Running Head: Plasma Cathelicidin and COPD Outcomes
Funding Support: SPIROMICS was supported by contracts from the National Institutes of Health and the National Heart, Lung and Blood Institute (HHSN268200900013C, HHSN268200900014C, HHSN268200900015C, HHSN268200900016C, HHSN268200900017C, HHSN268200900018C, HHSN268200900019C, HHSN268200900020C), which were supplemented by contributions made through the Foundation for the National Institutes of Health from AstraZeneca, Bellerophon Therapeutics, Boehringer-Ingelheim Pharmaceuticals, Inc., Chiesi Farmaceutici SpA, Forest Research Institute, Inc., GSK, Grifols Therapeutics, Inc., Ikaria, Inc., and Nycomed. RMB is supported by National Institutes of Health, National Heart, Lung and Blood Institute grant F32HL143867. MBD is supported by National Institutes of Health, National Heart, Lung and Blood Institute grant R01HL125432-06.
Date of Acceptance: May 27, 2020
Abbreviations: chronic obstructive pulmonary disease, COPD; SubPopulations and InteRmediate Outcome Measures In COPD Study, SPIROMICS; body mass index, BMI; forced expiratory volume in 1 second, FEV1; confidence interval, CI; airway surface liquid, ASL; acute exacerbations of COPD, AECOPD; C-reactive protein, CRP; white blood cell count, WBC; modified Medical Research Council dyspnea scale, mMRC; COPD Assessment Test, CAT; bronchodilator, BD; hydroxy, OH; standard deviation, SD
Citation: Burkes RM, Ceppe AS, Couper DJ, et al. Plasma cathelicidin is independently associated with reduced lung function in COPD: analysis of the Subpopulations and Intermediate Outcome Measures In COPD Study Cohort. Chronic Obstr Pulm Dis. 2020; 7(4): 370-381. doi: http://doi.org/10.15326/jcopdf.7.4.2020.0142
Online Supplemental Material: Read Online Supplemental Material (251KB)
Introduction
The innate immune system of the airways is the first line of defense from inhaled and aspirated pathogens and noxious agents. Alterations of innate immunity may play a role in the progression of chronic obstructive pulmonary disease (COPD).1-3 Cathelicidin (also known in humans as LL-37) is an antimicrobial peptide of the innate immune system present in the airway surface liquid (ASL), secreted by airway epithelial and immune cells.4-8 Cathelicidin gene transcription is regulated by 1,25-hydroxy-vitamin D.9,10 Cathelicidin is expressed constitutively and induced in the presence of microbes, lipopolysaccharide, and inflammatory cytokines.11 Cathelicidin acts via direct microbial killing, immune cell signaling, increasing antigen-presenting cell activity, lipopolysaccharide neutralization, and signaling of epithelial cell apoptosis.8,12-15 Levels of both plasma and sputum cathelicidin have been shown to respond to the presence of pathogens or noxious stimuli.16-19
We have previously shown that reduced cathelicidin levels are associated with adverse pulmonary outcomes in non-COPD populations. In cross-sectional analysis of a cohort of current and former injection drug users with prevalent smoking, lower plasma cathelicidin levels were associated with lower forced expiratory volume in 1 second (FEV1) and patient-reported pneumonia events.2 In longitudinal analyses of smokers without COPD, lower plasma cathelicidin has been associated with reduced FEV1 at 6 and 18 months.3 Studies have reported conflicting results regarding the association between cathelicidin level and COPD exacerbation (acute exacerbation of COPD [AECOPD]) risk.17,20 To our knowledge, there has been no investigation of the association between cathelicidin levels and longitudinal pulmonary outcomes in well-characterized COPD cohorts.
