Running Head: Comorbid Disease on Lung Cancer Screening Scans
Funding Support: The project described was supported by Award Number U01 HL089897 and Award Number U01 HL089856 from the National Heart, Lung, and Blood Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Heart, Lung, and Blood Institute or the National Institutes of Health. Dr. Budoff and the coronary artery calcium measurements were supported by Tobacco-Related Disease Research Program (TRDRP) – Grant # 20XT-0014. The COPDGene® project is also supported by the COPD Foundation through contributions made to an Industry Advisory Board comprised of AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Novartis, Pfizer, Siemens and Sunovion.
Date of Acceptance: April 4, 2019
Abbreviations: lung cancer screening, LCS; computed tomography, CT; COPD Genetic Epidemiology, COPDGene®; coronary artery calcification, CAC; myocardial infarction, MI; National Lung Screening Trial, NLST; chronic obstructive pulmonary disease, COPD; dual energy X-ray absorpticometry, DXA;forced expiratory volume in 1 second, FEV1; forced vital capacity, FVC; Global initiative for chronic Obstructive Lung Disease, GOLD; preserved ratio impaired spirometry, PRISm; low attenuation area, LAA; Hounsfield units, HU; quantitative CT, QCT; bone mineral density, BMD; longitudinal follow-up, LFU; coronary artery disease, CAD; confidence interval, CI
Citation: Regan EA, Lowe KE, Make BJ, et al and the COPDGene investigators. Identifying smoking-related disease on lung cancer screening CT scans: increasing the value. Chronic Obstr Pulm Dis. 2019; 6(3): 233-245. doi: http://doi.org/10.15326/jcopdf.6.3.2018.0142
Online Supplemental Material: Read Online Supplemental Material (306KB)
Introduction
Screening low-dose chest CT scans were shown in the National Lung Screening Trial (NLST) to reduce all-cause and lung cancer mortality in high-risk participants1,2 and are now covered in the United States by both Medicare and private insurers. Estimates of the number of individuals potentially eligible for lung cancer screening (LCS) in the United States are substantial, ranging from 8 to 37 million people,3,4 depending on the criteria for screening. Implementation of LCS is complex and costly. Final decisions regarding lung cancer screening in European countries are pending,5 and in the United States, recognition of the costs and complexity may be tempering enthusiasm.6
Although the primary goal of LCS has been to reduce lung cancer mortality, cigarette smoking affects multiple organ systems, and is associated with increased risk of chronic diseases including cardiovascular disease, chronic obstructive pulmonary disease (COPD), and osteoporosis. These conditions lead to adverse outcomes that increase morbidity, mortality and health care costs. Thus, it is reasonable to consider the value of systematically interpreting the additional diagnostic information that is present on these CT scans, to determine whether key diagnoses can be identified and how often the diagnosis by CT was “new” for a patient, i.e., beyond their clinically known and treated conditions.
In addition to detecting lung cancers and high-risk lung nodules,7-9 chest CT scans can provide information from either visual scoring10 or quantitative methods11,12 on the extent of emphysema. Undiagnosed COPD is common among smokers and is associated with exacerbation-like respiratory events and increased use of health care resources.13 Currently, population screening for COPD is not recommended14 although case finding in high-risk participants (i.e., smokers) is supported. Further, even smokers with normal spirometry may have lung disease, including emphysema, visible on CT scan, or have significant respiratory symptoms including exacerbation-like events.15 Emphysema and/or COPD are associated with greater risk of lung cancer, and identification of these may provide additional information for interpreting cancer risk.16-18
Ungated chest CT scans can provide reliable estimates of coronary artery calcification (CAC),19 a strong predictor of future cardiovascular events.20 Smokers are as likely to die of cardiovascular disease as pulmonary disease.21,22 They also are at risk of adverse events including myocardial infarctions (MIs), strokes and congestive heart failure. Low dose, ungated lung cancer screening CT scans can identify cardiovascular risk based on CAC and provide risk prediction similar to Agatston scores derived from gated scans.23-25
Osteoporosis and fragility fractures in older adults are associated with death, loss of independence and declines in physical function.26,27 Osteoporosis is itself associated with increased mortality and morbidity in the elderly, especially among patients with COPD.28,29 Although there are established screening recommendations for osteoporosis based on age, female gender, and current smoking, we and others have previously shown male smokers, former smokers and COPD patients are at increased risk, but are not likely to be assessed or treated under current guidelines.30-32 Quantitative bone measurements from chest CT scans can estimate the potential for osteoporosis-related fracture risk from chest CT scans.33-35CT attenuation values in the vertebral bodies have been shown to correlate well with dual energy x-ray absorptiometry (DXA) results and predict fracture risk.36-38
Based on those considerations, we postulated there should be radiographic evidence of these specific comorbid diseases and related outcomes or events in LCS eligible smokers. The adverse outcomes of interest were: myocardial infarction or stroke from cardiovascular disease, respiratory exacerbations or pneumonia from smoking-related lung disease, and vertebral fractures or hip fractures from osteoporosis. Little is known about the occurence of these adverse events and comorbid conditions in the context of usual clinical care screening and prevention strategies among the current and former smokers included in the LCS cohorts.
