Running Head: Asthma & COPD Overlap: Definitions, Measures
Funding Support: This study was funded by GlaxoSmithKline (Study number WEUSKOP7057).
Date of acceptance: Oct 28, 2016
Abbreviations: asthma-COPD overlap syndrome, ACOS; National Health and Nutrition Examination Survey, NHANES; odds ratio, OR; confidence interval, CI; Global Initiative for Asthma, GINA; Global initiative for chronic Obstructive Lung Disease, GOLD; forced expiratory volume in 1 second, FEV1; forced vital capacity, FVC; fractional exhaled nitric oxide, FeNO; parts per billion, ppb; body mass index, BMI; lower limit of normal, LLN
Citation: Mannino DM, Gan WQ, Wurst K, Davis KJ.Asthma and chronic obstructive pulmonary disease overlap: The effect of definitions on measures of burden. Chronic Obstr Pulm Dis. 2017; 4(2): 87-96. doi: http://doi.org/10.15326/jcopdf.4.2.2016.0159
Online Supplemental Material: Read Online Supplemental Material (69 KB)
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are 2 distinct and common respiratory diseases that share both common risk factors and clinical presentations.1 Although the overlap between asthma and COPD in population-based databases has been recognized for years,2-4 this overlap has only recently been given a name, asthma-COPD overlap syndrome (ACOS) and become better defined.1,5-11 The Global Initiative for Asthma (GINA) and the Global initiative for chronic Obstructive Lung Disease (GOLD) have recently released guidance for the classification of asthma, COPD, and ACOS.12 These guidelines have been the focus of recent review articles.13,14
In a recent review, Tho et al14 highlighted some of the challenges related to the definition and estimate of disease burden of ACOS. Chief among these were the precise definition of asthma, COPD, and ACOS. Prevalence estimates of ACOS in the general population ranged from 1.8%15 to 2.7%,16 with the former estimate based on spirometric criteria and the later estimate based on self-reporting of a physician diagnosis. Among populations of patients with clinical asthma, the estimated prevalence of concurrent COPD ranged from 16% to 61%,17,18 and among those with clinical COPD, the prevalence of current asthma ranged from 5% to 55%.19,20
This analysis used data from the National Health and Nutrition Examination Survey (NHANES) from 2007-2012 to determine population estimates of ACOS in U.S. adults using 2 different definitions of the syndrome, and to determine how other factors, such as lung function impairment and health care utilization, varied by ACOS definitions.
Methods
NHANES is a cross-sectional survey of the civilian, non-institutionalized U.S. population conducted by the National Center for Health Statistics of the Centers for Disease Control and Prevention. Data were collected via household interviews and standardized physical examinations in specially equipped mobile examination centers. The NHANES survey samples are selected through a complex, multi-stage, probability design, and each 2-year sample is nationally representative. The procedures to select the sample and conduct the interview and examination have been specified elsewhere.21 Informed consent was obtained from all participants and the National Center for Health Statistics Research Ethics Review Board approved the protocol.
The response rates for NHANES were as follows: 78% for the interview, and 75% for examination in the 2007-2008 survey; 79% and 77% in the 2009-2010 survey; and 73% and 70% in the 2011-2012 survey. NHANES 2007-2012 included spirometry of individuals aged 6-79 years. We limited our analysis to adults aged 20-79 years. Spirometry was obtained using an Ohio 822/827 dry-rolling seal volume spirometer following similar protocols across surveys. NHANES reported pre-bronchodilator values of forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) in ml. A subset of individuals (those with obstruction present and no contraindication) qualified for the administration of a bronchodilator (albuterol) and had spirometry repeated afterwards. The FEV1 and FVC predicted values were computed using predictive equations.22
Definitions
Current asthma was defined by answering yes to the following 2 questions: “Has a doctor ever told you that you have asthma?” and “Do you still have asthma?” Self-reported COPD was defined by answering yes to either of the following 2 questions: “Has a doctor ever told you had emphysema?” or “Do you still have chronic bronchitis?” Spirometry-confirmed COPD was defined as having an FEV1/FVC <0.70, based on pre-bronchodilator lung function. (This is technically spirometric obstruction but will be referred to as spirometry-confirmed COPD for simplification).
We evaluated 2 ACOS definitions: ACOS1 was self-reported COPD and current asthma; ACOS2 was spirometrically-confirmed COPD (FEV1/FVC < 70%) and current asthma.