The SubPopulations and InteRmediate Outcome Measures In COPD Study (SPIROMICS) is a prospective, multicenter cohort study with available baseline plasma and serum samples in addition to well-characterized demographic and clinical variables.21 SPIROMICS collects cross-sectional and longitudinal lung function measurements along with historical and prospective COPD exacerbations data. The SPIROMICS cohort allows for determination of relationships between cathelicidin levels and longitudinal lung function and exacerbation outcomes, as well as the association between cathelicidin and vitamin D, a regulator of cathelicidin. We hypothesize that low plasma cathelicidin will be associated with reduced lung function and increased COPD exacerbations at enrollment and after one year of follow-up.
Methods
Study Participants
SPIROMICS (total cohort n=2974) is a National Heart, Lung and Blood Institute-funded, multicenter, observational, prospective cohort developed to assess intermediate outcome measures and identify COPD subphenotypes.21 The cohort includes participants recruited from 12 clinical centers who are current or former smokers (≥ 20 pack years) with or without chronic airflow obstruction and non-smoking controls between the ages of 40-80. The analytical cohort consists of 1609 participants with available plasma cathelicidin measurements and spirometry confirmed COPD (post-bronchodilator FEV1 to forced vital capacity [FVC] ratio < 0.70) (Online Supplement Figure 1). Institutional review boards at each center approved SPIROMICS and participants provided informed, written consent.
Data Collection
Demographic data, medical history, self-reported smoking status (current versus former), smoking history (in pack years), and body mass index (BMI) were collected at enrollment. Post-bronchodilator spirometry values (FEV1, FVC, FEV1/FVC) were collected and standardized according to published standards.22 Historical AECOPD events were participant-reported at enrollment and involve any or severe AECOPD events in the 365 days prior to enrollment. Longitudinal AECOPD events were collected via participant self-report by quarterly phone calls and at a 1-year follow-up visit 365 days after study enrollment. An AECOPD event was defined as the need for any increased therapy due to COPD symptoms. Severe AECOPD was defined as an acute worsening of COPD symptoms leading to an emergency department visit or hospitalization. Stored baseline plasma samples were used to measure cathelicidin using ELISA (Hycult, Inc; limit of detection: 0.14 ng/ml, standard range: 0.14 ng/ml to 100 ng/ml) and serum 25-hydroxy vitamin D via radioimmunoassay (iDS, Enzyme Immunoassay; Intra-assay coefficient of variance: 8.14%, limit of blank: 1.8 ng/ml and limit of detection: 3.7 ng/ml).
Statistical Approach
The cathelicidin distribution within the cohort was characterized using descriptive statistics. Cathelicidin levels were dichotomized using a cut-point of the lowest quartile (< 50 ng/ml) versus the highest 3 quartiles (≥ 50 ng/ml) from SPIROMICS measurements conducted for this analysis, as well as continuously (per 10 ng/ml). Two-sample t-test and Mann-Whitney U testing for continuous variables (depending on skew of data) or chi-squared testing (for categorical variables) was performed to identify the differences in demographic and clinical factors between participants with cathelicidin < 50 ng/ml versus ≥ 50 ng/ml. Counts and percentages presented in Table 1 account for variable missingness, which is less than 10% for each variable and assumed to be random. For analyses, FEV1 was modeled continuously as % predicted FEV1. Linear regression modeling was used to determine bivariate relationships between cathelicidin and baseline % predicted FEV1. Fixed-effect multilevel modeling was used to determine multivariable relationships between continuous and categorical cathelicidin and % predicted FEV1 at baseline and rate of change over one year. Covariates in multivariable multilevel regression models were selected based on clinical relevance23 and include pack years smoked and current smoking status (age, race, sex, and height are included in the calculation of FEV1 % predicted). Any and severe AECOPD outcomes were modeled dichotomously (0 versus ≥ 1 event) in logistic regression analysis. Multivariable models assessing the relationship between cathelicidin and AECOPD included age, race, sex, smoking status, pack years smoked, and BMI. Correlation between cathelicidin and vitamin D as well as other blood markers implicated in COPD progression and outcomes in prior publications24,25 were assessed by Pearson’s correlation. These blood-based markers included C-reactive protein (CRP), white blood cell (WBC) count, fibrinogen, interleukin-6, S100 calcium-binding protein, and club cell secretory protein-16.
