-no effort and when carrying out moderate-to-no tasks at all.7In these patients, symptoms can affect ability to work and carry out normal activities.7
In dyspneic patients with mild-to-moderate airflow limitation and no exacerbation history (GOLD Group B), monotherapy with a long-acting bronchodilator provides significant improvements in dyspnea, lung function, and rescue medication use compared with placebo.19 However, some patients continue to experience dyspnea despite receiving bronchodilator monotherapy.20,21 According to GOLD, a combination of long-acting bronchodilators may be considered if symptoms such as dyspnea are not improved with monotherapy.2 Although analyses based on severity of airflow limitation were not performed, the majority of dyspneic patients in our analysis had moderate airflow limitation and no exacerbation history, and would therefore be categorized as GOLD Group B.
Our post-hoc analysis indicates that the LABA/LAMA combination IND/GLY was more effective than tiotropium monotherapy at improving dyspnea, lung function, and rescue medication use in dyspneic patients with COPD.The findings support the results of the 6-week BLAZE cross-over study, which demonstrated that IND/GLY significantly improved dyspnea, lung function, and rescue medication use compared with placebo and tiotropium in dyspneic patients (patients with an mMRC grade ≥2 at screening).22
Post-hoc analysis of the TORCH study indicates that pharmacotherapy is most effective in patients with milder disease, compared with placebo.23 A considerable amount of data from clinical trials demonstrates that LABA/LAMAs as a class provide benefits beyond placebo and long-acting bronchodilator monotherapy with regard to lung function and patient-reported outcomes.14,24-29 In addition, IND/GLY has demonstrated significant improvements in lung function compared with placebo, independent of prior medication.30 We therefore suggest that the use of LABA/LAMA combinations be considered earlier in the course of disease, or even as first-line therapy.
LABA/ICS combinations are not recommended for use in patients in GOLD Group B, as the risks associated with ICS appear to outweigh the benefits of such treatment in patients at low risk of exacerbation.2 However, ICS use in GOLD Group B patients is widespread.20,31-36 In the dyspneic patients studied in our analysis,IND/GLY was significantly better than SFC at improving dyspnea and lung function, providing supportive evidence for the efficacy of a dual bronchodilator compared with a LABA/ICS in the treatment of GOLD Group B patients.
Analysis of patients with a BDI score ≤6 and ≤8 indicates that the relative efficacy of IND/GLY is largely consistent in patients with slightly worse or slightly milder dyspnea, respectively. However, the largest improvement in TDI was seen in the ≤7 subgroup. This possibly could be attributed to decreased room for improvement in the ≤8 group, and the ≤6 group being a smaller subgroup of patients.
The magnitude of response in terms of dyspnea improvement was slightly larger in the BDI ≤7 subgroups compared with the total study populations.14,15 The improvement in TDI total score observed with IND/GLY compared with SFC in the BDI ≤7 subgroup was 0.97 units; in the total ILLUMINATE population, improvement in TDI total score was 0.76 units.15In addition, improvement in TDI total score observed with IND/GLY compared with tiotropium in the BDI ≤7 subgroup was 0.59 units; in the total SHINE population, improvement in TDI total score was 0.51 units.14 Of interest, more patients in the BDI ≤7 subgroups had severe COPD compared with the total populations.In BLAZE, the magnitude of response in terms of dyspnea improvement was greater in patients with severe COPD than those with moderate COPD.22
Previous analyses have indicated that pharmacotherapy may be less effective in patients with severe-to-very severe COPD than in patients with milder COPD in terms of lung function improvements.23 If lung function improvements correlate with improvements in dyspnea, then it might be expected that pharmacotherapy would be less effective at improving dyspnea in patients with more severe disease, which is inconsistent with our findings. The correlation between lung function and symptoms has previously been identified as modest.4,5 Our findings may support the need for composite endpoints to more fully assess treatment efficacy in patients with COPD.37
In our analysis, the majority of the improvements observed with IND/GLY compared with tiotropium and SFC did not reach commonly accepted minimal clinically important differences. However, it may be argued that it is not realistic for comparisons between active treatments to achieve MCID values determined using data versus placebo.38 Instead, responder analyses may be a more appropriate measure of a clinically relevant improvement when comparing active treatments, or when adding one treatment to another.38 The proposed term for this parameter is “minimum worthwhile incremental advantage.”38
It is important to note that there were some limitations in the present post-hoc analysis. A similar analysis has already been carried out on the whole population; however, this post-hoc analysis adds to the field as it specifically analyzes patients with dyspnea. GOLD recommends LABA/LAMA as an alternative to monotherapy in patients with mild-to-moderate airflow limitation and symptoms.2 Subgroup analyses can result in small patient numbers in certain subgroups. For instance, there were fewer than 200 patients in the IND/GLY and SFC treatment arms in the BDI ≤7 subgroup in ILLUMINATE. Therefore, there may be a lack of power in these subgroups to detect differences, and the results should be interpreted with caution. In addition, subgroup analyses based on severity of airflow limitation were not conducted and therefore the patient populations do not model exactly to the GOLD classifications, which may limit the conclusions that may be drawn.
Conclusion
Our analysis indicated that IND/GLY provides significant benefits compared with tiotropium monotherapy in patients with COPD who are experiencing dyspnea. These data raise the consideration of using a LABA/LAMA combination earlier in the treatment algorithm, and even possibly as first-line therapy, in symptomatic patients categorized as GOLD Group B. Our analysis also indicated that IND/GLY was at least as effective as SFC in improving dyspnea and lung function, in a patient population in whom ICS use is not recommended due to the limited benefits and associated risks.
Acknowledgments
The authors were assisted in the preparation of the manuscript by Elizabeth Andrew, a professional medical writer at CircleScience, an Ashfield Company, part of UDG Healthcare plc (Tytherington, United Kingdom). Medical writing support was funded by Novartis Pharma AG (Basel, Switzerland).
Declaration of Interest
Donald Mahler has received consulting fees for advisory boards from Boehringer Ingelheim, GlaxoSmithKline, Novartis, Sunovion, and Theravance. He receives royalties from CRC Press, from Hillcrest Media Group Inc, and from MAPI Research Trust. He is on speakers’ bureaus for Boehringer Ingelheim, GlaxoSmithKline, and Sunovion. His website (http://www.donaldmahler.com) is an educational website for those with COPD and their families. Dorothy Keininger, Karen Mezzi, Robert Fogel, and Donald Banerji are full-time employees of Novartis. Robert Fogel and Donald Banerji own stock in Novartis.