Lung function outcomes were calculated based on pre-bronchodilator measurements, unless otherwise indicated and included: mean FEV1 as percentage of predicted, mean FEV1/FVC, proportion not having spirometry (for those defined using clinical criteria), proportion with obstruction (FEV1/FVC < 0.70), proportion with restriction (FEV1/FVC ≥ 0.70 and FVC < 80%), and proportion who were administered a bronchodilator (only a subset of participants who had spirometry were eligible [based on the presence of obstruction and no clinical contraindications] to be administered a bronchodilator and of those eligible, only a subset actually had a bronchodilator). Bronchodilator response was the mean percentage improvement in FEV1 from baseline and significant bronchodilator response was the percentage with a significant response (> 12% and > 200ml in FEV1 or FVC). We also evaluated eosinophil count as an absolute count and as a percentage ≥ 2% of the total white blood cell count and fractional exhaled nitric oxide (FeNO) in parts per billion (ppb).
The following outcomes were evaluated:
- Health care visits over past year, “During the past 12 months, how many times have you seen a doctor or other health care professional about your health at a doctor's office, a clinic, hospital emergency department, at home or some other place?”
- Overnight hospital stays over past year, “During the past 12 months, were you a patient in a hospital overnight?”
- Number of overnight hospital stays over past year, “How many different times did you stay in any hospital overnight or longer during the past 12 months?”
- Respiratory prescription medication use over past month, “In the past month have you used or taken medication for which a prescription is needed?”
- Wheezing medication use in past year, “In the past 12 months, have you taken medication prescribed by a doctor for wheezing or whistling?”
- Frequency of health care for wheezing attacks over past year, “In the past 12 months, how many times have you gone to the doctor's office or the hospital emergency department for one or more of these attacks of wheezing or whistling?”
- Frequency of wheezing or whistling attacks over past year, “In the past 12 months, how many attacks of wheezing or whistling have you had?”
- Missed work or school days due to wheezing over past 12 months, “During the past 12 months, how many days of work or school did you miss due to wheezing or whistling?”
Covariates included age (20-29, 30-39, 40-49, 50-64 and >65 years), gender (male, female), body mass index (BMI) (<18.5 kg/m2 [underweight], 18.5-24 kg/m2 [normal], 25-29 kg/m2 [overweight], ≥ 30 kg/m2 [obese]), race/ethnicity (non-Hispanic white, non-Hispanic black, Mexican American, and other), education attainment (less than high school, high school, and more than high school), cigarette smoking status (current smokers [had smoked ≥100 cigarettes in lifetime and still smoked at the time of interview], former smokers [had smoked ≥100 cigarettes in lifetime but did not smoke at the time of interview] and never smokers [had not smoked ≥100 cigarettes in lifetime]).
All analyses were conducted with SAS version 9.3 (SAS Institute, Cary, North Carolina) and SUDAAN version 10.0 (RTI, Research Triangle Park, North Carolina).
The descriptive analyses compared the different definitions of ACOS with regard to the outcomes of respiratory symptoms, lung function, and health care utilizations. The multivariate analyses examined predictors of ACOS (using the different ACOS definitions). Additional multivariate analyses examined how the various outcomes (respiratory symptoms, lung function, and health care utilizations) were related to the different ACOS definitions and their components (asthma and COPD).
All analyses were performed according to NHANES standards using the appropriate weights (typically, the mobile examination center final weights combined across the study periods included) to account for the complex sampling scheme and to generate estimates representative of the U.S. non-institutionalized population.
Results
Overall 16,526 adult participants in NHANES, representing 211.7 million U.S. adults, had data available to determine self-reported COPD and current asthma. Of these, 12,964, representing 174.2 million U.S. adults, had spirometry available and comprised the population used to determine spirometrically-confirmed COPD and current asthma.
In the total analytic population, 7.3% reported current asthma, 3.9% reported COPD, and 1.6% reported both (Figure 1). Using the definition of ACOS1, the proportions corresponded to a population estimate of 3.4 million ACOS-affected adults (Table 1). Of the 14.3 million adults with current asthma, 23.8% had ACOS1, whereas among the 8.3 million adults reporting COPD, 41.0% had ACOS1 (Figure 1A).