Several sensitivity analyses were performed. Serum vitamin D levels were included in statistically significant models. Statistical moderation and stratified analysis incorporating vitamin D levels (categorical vitamin D deficiency [< 20 ng/ml 25-hydroxy (OH)-vitamin D] versus. sufficiency as well as continuous 25-OH-vitamin D) were conducted. For all comparisons p < 0.05 was considered significant. Season of blood draw was added to statistically significant models. Statistical analysis was performed using SAS 9.4 (SAS, Cary, North Carolina).
Results
Description of Cohort
Demographic and clinical characteristics of the analytical cohort are summarized in Table 1. The mean age was 65 years old, 42% were female, 14% reported Black race, and 34% were current smokers with a median smoking history of 46 pack years (Q1–Q3, 30–57). The total cohort baseline mean % predicted FEV1 was 61.0% predicted (standard deviation [SD] 23% predicted) and % predicted FVC was 91.8% predicted (SD 18.1% predicted). In the cohort, 34% of participants were classified as Global initiative for chronic Obstructive Lung Disease (GOLD)26 stages 3 or 4. Any COPD exacerbation in the year prior to enrollment was reported in 31% of participants and 16% reported a severe exacerbation in the year prior to study enrollment.
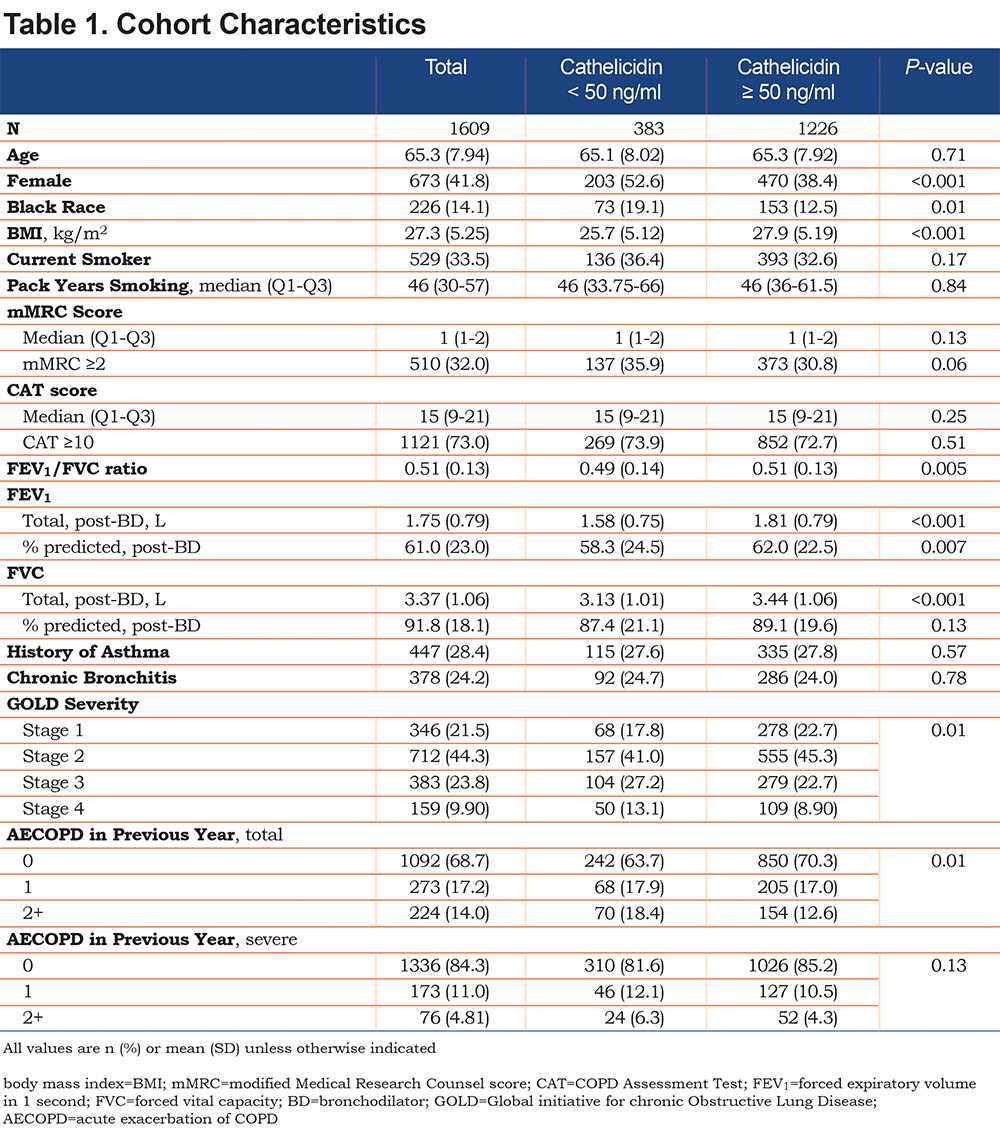
The median plasma cathelicidin level was 61.3 ng/ml (Q1–Q3, 50.6–74.4 ng/ml). There were 383 participants with cathelicidin < 50 ng/ml (lowest quartile) and 1226 with cathelicidin ≥ 50 ng/ml. Participants with cathelicidin < 50 ng/ml were more likely to be female (53% versus 38%; p < 0.001), Black (19% versus 13%; p < 0.001) and have a lower BMI (25.7 kg/m2 versus 27.9 kg/m2; p < 0.001). Those with cathelicidin < 50 ng/ml had