We tested these hypotheses in the COPD Genetic Epidemiology (COPDGene®) study, a longitudinal cohort of 10,192 smokers enrolled to identify genetic determinants and subtypes of COPD. We identified participants from the COPDGene® cohort who would have met criteria for LCS at their entry into the study. We used spirometry, clinical characteristics, pre-existing diagnoses, medication use, longitudinal follow-up data and CT findings from Phase 1 COPDGene® to construct descriptions of prevalent comorbid disease, outcomes and new diagnoses.
Methods
Cohort
The COPDGene® Phase 1 visit was conducted between 2007 and 2011 at 21 clinical centers in the United States as described previously.15 The research protocol was reviewed and approved by the local institutional review board at each clinical center, and participants signed written informed consent. Participants were recruited from local communities near the clinical centers and there was enhanced recruitment of COPD participants from pulmonary practices.
Spirometry
Pre- and post-bronchodilator spirometry was performed using Easy One spirometers (ndd, Andover, Massachusetts) and following American Thoracic Society guidelines.39 Participants were classified as having COPD based on forced expiratory volume in 1 second (FEV1) < 80% predicted and FEV1/ forced vital capacity (FVC) < 0.7 using the Global initiative for chronic Obstructive Lung Disease (GOLD) criteria.40 Smokers with normal FEV1/FVC ratio but FEV1 below 80% predicted were labeled as having preserved ratio impaired spirometry (PRISm)41 and were also included in the cohort. We have previously demonstrated smokers with normal spirometry (GOLD 0), PRISm and GOLD 1 groups are similar to a population cohort in their distribution of FEV1.15
COPDGene® Lung Cancer Screening Subset
We selected participants from the COPDGene® cohort who would meet LCS criteria based on characteristics at their baseline study visit (Phase 1). These participants were age 55-79, current smoker or former smoker who quit fewer than 15 years ago and had at least a 30 pack-year smoking history. There were 4078 eligible participants and 4055 of those had complete spirometry results.
Questionnaires
COPDGene® collected Phase 1 data on respiratory symptoms, quality of life, medications, exposures and medical history including comorbid disease identified by a physician (available at www.copdgene.org).
CT Scans
Inspiratory (200 mAs) CT scans were available on most of the LCS eligible participants. Details of the CT procedure, calibration by phantom and analysis have been published.12 Briefly, each scan passing quality assurance was analyzed for emphysema, which we defined as the percentage of low attenuation areas (LAA) at a threshold of -950 Hounsfield Units (HU) (LAA%@ -950 HU) considered to be significant emphysema when > 5%.
Coronary Calcium
CAC above a threshold of 130 HUs involving 3 contiguous voxels was used to identify a calcific lesion, giving a minimum lesion area of 1.02 mm2. The lesion score was calculated using the area density method, by multiplying the lesion area by a density factor derived from the maximal HU within the area as described by Agatston.42,43 A total CAC score was determined by summing individual lesion scores from each of 4 anatomic sites (left main, left anterior descending, circumflex and right coronary arteries).19 A CAC greater than 100 mg was considered clinically significant.