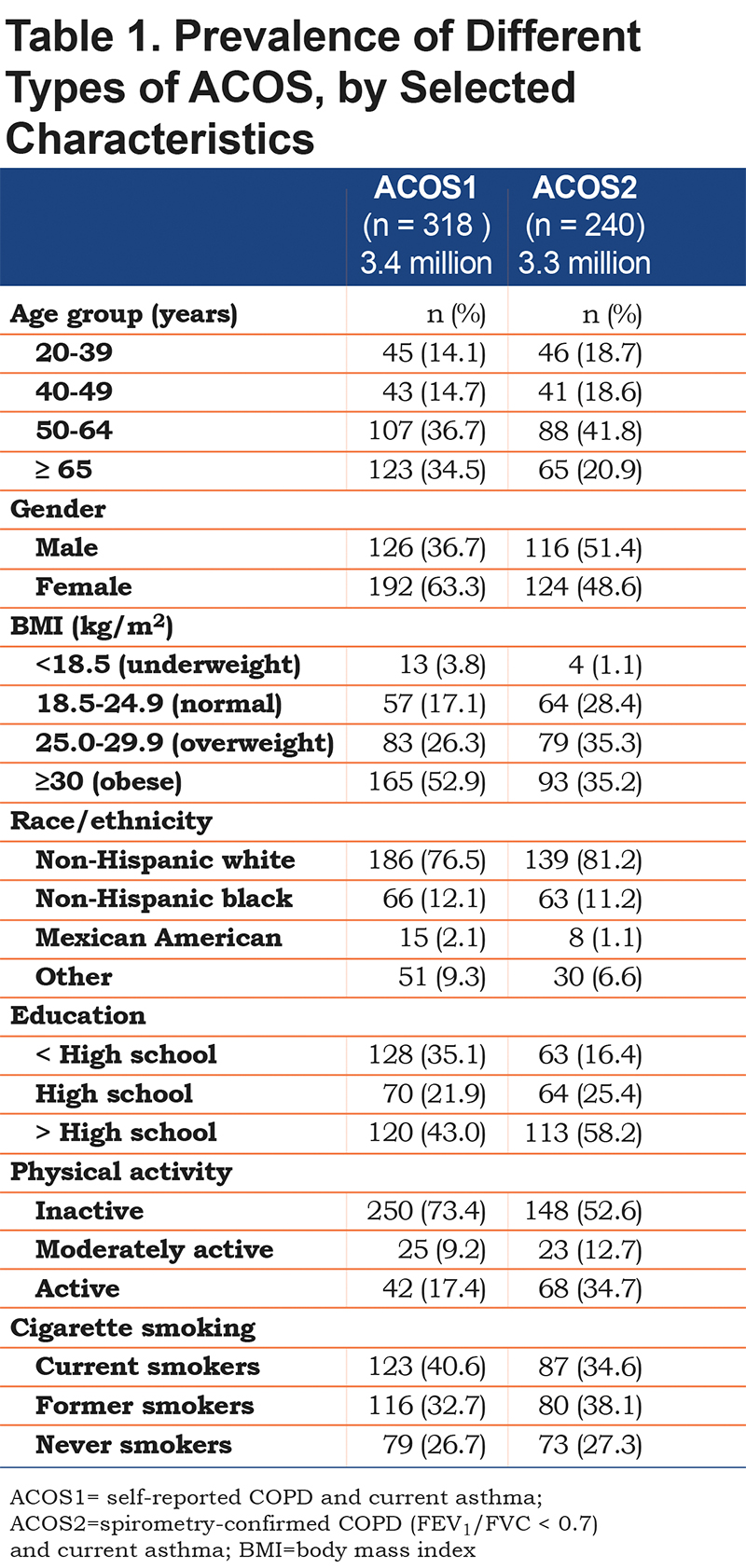
In the population with spirometry measurements, 6.9% reported asthma and 13.6% had spirometry-confirmed COPD (FEV1/FVC < 0.7); 1.9% of the population had both, corresponding to 3.3 million adults (Figure 1B, Table 1). Of the 12.1 million adults reporting current asthma, 27.5% had ACOS2 using this definition, and among the 23.7 million with spirometrically-confirmed COPD, 14.0% had ACOS2 (Figure 1B).
Tables 1 and 2 display the variation in demographic factors by the 2 different ACOS case definitions. A higher age, current or former smoking status, and race were consistently associated with ACOS across both definitions. Being female, obese, inactive, or having less than a high school education were associated with ACOS in the non-spirometrically defined population (ACOS1, Table 2).