a lower baseline FEV1 (1.58 L versus 1.81 L; p < 0.001), FVC (3.13 L versus 3.44 L; p < 0.001), and FEV1/FVC (0.49 versus 0.51; p = 0.005). The % predicted post-bronchodilator baseline FEV1 was also lower among those with cathelicidin < 50 ng/ml (58% predicted FEV1 versus 62% predicted FEV1, p = 0.007). Those with cathelicidin < 50 ng/ml were more likely to have GOLD stage 3 or 4 lung disease (40% versus 32%, p = 0.01), with lower median cathelicidin in more severe GOLD stages (Figure 1). Participants with cathelicidin < 50 ng/ml were more likely to report at least 1 total COPD exacerbation in the year prior to admission (36% versus 30%, p = 0.01). Serum vitamin D and plasma cathelicidin levels were not correlated (r = 0.03, p = 0.23). Of the blood markers previously associated with COPD progression and outcomes, only CRP (r = 0.14, p < 0.001) and total WBC count (r = 0.16, p < 0.001) were statistically correlated with cathelicidin levels, with low correlation coefficients (Online Supplement Table 1).
Associations Between Cathelicidin Lung Function
At baseline, in bivariate analysis cathelicidin < 50 ng/ml was associated with a 3.61% lower % predicted FEV1 (95% confidence interval [CI] -6.26% to -0.97% predicted FEV1; p = 0.007). When modeling cathelicidin continuously, in bivariable linear regression, every 10 ng/ml lower cathelicidin was associated with 0.53% lower % predicted FEV1 (95% CI -0.91% to -0.16% predicted FEV1, p = 0.006) at baseline. At 1 year, in bivariate analysis, cathelicidin < 50 ng/ml was associated with a 4.00% lower % predicted FEV1 (95% CI -7.03% to -0.97% predicted FEV1; p = 0.01). Each 10 ng/ml lower cathelicidin was associated with a 0.53% lower % predicted FEV1 (95% CI -0.97% to -0.16% predicted FEV1; p = 0.005) at 1 year.