Bone Density
Cancellous bone density was estimated using 2 integrated methods, quantitative CT (QCT) and bone attenuation values in the cohort. A calibrated QCT measurement was done in 3321 participants using automated software, N-VivoTM (Image Analysis) and a calibration phantom as previously described.44 A mean volumetric bone mineral density test (BMD) (mg /cm3) was calculated using a minimum of 3 vertebral bodies from T6 to L1. The second method was based on CT attenuation values using TeraRecon software (TeraRecon, Foster City, California to determine mean bone attenuation value from T7 – L1 for the full cohort. We derived BMD prediction equations (both by scanner and overall) by means of the calibrated QCT BMD and attenuation values, which we used to calculate an equivalent BMD for each participant. BMD equivalent value of < 120 mg/cc was considered low bone density. Vertebral fractures were scored on CT scans using the Genant criteria with 2 readers and an adjudicator as previously described.30
High Dose to Low Dose Computed Tomography Comparison
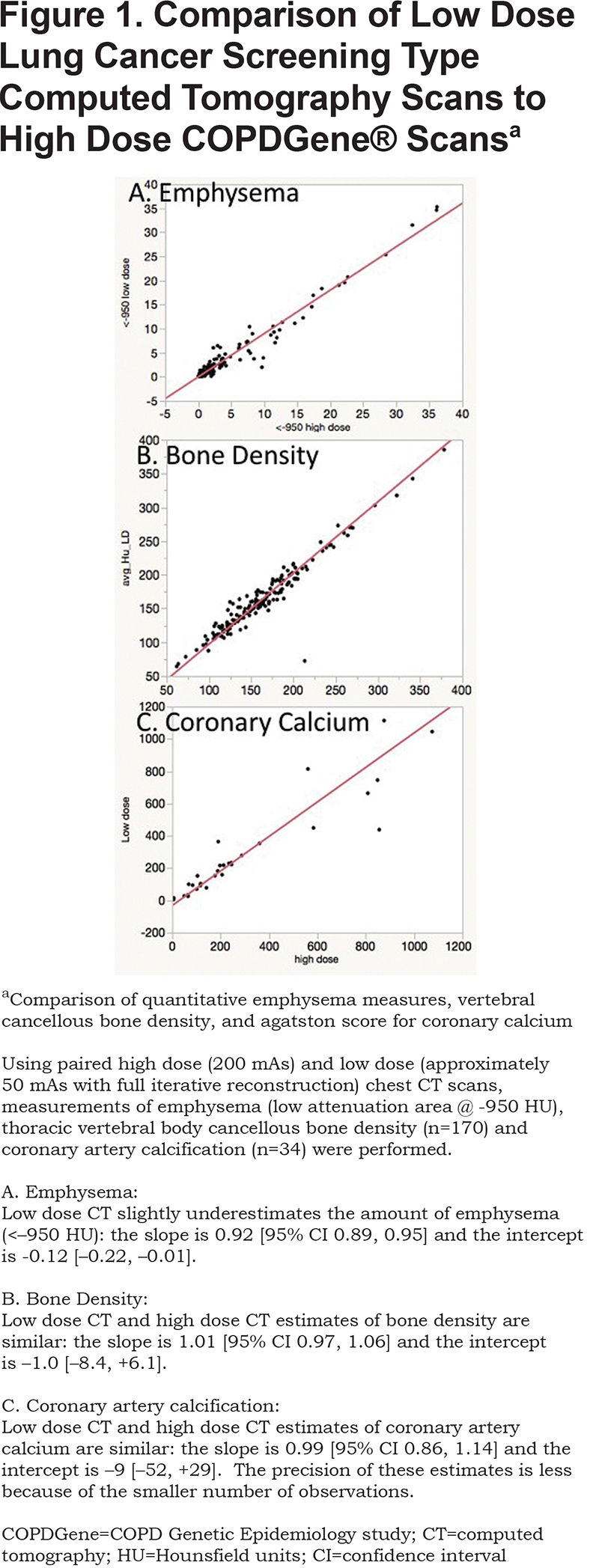
To compare the original 200 mAs inspiratory scans to low dose CT scans used in LCS programs, a subset of participants (n=170) from 5 sites were scanned using both the original CT protocol and a reduced dose protocol with dose modulation similar to low dose LCS scans. Reduced dose scans were reconstructed using filtered back projections and with “moderate” and “strong” levels of iterative reconstruction. Comparative measures of emphysema (n=170), CAC using L coronary artery (n=34), and cancellous bone density (n=170) were performed on both the original scans and reduced dose scans.
Longitudinal Data Collection
COPDGene® participants consented to be contacted by telephone or email every 6 months following their enrollment as part of the longitudinal follow-up (LFU) program.45 Participants were queried about hospitalizations, MIs, strokes, hip fractures, respiratory exacerbations that required glucocorticoids or antibiotics and episodes of pneumonia. Of the 4078 eligible participants, 388 had not participated in the LFU program leaving 3690 for analysis of the adverse events.