Table 3 examines spirometric outcomes in the entire population for ACOS1. Of note, only 82.3% of the population sampled had spirometry measurements, although this proportion was significantly lower in people with either ACOS or COPD (odds ratio [OR] 0.5, 95% confidence interval [CI] 0.4, 0.6 and OR 0.7, 95% CI 0.5, 0.9, respectively). Of those who had spirometry, 44.2% of those with ACOS1 had obstruction and an additional 17.8% had restriction (Table 4), yet only 1.9% had a significant response to a bronchodilator. Confidence intervals are available in the online supplement, eTable 1.
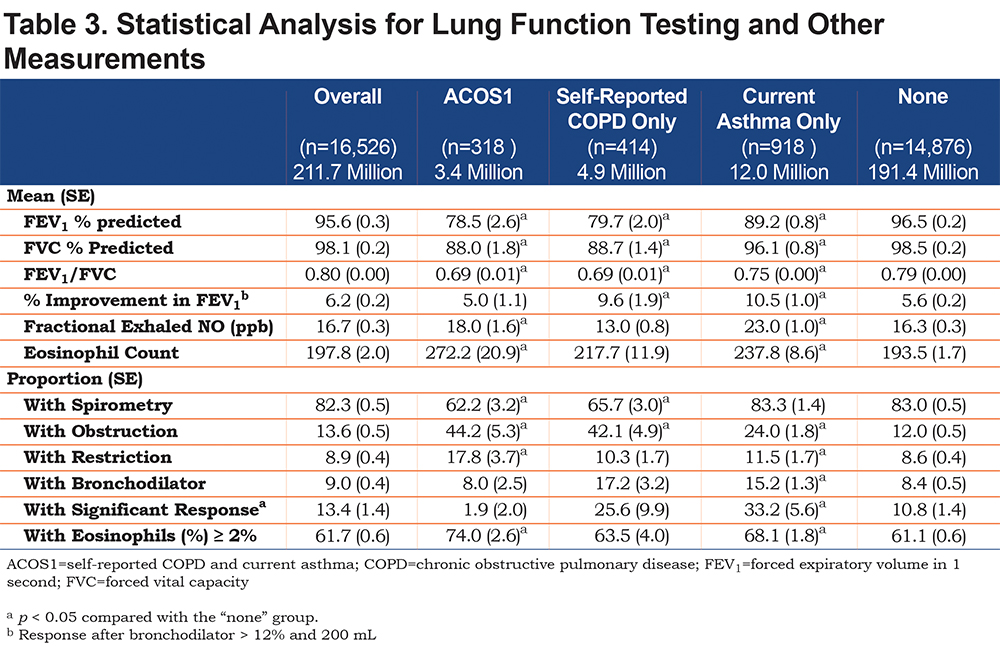
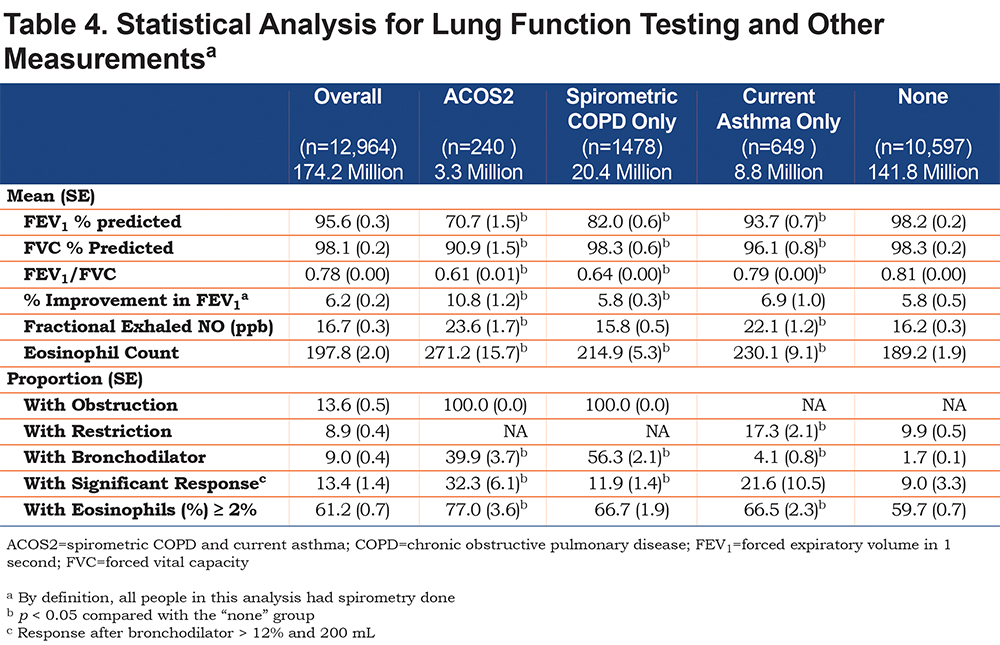
Table 4 examines spirometric outcomes in the population that had spirometry (which was required for the ACOS2 definition). Confidence intervals are available in the online supplement, eTable 2.