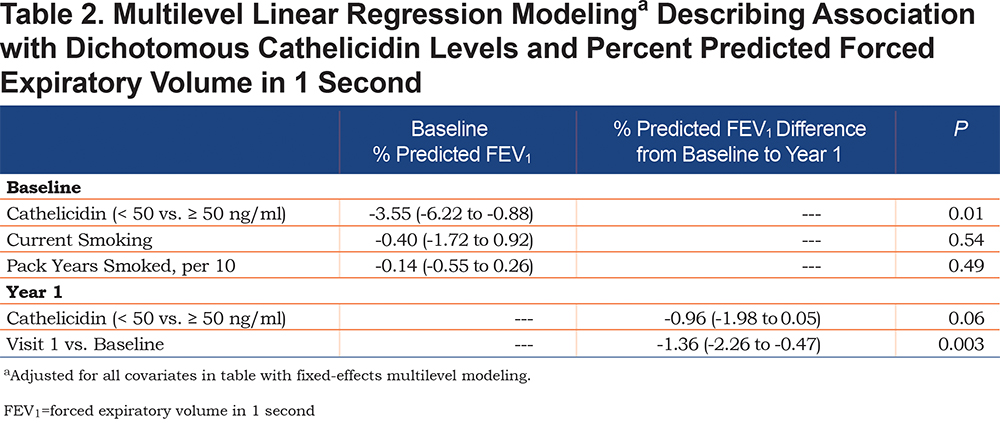
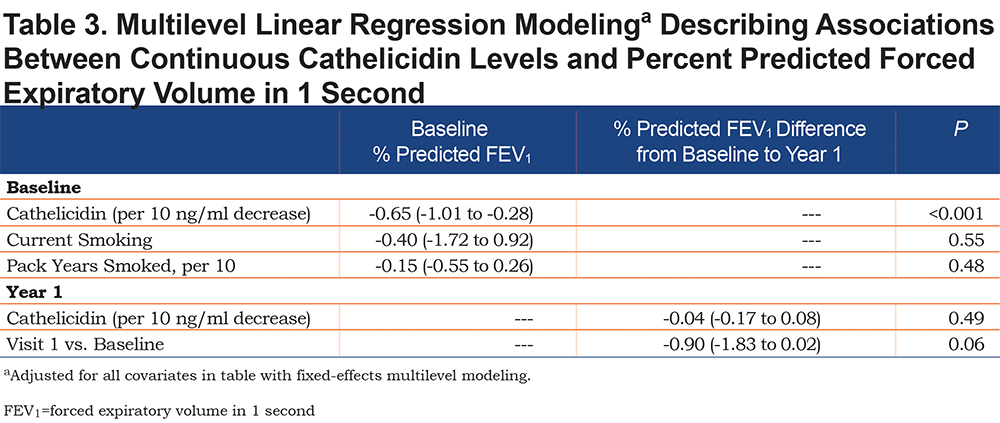
In fixed-effect multivariable linear regression, controlling for current smoking and pack years smoked, cathelicidin < 50 ng/ml was associated with a 3.55% lower baseline % predicted FEV1 (95% CI -6.22% to -0.88% predicted FEV1; p = 0.01) (Table 2). Likewise, every 10 ng/ml lower cathelicidin was associated with a 0.65% lower baseline % predicted FEV1 (95% CI -1.01% to -0.28% predicted FEV1; p < 0.001) (Table 3). Dichotomous and continuous cathelicidin were not associated with lower lung function at 1 year (0.96% lower % predicted FEV1 [95% CI -1.98% to 0.05% predicted FEV1; p = 0.06] and 0.04% lower % predicted FEV1 [95% CI -0.17% to 0.08% predicted FEV1; p = 0.49], respectively).