Data Analysis
The participants who were eligible for LCS in the COPDGene® cohort were grouped by severity of lung disease using the GOLD spirometric criteria.46 Key clinical characteristics were identified for each group, and group means were compared using analysis of variance and Student’s t-test for continuous variables and Chi Square or Fisher’s exact test for categorical variables. The GOLD 0 group of smokers with normal spirometry served as the reference group. Incident disease-associated clinical events (MI, stroke, respiratory exacerbation, pneumonia, and hip fracture) were identified by self report from the LFU data over the entire period of follow-up for each participant. These outcomes are presented by GOLD stage groups and totals, along with person-years of follow-up and number of recorded surveys. Vertebral fractures were visually scored in a subset of CT scans and used as an event associated with low bone density.
Ascertainment of pre-existing coronary artery disease and osteoporosis were based on either self-report of physician-diagnosed disease, use of lipid-lowering medication or osteoporosis medications, respectively. New CT-identified diagnoses were the difference between number with CT-defined disease and those with a previous diagnosis. Analyses were performed using STATA 15.1, SAS 9.3 and JMP 12.1 (both SAS Institute Inc, Cary, North Carolina).
Results
There were 4055 COPDGene® participants who met the criteria for LCS and had complete spirometry data (23 of the 4078 participants selected had failed spirometry ). Pulmonary nodules were identified in 29% (1195/4055). The clinical characteristics are shown in Table 1. Forty-seven percent (1919 participants) of this COPDGene® LCS cohort had either no, or mild COPD (GOLD 0, PRISm, GOLD Stage 1) as defined by spirometry. We found that GOLD 2-4 participants eligible for LCS screening were, unsurprisingly, characterized by greater dyspnea, more chronic bronchitis symptoms, more episodes of respiratory exacerbations and greater use of respiratory medications, than those with mild or no COPD.
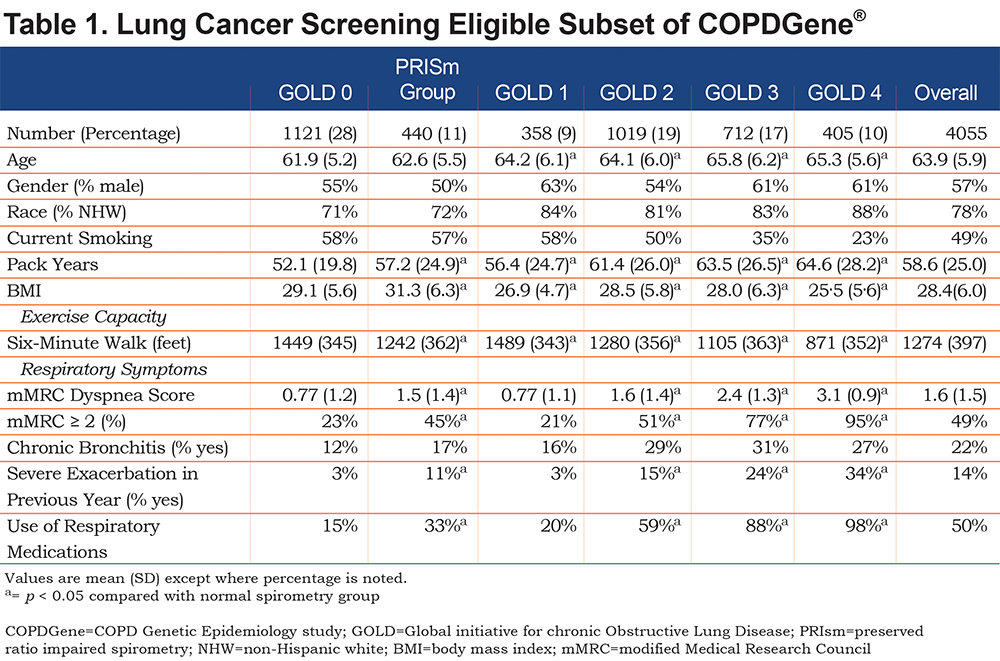
Significant adverse clinical events related to cardiovascular disease, osteoporosis or lung disease were common (Table 2). Among GOLD 0 smokers, exacerbations were reported by 21%, MIs by 5%, stroke by 4% and hip fracture by 3%. PRISm and the GOLD 1-4 groups had higher rates, with 9% and 8% MI, 8% and 7% stroke, 5% and 4% hip fractures and 38% and 56% exacerbations, respectively.
