Running Head: COPD Maintenance Medication Patterns/Adherence in Medicare
Funding/Support: This work was supported by the Agency for Healthcare Research and Quality (R01-HS020642), the Patient-Centered Outcomes Research Institute (1R24HS022134-03), the Clinical and Translational Science Award (1UL1TR001439-01) from the National Center for Advancing Translational Sciences, and the Claude D. Pepper OAIC grant # P30 AG024832-11. Funders had no role in the preparation, approval or submission of the results.
Date of Acceptance: September 1, 2017
Abbreviations: chronic obstructive pulmonary disease, COPD; inhaled corticosteroids, ICSs; long-acting beta2-agonists, LABAs; long-acting muscarinic antagonists, LAMAs; proportion of days covered, PDC; odds ratio, OR; confidence interval, CI; phosphodiesterase, PDE-4; durable medical equipment, DME; International Classification of Diseases- 9th revision, ICD-9; generalized estimate equation, GEE; metered dose inhaler, MDI
Citation: Nishi SPE, Maslonka M, Zhang W, Kuo Y-F, Sharma G. Pattern and adherence to maintenance medication use in Medicare beneficiaries with chronic obstructive pulmonary disease: 2008-2013. Chronic Obstr Pulm Dis. 2018; 5(1): 16-26. doi: http://doi.org/10.15326/jcopdf.5.1.2017.0153
Introduction
Pharmacological management of patients with chronic obstructive pulmonary disease (COPD) is based on symptoms, disease severity and risk of exacerbation. Long-acting muscarinic antagonists (LAMAs), long-acting beta2- agonists (LABAs), phosphodiesterase (PDE-4) inhibitors and the combinations fixed-dose inhaled corticosteroids (ICSs) with a LABA and LABA/LAMA are approved as maintenance medications to manage patients with moderate to severe COPD.1 These medications have been shown to provide symptomatic relief, improve lung function and reduce the risk of exacerbations.1-3
Current knowledge of the use of and adherence to maintenance medication comes from randomized controlled trials.4-6 In the real-world setting, adherence to clinical practice guidelines is low in patients with COPD.7,8 The high burden of comorbidities, complex inhaler regimens, co-existing psychosocial disorders and cost all negatively impact adherence.9-15 Moreover, a high proportion of patients prescribed these medications have not undergone spirometry to confirm the diagnosis of COPD.16 In general, patients with COPD are undertreated.17
We used a 5% national sample of Medicare beneficiaries with COPD to examine the pattern of maintenance medication use and adherence among maintenance medication users from 2008-2013.We also examined factors associated with the receipt of and adherence to maintenance medication. Our hypothesis was that the overall use of maintenance medication would increase during the study period, but the magnitude of increase would be impacted by the financial burden to the patient.
Methods
Data Source
This is a retrospective, cross-sectional study of maintenance medication use in individuals with COPD using a 5% Medicare beneficiary population, 2008–2013. This study was approved by the University of Texas Medical Branch Institutional Review Board and informed consent was waived due to the nature of the study. Over 98% of adults in the United States age ≥65 years are enrolled in Medicare, which includes >45 million beneficiaries. The Centers for Medicare and Medicaid Services selects a random sample of 5% Medicare beneficiaries based on the eighth and ninth digits (05, 20, 45, 70, 95) of their health insurance claim number. The resulting standard data is available for research purposes and has been shown to be representative of the entire cohort.18
Data from the following files were used for this study: 1) Denominator file (Medicare enrollment information and demographic data); 2) Medicare Provider Analysis and Review file (claims for hospital inpatient and skilled nursing facility stays); 3) Outpatient Standard Analytic File (hospital outpatient services); 4) 100% Physician/Supplier file (physician and other medical services); 5) Durable Medical Equipment (DME) file; and 6) Part D Drug Event file.
Study Cohort
A patient met the diagnosis of COPD who had any of the following:
1) At least 2 outpatient or consultation visits (Evaluation and Management codes 99201-99205, 99211-99215) with an encounter diagnosis of COPD at least 30 days apart within a year;
2) One acute care hospitalization with a primary discharge diagnosis of COPD based on the following International Classification of Diseases, 9th revision (ICD-9) codes: 491.x (chronic bronchitis), 492.x (emphysema) or 496 (chronic airway obstruction); or
3) An acute care hospitalization for respiratory failure (ICD-9 codes 518.81, 518.82, 518.84) as the primary discharge diagnosis and COPD listed as the secondary diagnosis.