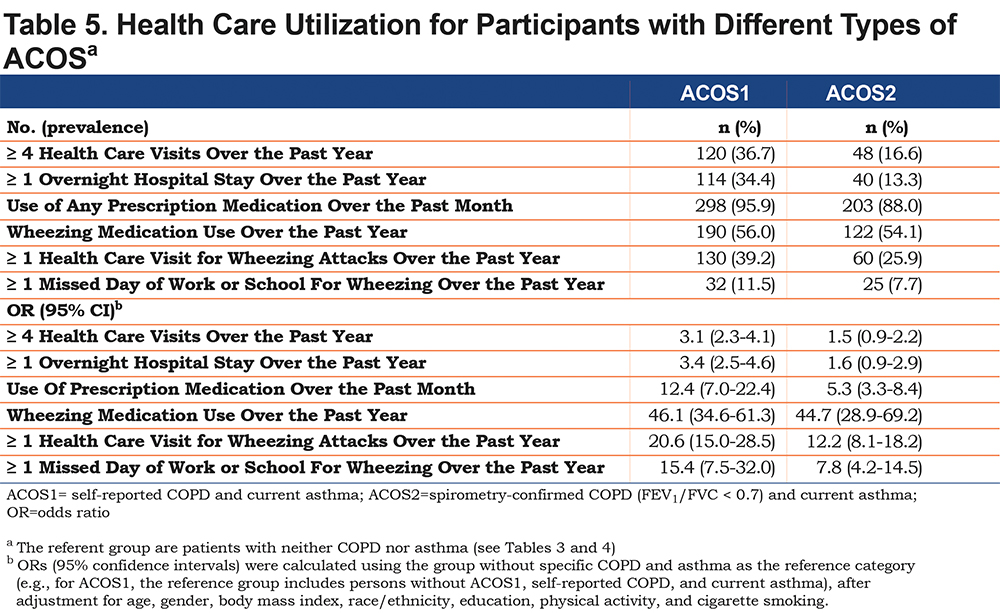
Finally, Table 5 shows health care utilizations in the 2 ACOS groups. The measures of burden (>4 health care visits or >1 overnight hospital stay were significantly higher in ACOS1 than in ACOS2).
Discussion
We used NHANES data from 2007-2012 to calculate estimates of the population-based prevalence of ACOS among U.S. adults using 2 different case definitions: ACOS1, self-reported COPD and current asthma; ACOS2, spirometric-confirmed COPD (FEV1/FVC < 70%) and current asthma. While these 2 definitions gave population estimates of disease burden that were similar in magnitude (1.6% for ACOS1 and 1.9% for ACOS2), this analysis highlighted how different these populations were with respect to clinical characteristics and outcomes. Further, it highlighted some of the pitfalls of using national survey data like NHANES for this type of analysis.
Our overall estimates of ACOS disease prevalence of 1.3% to 1.9% are comparable to the ranges reported in other population-based studies of 1.8%,15 2.3%23 and 2.7%.16 Similarly, the range of estimated COPD among patients with asthma was 19.0% to 27.5%, comparable to the estimate of 29% reported by Milanese et al18 and the estimates of 16%-30% reported by de Marco for patients 20-44 and 45-64,24 but less than the 61% reported by de Marco for 65-84 year olds.24 Among those with COPD (using the various definitions), we found 14.0% to 41.0% reported comorbid asthma. Other previous studies report a very wide range for asthma prevalence among those with COPD from 5.0%19 to 55%.20 This large difference in estimates is a function of the definition used. For example, the previous studies reporting lower estimates19,25 had fairly stringent criteria for asthma, such as a bronchodilator response of > 400 mL or elevated FeNO whereas the higher estimates used a physician diagnosis of asthma in conjunction with current symptoms.20 In our analysis, the highest estimate of comorbid asthma (41.0%) was observed in those with self-reported COPD (ACOS1).