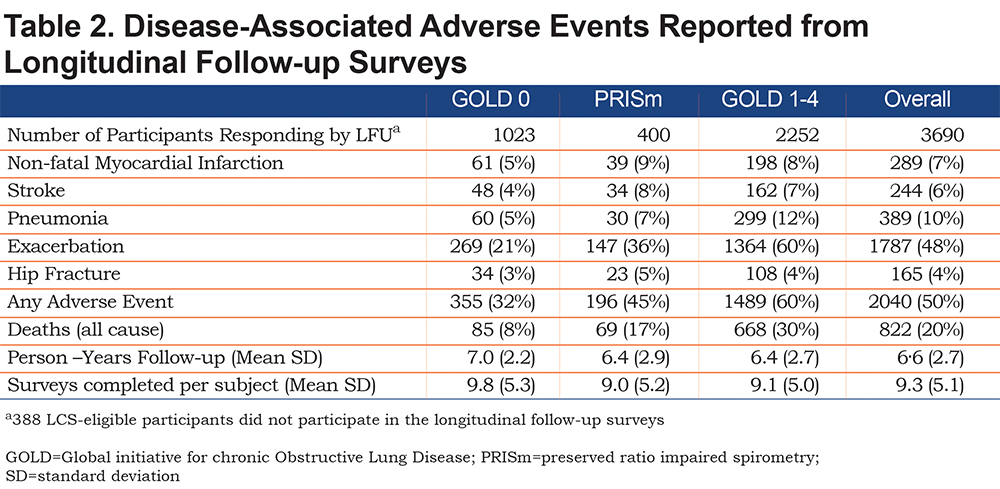
CT-based diagnoses showed 76% of participants had some combination of coronary artery disease, lung disease (emphysema), or osteoporosis (Table 3). We found 44% had abnormal (>100 mg) calcium in their coronary arteries. Emphysema was seen in 45% of participants and 56% of LCS-eligible participants had reduced bone attenuation values, suggesting osteoporosis in the thoracic spine.

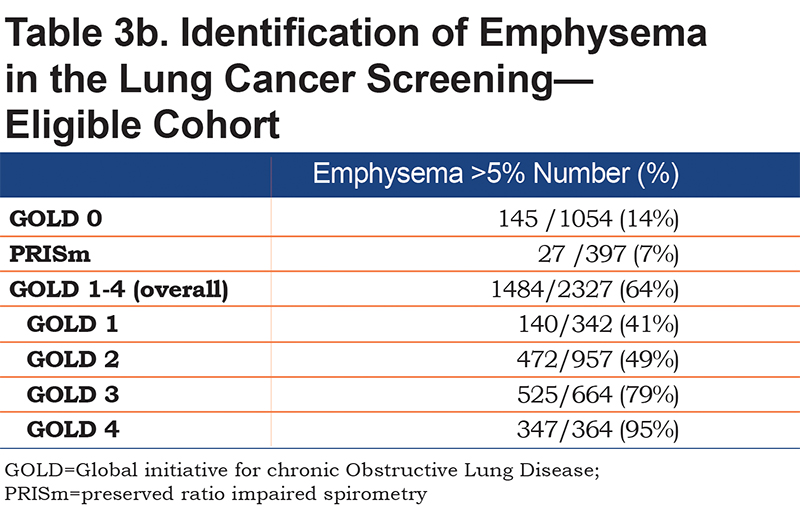
New diagnoses based on the CT analysis were identified for coronary artery disease (CAD), COPD based on emphysema, and osteoporosis (Table 4) taking into account pre-existing diagnoses. Participants who reported a physician diagnosis of CAD, emphysema or osteoporosis, or who used a medication related to the condition (such as statins or bisphosphonates) were compared to all those with abnormal CAC, emphysema or low bone density. A new diagnosis was presumed if there was no report of appropriate medication use or previous diagnosis.