A separate denominator file of beneficiaries with COPD was created for each calendar year (2008-2013). Each file was composed of participants in the year of interest who:
1) Had a diagnosis of COPD;
2) Were age ≥66 years and alive by the end of the year;
3) Had complete Medicare enrollment (Part A, Part B and Part D) and not enrolled in a health maintenance organization in the previous year and current year;
4) Were not a resident of a nursing facility in the previous year and current year; and
5) Were residents in 1 of 9 United States geographic regions.
Variables
Medicare enrollment files were used to categorize individuals by age (66–74, 75–84, ≥85 years), gender (male, female) and race/ethnicity (white, black, other). Financial burden of medication cost to the patient was determined by dual eligibility status. Dual-eligible beneficiaries refers to individuals qualifying for Medicare Part A and/or Part B who are also eligible for some form of Medicaid benefit. Dual eligibility status was identified by either state buy-in in the enrollment file or low-income subsidy Part D program. The education level at the zip code of residence was obtained from the American Community Survey estimates of the U.S. Census. A comorbidity score (0, 1, 2, ≥3) was generated using the Elixhauser comorbidity score (excluding COPD) from inpatient and outpatient billing data.19
A spirometry examination was identified through Current Procedural Terminology codes (94010, 94014, 94015, 94016, 94060, 94070, 94620). Oxygen therapy use was defined from Healthcare Common Procedure Coding System codes E1390-E1392. Outpatient pulmonary specialist visit was defined as any outpatient visit with a Health Care Financing Administration provider specialty code (“29”) during the study year.
Outcome Measures
Our outcomes of interest were receipt of COPD maintenance medication and maintenance medication adherence in each study year of interest for individuals with COPD. The COPD maintenance medications included LABAs, LAMAs, ICSs, and fixed-dose LABA/ICS combinations. Medication prescriptions and prescribed dosages were extracted from Part D and DME prescriptions filled. We chose to look at DME files as nebulized medication is covered under DME benefits. For each year, the proportion of days covered (PDC), defined as the proportion of days in a study year that a patient had COPD maintenance medication available from the index prescription filled date, was calculated. The index date was defined as the earliest date a maintenance medication prescription was filled during the study year. For prescriptions extending beyond the end of the year, the days covered were truncated at the end of the year. Beneficiaries were classified as adherent if their PDC ≥80%.
Statistical Analysis
Patient characteristics were summarized from 2008-2013 using counts and percentages of categorical variables. A generalized estimate equation (GEE) with binomial distribution model analysis was used to assess the trend in outcomes, adjusted for patient characteristics (age, gender, race, dual eligibility status, education level and comorbidity) and health care measures (COPD hospitalization in the previous year, having a spirometry test, oxygen therapy treatment and pulmonary specialist visit). The GEE accounted for the cluster effect of individuals.20 A priori 2-way interactions on maintenance medication use and adherence was tested between dual eligibility status and education level, and between dual eligibility and study years. All analyses were performed using SAS version 9.4 (SAS Inc., Cary, North Carolina). All reported p-values were two-sided with p<0.05 considered statistically significant.
Results
Table 1 presents the baseline characteristics of beneficiaries with COPD by study year. The number of individuals with COPD increased from 13,957 to 18,044. The majority were female, non-Hispanic white and had ≥2 comorbidities. Over a third of beneficiaries were prescribed home oxygen therapy, were seen by a pulmonary specialist and had spirometry testing.