Moving from a case definition based only on reported disease (ACOS1) to one that incorporated the objective measurement of lung function (ACOS2) changed several correlates of disease. For example, women were nearly twice as likely as men to have ACOS1 (2.0% versus 1.2%). This gender difference may explain why a higher proportion of those in ACOS1 reported symptoms relative to those in ACOS2, since women are generally more likely to report symptoms than men.26,27 One of the biggest differences between the case definitions was the requirement for spirometry in ACOS2. People with ACOS1 were significantly less likely to have spirometry measurements included than those with neither self-reported COPD nor current asthma (OR 0.5, 95% CI 0.4, 0.6). This discrepancy was due to the fact that the NHANES protocol specifically excluded people with known lung disease from having spirometry tests.28 Thus, in this database, those who had the worse lung disease would be excluded from the ACOS2 dataset. This bias would, again, explain why most utilization was higher in the ACOS1 group relative to the other groups (i.e., individuals with more severe impairment were excluded from having spirometry performed). Our estimated percentage of the population with ACOS2 based on pre-bronchodilator spirometry was 1.9% (3.3 million). Based on a post-bronchodilator spirometry (data not shown) this would be 0.4% (0.7 million), although most of this reduction is related to the absence of bronchodilator administration (1.1%, 2.0 million) and the rest is related to the bronchodilator response (0.4%, 0.6 million). Another bias was that in NHANES, bronchodilators were only administered to people who were obstructed. Thus, it was not possible to look at bronchodilator response in individuals with asthma who were not obstructed. Finally, we used the fixed ratio (FEV1/FVC < 0.70) rather than the lower limit of normal (LLN) to define obstruction. A separate analysis using the LLN (data not shown) showed the prevalence of ACOS2 to remain unchanged at 1.9%.
Another potential bias is related to health care utilization and diagnoses: people with higher utilization rates have more opportunities to receive diagnoses.29 The absence of stringent criteria for the diagnosis of asthma and COPD in clinical practice (and the reality that many are diagnosed in the absence of meeting clinical criteria) adds to the potential for this diagnostic bias.30,31
Our findings are subject to a number of limitations inherent with this cross-sectional study of the non-institutionalized U.S. population. Surveys such as NHANES may not capture the sickest segment of the population if they are unable or unwilling to participate in a survey including clinical examinations; such a selection bias could potentially underestimate the true burden of disease. Relying on self-reporting of a physician’s diagnosis may potentially overestimate the true burden of disease. As is noted above, there is no single diagnostic test that determines when an individual has asthma. NHANES did not have other evaluations done (peak flow variability, sputum evaluation, allergy testing, etc.) that have been proposed as part of a case definition for ACOS. Even for COPD, the presence of obstruction on spirometry may not always mean that the person has COPD, and some cases of COPD may not have obstruction on spirometry.32,33
This analysis has demonstrated that ACOS, which involves the overlap of 2 common diseases, is highly influenced by the precise definitions used for those component diseases. These results are generally consistent with the published literature from clinical cohorts; asthma-COPD overlap syndrome (ACOS) is found in 1%-2% of the U.S. adult population, comprising about 3 million affected individuals. Studies that further attempt to determine risk factors, outcomes, and prevention strategies for ACOS will have to carefully consider the case definition and the implications of differing definitions on study design and outcomes.
Acknowledgements
All authors contributed to study design, data analysis, interpretation of data, creating the manuscript and approving the final manuscript. All authors are guarantors of the paper, taking responsibility for the integrity of the work as a whole, from inception to published article.
Declaration of Interest
DM has received honoraria/consulting fees and served on speaker bureaus for GlaxoSmithKline plc, Novartis Pharmaceuticals, Pfizer Inc., Boehringer-Ingelheim, AstraZeneca PLC, Forest Laboratories Inc., Merck, Amgen, and Sunovion. Furthermore, he has received royalties from Up-to-Date, is on the Board of Directors of the COPD Foundation, and has been compensated as a medical expert in legal cases. WG reports no conflicts of interest. KJD and KW are employees and shareholders of GlaxoSmithKline plc.