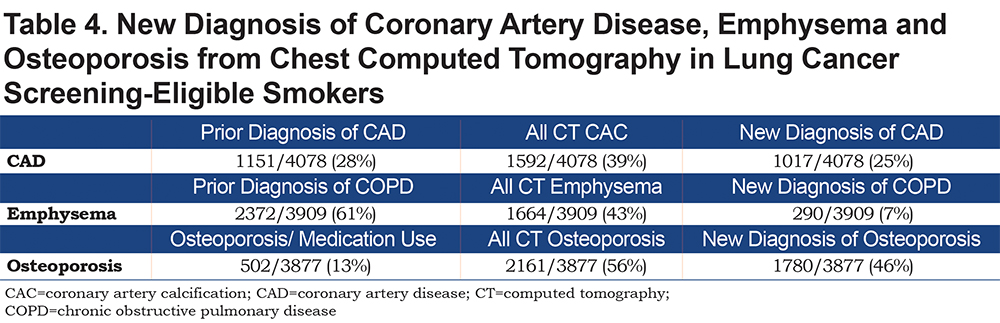
A new diagnosis of CAD was made in 111 (9.3%) GOLD 0 participants, 56 (12.6%) PRISm participants and 648 (24.8%) participants in the GOLD 1-4 groups. For osteoporosis, there were similar findings with 344 (28.9%) new diagnoses in GOLD 0 group, 116 (26.1%) in the PRISm group and 1066 (41.3%) in the GOLD 1-4 groups. In total, of the 1592 patients with an elevated CAC, for 1017 (25%) this was a new finding. For the participants with emphysema and osteoporosis, 7% and 46% of the cases were new, respectively (Table 4).
More events of MI, stroke, exacerbations, pneumonia, and fractures were seen in the participants with these CT diagnoses (Table 5). In the group with no history of CAD or statin use, 4% had either an MI or stroke reported. However, the additional presence of CAC on CT was associated with almost a 50% increase in events to 7% of the participants. Similarly, in the participants who had reported prior CAD diagnosis or statin use, CAC was associated with more events (10% versus 14%). A Cox regression model adjusting for age, gender, race, pack years smoking, current smoking and FEV1, showed a significant hazard ratio for a new diagnosis of CVD risk due to CAC of 1.5, confidence interval (CI) 1.1- 2.0, p=0.006.
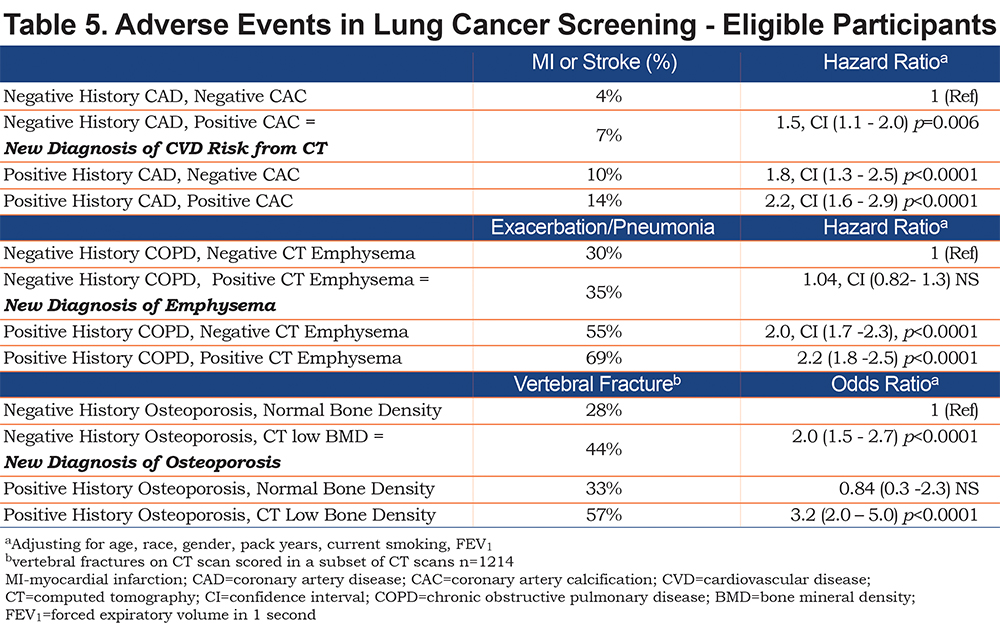
Prevalent vertebral fractures were strongly associated with the new diagnosis of low vertebral bone density from CT (28% versus 44%, p<0.0001 by Chi Square), and in the adjusted logistic regression model giving an odds ratio of 2.0, CI 1.5 – 2.7, p<0.0001. A positive history of osteoporosis and normal CT measurement of bone density was associated with lower risk of fracture and non-significant odds ratio of 0.8, but the history of prior diagnosis and low bone density on CT scan was seen to have the highest odds ratio (3.2, CI 2.0 – 5.0, p<0.0001) for prevalent vertebral fractures. Incident hip fractures were evaluated for an association to CT bone density but the results were not significant likely due to the small numbers of hip fractures reported (n=150).