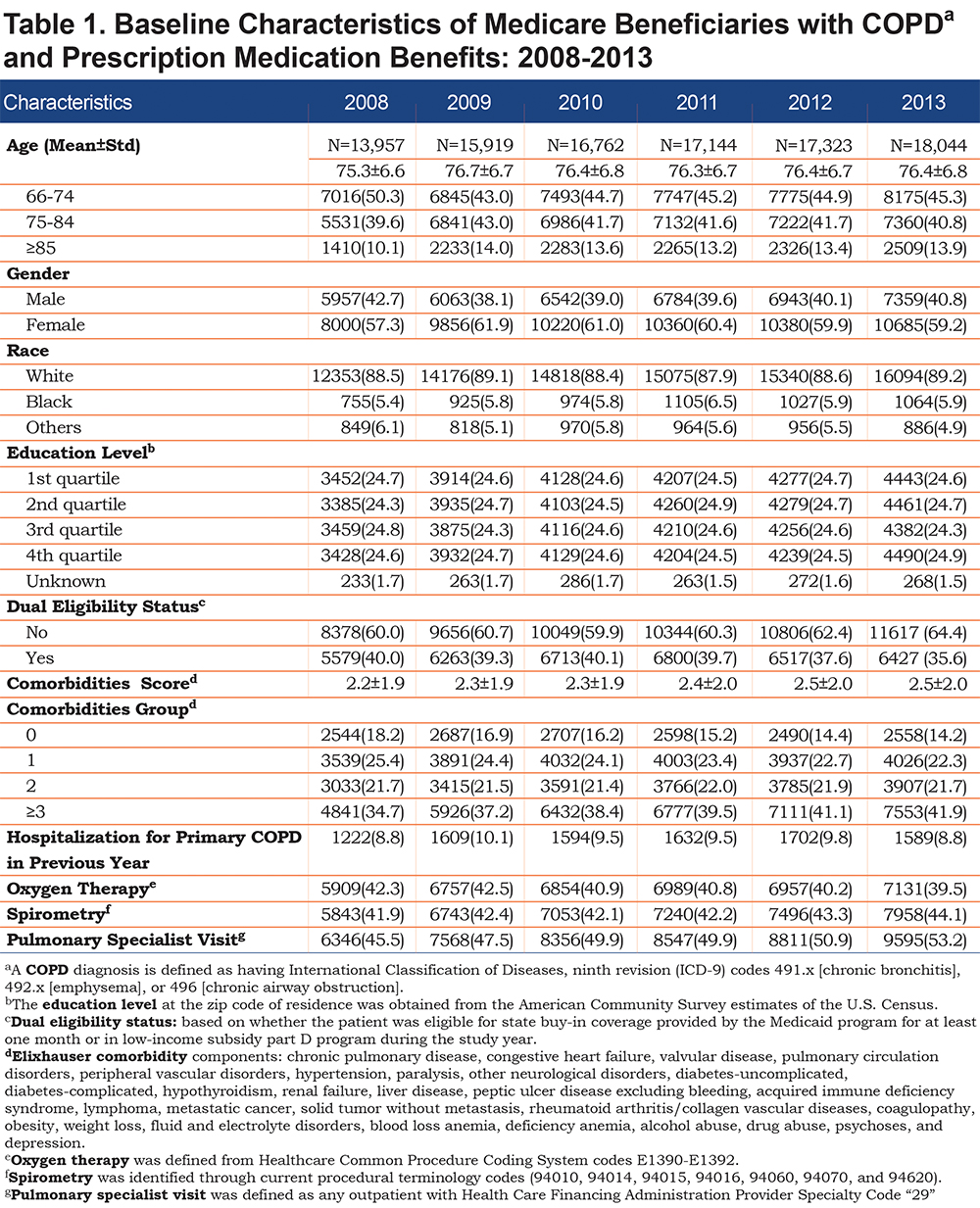
Table 2 presents receipt of COPD-related medications during the study period. Receipt of any COPD-related medication increased from 85.6% in 2008 to 87.1% in 2013. Approximately 72% of individuals filled some reliever medication, most commonly short-acting beta2-agonists (SABAs) as metered dose inhaler and SABA/SAMA nebulized solutions.