Although the GOLD 1-4 participants were all diagnosed with COPD based on spirometric airway obstruction, not all of those participants have emphysema on CT scan (61.3% overall in GOLD 1-4). The proportion of COPD participants who have emphysema on CT scan increases over the GOLD stages (GOLD 0 – 11.8%, GOLD 1 – 38.4%, GOLD 2 – 47.1%, GOLD 3 - 77.3%, GOLD 4 – 94.5%). Emphysema was frequently identified in the GOLD 0 and PRISm groups - in spite of having spirometry values that did not classify them as having COPD. In the GOLD 0 group 12% had emphysema > 5%; in the PRISm group 7% had emphysema (Table 3b). The presence of emphysema was not associated with an increase in respiratory events (data not shown).
Low dose scans with iterative reconstruction were compared to the standard research scans for CAC, emphysema and bone density measures (Figure 1). There was good agreement in these measures between the low dose and research scans.
Discussion/Conclusions
Our analysis of a large cohort who would be eligible for lung cancer screening has 2 principal findings. First, the 3 comorbid diseases (CAD,47-49 emphysema, and osteoporosis) were highly prevalent on CT scans, with 75% of participants having 1 or more of these conditions. Though not fully reversible, these conditions are eminently treatable with reasonable expectation of reduced mortality, improved function and quality of life, and reduced fracture risk. Importantly, CAD and osteoporosis were new diagnoses (as assessed by the absence of any self-report of physician diagnosis or treatment) in 25% and 46% of participants. Emphysema was not a frequent new diagnosis in this cohort but might be in an actual LCS cohort.
Second, significant adverse clinical outcomes related to these 3 comorbid diseases occurred very frequently over a mean of 6 years of follow-up. Even in the GOLD 0 and PRISM groups, i.e., those not meeting the current definition of COPD, 32% and 45% respectively experienced 1 or more disease-related outcome. These results illustrate the potential added value that could be obtained from LCS scans with minimal additional effort.
We found that a new diagnosis of either CAD risk or osteoporosis was associated with an increased hazard ratio for the associated adverse event after adjustment for baseline variables. Participants with previously diagnosed COPD showed higher hazard ratios for exacerbation/ pneumonia that were not affected by the finding of emphysema, but finding emphysema in participants without a previous diagnosis of COPD was not associated with greater risk. For CAD, those with a positive history of disease still had greater risk than the new diagnosis participants.
In addition to the expected pulmonary nodules that were similar to other LCS cohorts, we found evidence of important smoking-related diseases on CT scans. Given the frequency of new diagnoses that we demonstrate and potential for treatment, the value of LCS scans could be increased. These results define a rationale for enhanced readings in community-based LCS programs. In terms of feasibility, COPDGene® utilized clinical radiation dose (rather than low-dose screening) with automated analysis of lung parenchyma that may not be appropriate for all LCS programs, but visual assessment of low-dose LCS scans can provide similar information about emphysema. Both CAC and bone attenuation values have been successfully determined from low dose screening scans.8,23,38
The current diagnostic standard for osteoporosis is DXA, but there is evidence to suggest QCT permits better fracture prediction in certain populations.30,33 Alternatively, after LCS scans identify low bone attenuation in the vertebral bodies, a separate DXA scan could be performed for confirmation and to assess bone density in the hips. Both male and female smokers are at increased risk of osteoporosis and fractures. Concerns about radiation exposure have limited the use of QCT for assessing bone density but in the context of LCS and its existing data, it may be an optimal choice or a strong adjunct, especially for vertebral fractures.
Howard et alstudied comorbidities in LCS eligible individuals and argued the expected population for LCS would be older and have more comorbid diseases than seen in the NLST.50 They concluded that this population of LCS participants would derive more benefit from lung cancer screening and would have more current smokers, comorbidities and shorter life expectancies. Our participants reflect those different demographics and outcomes.