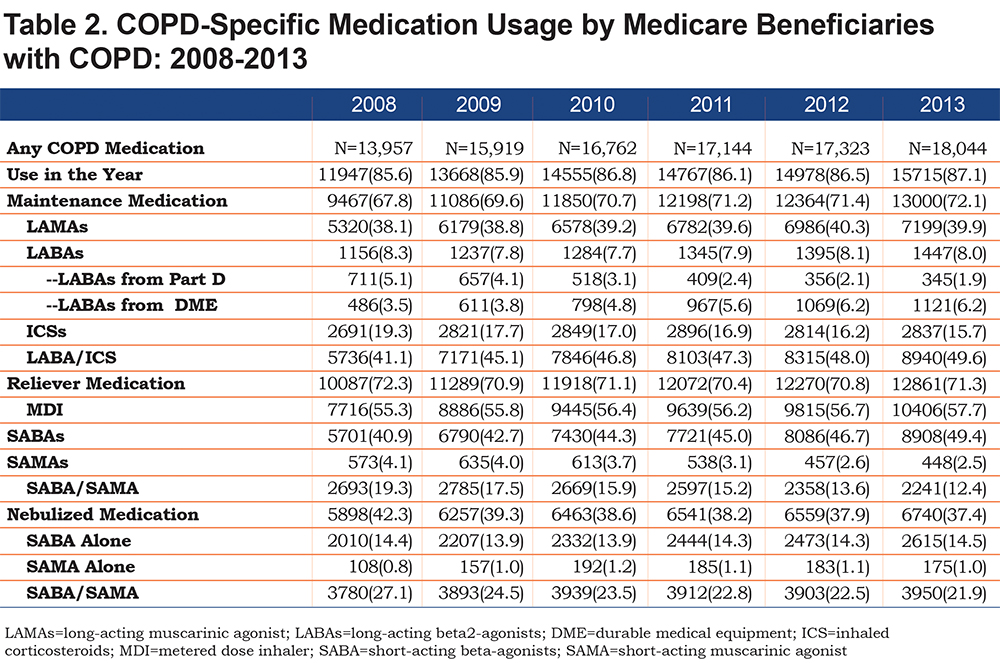
Maintenance medication (LAMAs, LABAs, ICSs and/or LABA/ICS combos) increased from 67.8% in 2008 to 72.1% in 2013. The increase in any maintenance medication during the study period was related to the increase in LABA/ICS combination use, which rose from 41.1% in 2008 to 49.6% in 2013. In contrast, use of an ICS alone decreased from 19.3% in 2008 to 15.7% in 2013. Overall, receipt of LABA prescriptions did not change over the study period. However, handheld device LABA prescriptions decreased, whereas nebulized LABA prescriptions increased, from 3.5% in 2008 to 6.2% in 2013. The receipt of maintenance medications by beneficiaries with COPD seen by a pulmonary specialist was high (77.1%-79.0%) but remained unchanged during the study period. In contrast, maintenance medication prescriptions filled by beneficiaries seen by primary care providers increased from 60.1% in 2008 to 64.6% in 2013. Across all study years, a higher proportion of women than men received maintenance medication. By gender, maintenance medication prescriptions filled increased from 68.8% in 2008 to 73.1% in 2013 in women and from 66.6% in 2008 to 70.6% in 2013 in men.
Figure 1 presents maintenance medication use (A) and adherence (B) by dual eligibility status. During the entire study period, a higher percentage of beneficiaries with dual eligibility status had maintenance medications filled compared to those who had Medicare only, suggesting that co-payment or deductible plays a role in the use of higher cost prescriptions. To examine adherence (B), we performed a subset analysis using beneficiaries who filled a prescription of any maintenance medication during the calendar year. As shown in the figure, individuals with dual eligibility status had a higher mean PDC over the study period, but the mean PDC of either group did not change over the study period.
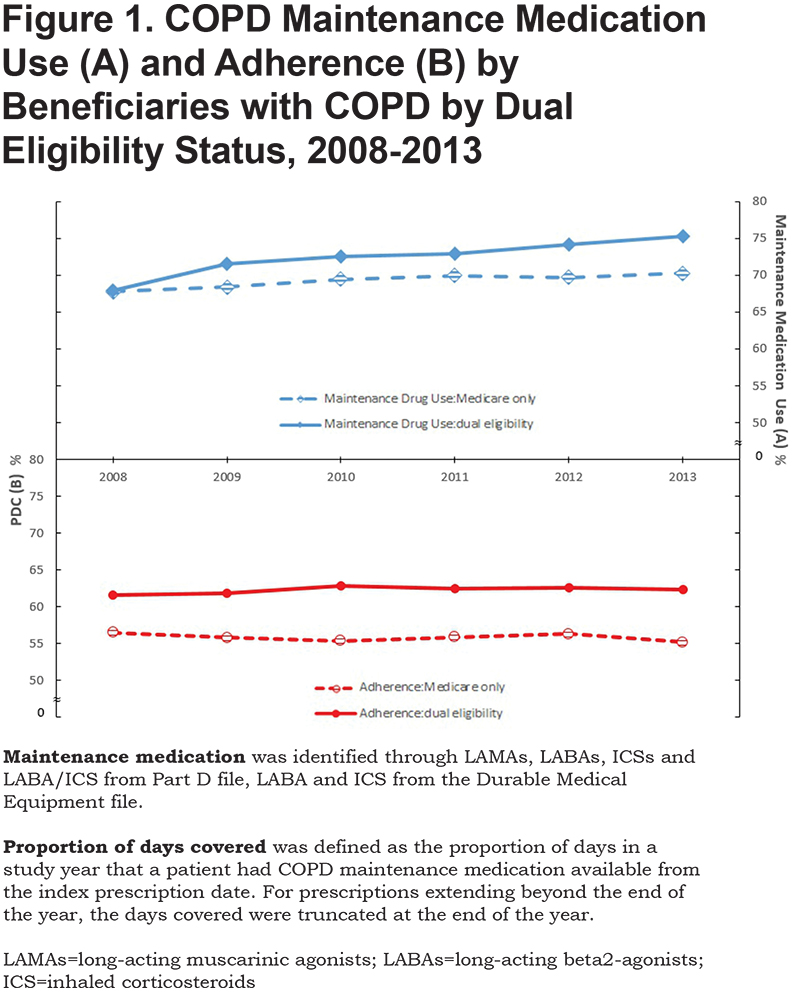
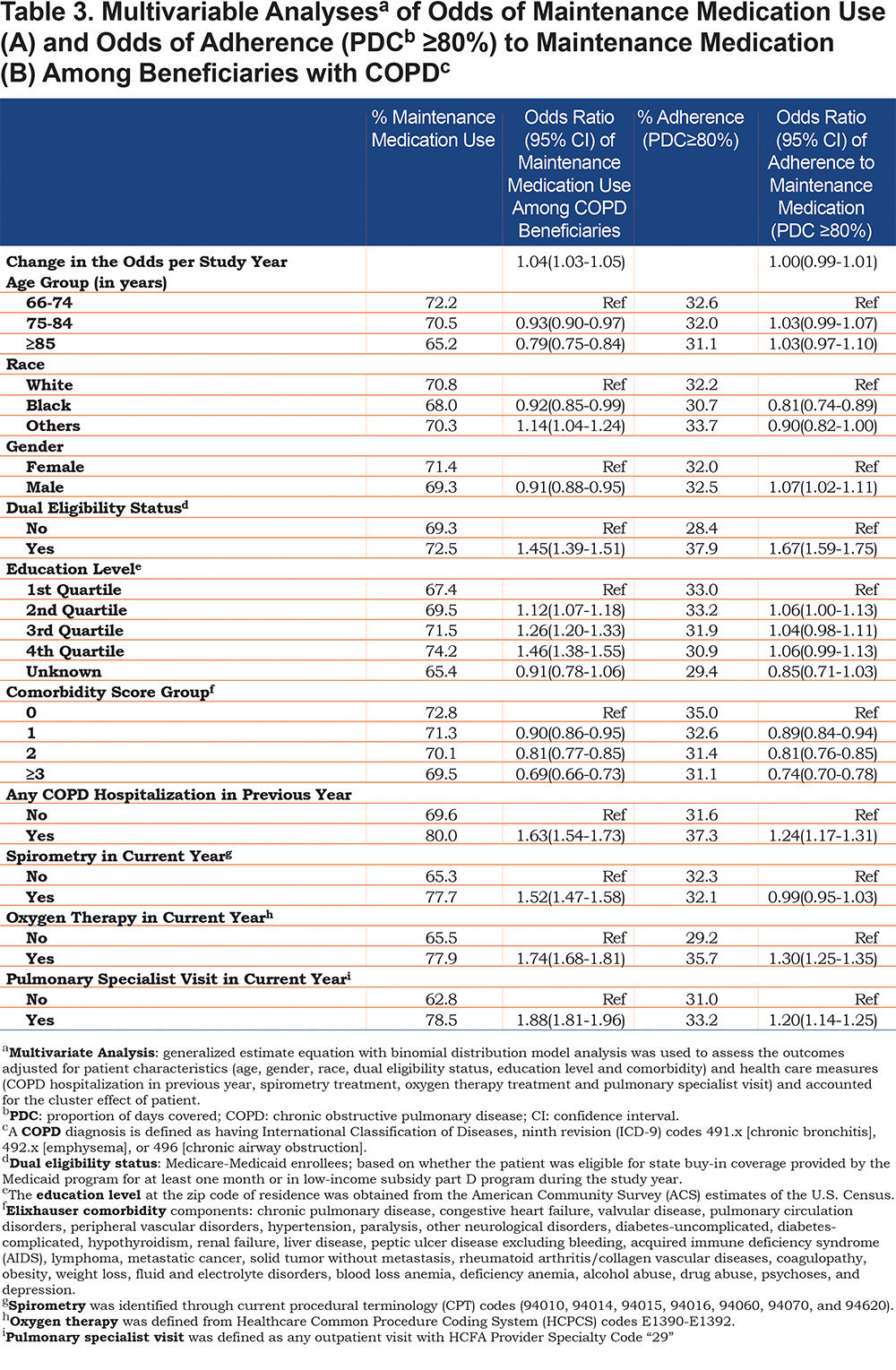
Table 3 presents the multivariable analyses of odds of receipt of prescription (A) and odds of adherence (B) (defined as PDC ≥80%) among beneficiaries who filled a maintenance medication prescription. There was a 4% (odds ratio [OR] 1.04; 95% confidence interval [CI] 1.03-1.05) increase in the odds of receiving a maintenance medication prescription per year from 2008 to 2013. The odds of receiving a maintenance medication were higher among individuals who were younger, female, with dual eligibility status (OR 1.45; 95% CI 1.39-1.51), with more education and those with fewer comorbidities. Beneficiaries who had a COPD hospitalization in the previous year (OR 1.63; 95% CI 1.54-1.73), had spirometry testing (OR 1.52; 95% CI 1.47-1.58), received oxygen therapy (OR 1.74; 95% CI 1.68-1.81) or were seen by a pulmonary specialist provider (OR 1.88; 95% CI 1.81-1.96) also had higher odds of receiving a prescription for a maintenance medication. In a subset analysis using individuals who filled a maintenance medication prescription, overall, there was no increase in the odds of adherence to maintenance medications during the study period. Factors associated with higher odds of adherence among individuals prescribed a maintenance medication include: dual eligibility status (OR 1.67; 95% CI 1.59-1.75), lower comorbidity burden, prior COPD hospitalization (OR 1.24; 95% CI 1.17-1.31), receipt of oxygen therapy (OR, 1.30; 