Our data confirm and extend those of the NELSON LCS cohort51 which showed that low-dose CTs could identify COPD with a sensitivity of 63% and specificity of 88%. CT emphysema was a major factor in their model but they also included gas trapping. However, determining gas trapping requires an expiratory scan which current LCS guidelines omit to minimize radiation exposure. Accordingly, we chose to focus on emphysema as a key marker of COPD. CT emphysema was associated with all-cause mortality in participants from the MESA study, even without overt COPD defined by spirometry.52 Our data shows that emphysema prevalence increased steadily in association with more severe obstruction, becoming nearly universal among GOLD stage 4 participants.
Because the prevalences of the 3 conditions we investigated here are much higher than that of lung cancer, we would expect that efforts to identify and treat them should have a sizable public health impact. Our participants have heavy smoking exposures and current treatments to prevent adverse events would not be expected to be completely effective, however, a sizable number had undiagnosed comorbid disease and could benefit from treatment. Additionally, case-finding for these conditions provides concrete information to current smokers about these diagnoses to encourage smoking cessation – rather than providing false reassurances from a report of no cancer.
A limitation of this cohort is its enrichment for severe COPD, such that the overall prevalence of both lung disease and comorbid conditions associated with COPD are substantially increased. Thus, our results cannot be directly extrapolated to all LCS-eligible populations. However, prevalence of these comorbidities in our normal spirometry and PRISm participants do approximate what can be identified in community-based LCS programs. Moreover, some proportion of the participants in any LCS program will likely have COPD and also potentially benefit from treatment of their comorbid cardiovascular and bone diseases. Other limitations include generalizability of CT findings. The CT analysis for COPDGene® was research based, higher radiation dose, relied on quantitative analysis of CT scans, and the results may differ from methods used in clinical practice. Nonetheless, visual scoring of emphysema and CAC are feasible.23,53
Because LCS programs represent a fixed investment both in cost and radiation exposure, the opportunity to increase their value is attractive. Further analysis and research are clearly needed to define subsequent testing strategies. Current guidelines recommend lipid lowering therapy be applied to patients with increased CAC.54 Information about emphysema may ultimately inform a more refined assessment of cancer risk and screening intervals, but also could be used to enhance smoking cessation efforts.
In summary, we provide evidence that expanded readings of LCS CT scans to include assessment of coronary calcium, emphysema and osteoporosis could favorably impact the health and well-being of large numbers of ever-smokers. Identification and effective treatments might reduce the number and costs of adverse events among these patients at high risk. These benefits could be obtained without change in radiation burden or facility costs of the procedure, and with modest impact on workflow. What is needed is an organized effort to collect and disseminate information to patients and their physicians that currently is being ignored. The existing structure of LCS programs could be modified to provide expanded written reports on the associated comorbid diseases. Furthermore, as LCS patients are expected to return for annual visits, it is feasible to evaluate directly the impact of interventions on the frequencies of cardiovascular events, respiratory exacerbations, and new fractures.
Acknowledgements
Author Contributions: Concept: EAR, BJM, JDC, JHF; data analysis: KEL, EAR, DAL, DCE, MJB, SSM, SH: manuscript development, writing and editing: EAR, KEL, BJM, DAL, GLK, MJB, SSM, DD, JLC, RPB, MKH, THB, JEH, EK, SH, DCE, EJRvB, EKS, JDC, JHF
Declaration of Interest
Jeffrey L. Curtis reports grants from the National Heart Lung and Blood Institute, the National Institute of Allergy and Infectious Diseases and the Departments of Defense and Veterans Affairs, outside the scope of this project. Russell P. Bowler reports serving on advisory boards for GlaxoSmithKline, Boehringer Ingelheim and Mylan Pharmaceuticals and has received research grants from GlaxoSmithKline and Boehringer Ingelheim. Meilan K. Han reports consulting work for GlaxoSmithKline, Boehringer Ingelheim, Astra Zeneca and Mylan Pharmaceuticals and research support from Novartis and Sunovion. Edwin K. Silverman reports grant and travel support from GlaxoSmithKline in the past 3 years. Ms. Lowe and Drs. Regan, Make, Lynch, Kinny, Budoff, Mao, Dyer, Beaty, Hokanson, Kern, Humphries, Curran-Everett, van Beek, Crapo and Finigan have nothing to declare.