95% CI, 1.25-1.35) and evaluation by a pulmonary specialist (OR 1.20; 95% CI 1.14-1.25). We further looked at the interaction of dual eligibility status by education level and dual eligibility status by year and found no significant effect on the interaction between dual eligibility status and education level in either model (maintenance medication prescription or adherence). However, the interaction of dual eligibility status by year had a significant effect on the maintenance medication prescription model. After adjusting for covariates as shown in Table 3, beneficiaries with dual eligibility status had a greater increase (OR 1.12; 95% CI 1.09-1.16) in the rate of receiving a maintenance medication prescription per year than those with Medicare only (OR 1.02; 95% CI 1.01-1.04); But in the adherence model, the interaction effects in both groups (with dual eligibility status and Medicare only) were not significant (OR 1.01; 95% CI 0.98-1.04 versus OR 0.99; 95% CI 0.98-1.01).
Discussion
The results of our study can be summarized as follows:
- maintenance medication use in beneficiaries with COPD is increasing;
- the largest increases can be attributed to increased LABA/ICS combination therapy;
- dual eligible (i.e., dual eligibility status) beneficiaries with COPD are more likely to receive a maintenance medication prescription and had a higher rate of increase in receipt of maintenance medications over time.
- Overall adherence to maintenance medication in this population remains suboptimal.
For effective chronic disease management of COPD, appropriate use of recommended prescription medication is crucial.21 Studies show low use of and adherence to COPD maintenance medication.8,10 Patient-related factors associated with risk of poor adherence include unemployment status, less education, low income, living alone and psychosocial comorbidity.13 Cross sectional and longitudinal studies examining the role of depression in adherence to COPD maintenance medication have shown that adherence to COPD maintenance medication falls precipitously within 6 months of use and depression is associated with lower adherence compared to those without depression.10,22,23 Other factors related to lower COPD maintenance medication adherence include lack of perceived or actual benefit from the prescribed medication,12,23 inability to follow instructions either due to cognitive15 or physical inability to use different inhalation devices and out-of-pocket cost of these medications.11,14,24
In our study, women were more likely than men to get a prescription for a maintenance medication. However, their adherence to maintenance medication was similar to that of men. This is congruent with prior reports which also showed no association between gender and adherence.8,12 One study did show lower adherence in women, but results were confounded by the higher prevalence of depression in women, as the latter is associated with lower adherence.10
Our findings of increased maintenance medication use in dual eligible individuals with COPD are worth exploring. Currently, none of the COPD maintenance medications are generic or covered under $4 generic prescription programs. However, dual eligible beneficiaries pay no premium and face no deductible under Part D, making these medications affordable under the plan. Beneficiaries who are not dual eligible may not be able to afford these medications. This situation is similar to that of cardiovascular prescriptions in which dual eligible beneficiaries are shown to have higher adherence.25
Potential strategies to improve medication adherence will be most successful if undertaken within a multidimensional approach. As shown in our study, the increased adherence over time of dual eligible beneficiaries supports the importance of medication coverage. Health care policy changes should focus on value-based insurance plans and improved prescription drug coverage for Medicare Part D beneficiaries, of whom 17% enter the coverage gap by October.24 Providers should personalize inhaler devices to optimize medication use and delivery,11 reduce the complexity of medication regimens whenever possible,8,14 decrease the number of device types prescribed26 and instill inhaler competency for themselves in order to educate patients.27-29 Pharmacist-led interventions may include inhaler education and medication synchronization, where all patients’ prescriptions are filled on the same day and scheduled closer to the receipt of their income payment.30-32 Treatments should ideally include the right medication-device combination for patients rather than a manufacturer proprietary device or insurance formulary determining the availability of a specific medication. Finally, monitoring and feedback about inhaler use through electronic medication delivery devices may assist in medication adherence.32
Limitations
Our study has several limitations. The analysis is limited to fee-for-service Medicare beneficiaries who had Medicare Parts A, B and D and is non-generalizable to managed Medicare or commercial insurance populations with COPD. Actual spirometry values and dyspnea scores cannot be assessed using administrative claims to verify a diagnosis or severity of COPD. However, we used surrogates for disease severity such as oxygen use, prior COPD hospitalization and clinic assessment by a pulmonary physician. The medication “use” is based on a prescription filled. This does not account for medications prescribed and never filled or medications used appropriately. Dual eligibility status is based on Medicaid coverage for at least 1 month during the entire year and may not be a true representation of the patient’s poverty status. Nonetheless, this measure is shown to be a good surrogate of the socioeconomic status of Medicare beneficiaries.33
In summary, maintenance medication use in fee-for-service Medicare beneficiaries increased during the study period and is higher in dual eligible beneficiaries. However, the overall adherence to maintenance medications is suboptimal and remains unchanged.
Acknowledgements
The authors thank Sarah Toombs Smith, PhD, ELS for help with preparation of the manuscript. Dr. Toombs Smith received no compensation for her work beyond her salary as a University of Texas Medical Branch employee. Authorship/Contributorship:
SPEN: Made substantial contributions to 1) conception and design, interpretation of data; 2) drafting the article or revising it critically for important intellectual content; and 3) final approval of the version to be published; guarantor of the paper; MM: 1) interpretation of data; 2) drafting the article or revising it critically for important intellectual content; and 3) final approval of the version to be published; WZ: 1) acquisition of data, 2) analysis and interpretation of data, 3) final approval of the version to be published; Y-FK: 1) substantial contributions to conception and design, 2) analysis and interpretation of data, 3) final approval of the version to be published; GS: 1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; 2) drafting the article or revising it critically for important intellectual content; and 3) final approval of the version to be published.
Declaration of Interest
GS serves on the advisory board of Theravance Biopharma, Mylan and Sunovion pharmaceutical companies. The other co-authors have no financial conflicts of interest to disclose.