Associations Between Cathelicidin and Acute Exacerbation of COPD
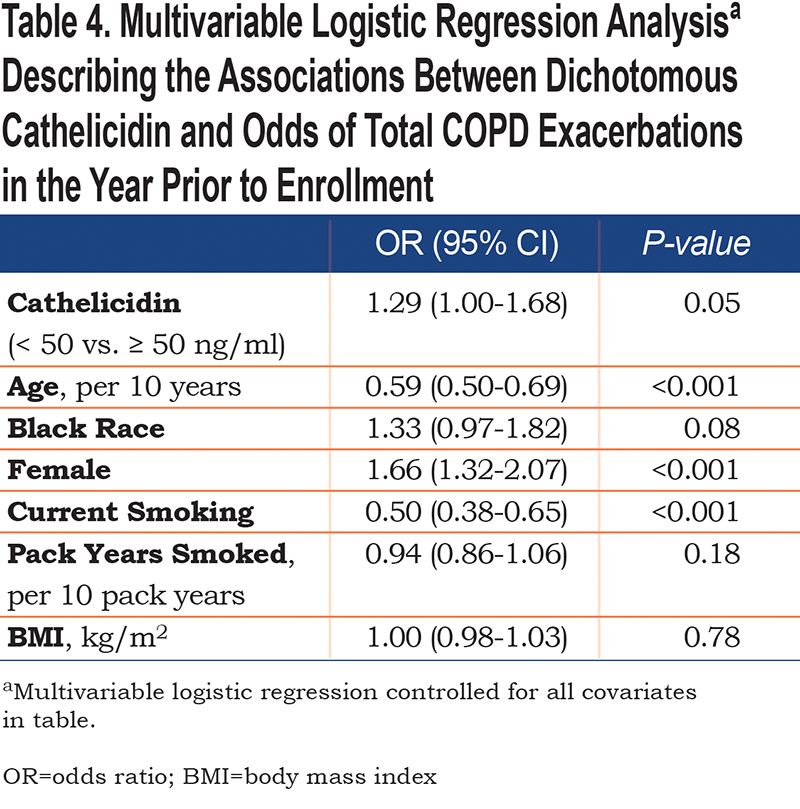
Logistic regression was used to determine the association between dichotomized and continuous cathelicidin levels with the odds of any AECOPD. In bivariate analysis, cathelicidin < 50 ng/ml was associated with a 35% increase in odds of total AECOPD in the past year (95% CI 1.06-1.72; p = 0.02). When controlled for age, race, sex, current smoking, pack years smoked, and BMI, the association was attenuated (odds ratio [OR] 1.29 [95% CI 1.00-1.68]; p = 0.05) (Table 4). Continuous cathelicidin was not associated with increased odds of exacerbation in bivariable logistic regression or when adjusted for the covariates above (OR 1.01 [95% CI 0.97-1.05]; p = 0.76). When assessing associations between continuous or dichotomized cathelicidin levels and longitudinal total AECOPD events, there were no statistically significant bivariate or multivariable relationships observed (Table 5). There was no association between cathelicidin (dichotomized or continuous) and historical or longitudinal severe AECOPD (Online Supplement Tables 2 and 3, respectively).

Sensitivity Analysis
The addition of vitamin D to the multilevel models did not attenuate the associations between continuous cathelicidin and % predicted FEV1 at baseline (0.62% lower % predicted FEV1 for every 10 ng/ml lower cathelicidin [95% CI -0.98% to -0.26% predicted FEV1]; p = 0.001). The relationship between cathelicidin < 50 ng/ml and baseline lung function was attenuated with the addition of vitamin D deficiency to the model (0.96% lower % predicted FEV1 [95% CI -1.96% to 0.05% predicted FEV1]; p = 0.06). The addition of season did not attenuate the association of either categorical or continuous cathelicidin with baseline % predicted FEV1. There was no statistically significant interaction between cathelicidin and vitamin D levels with baseline or longitudinal lung function.
Discussion
In this analysis of 1609 COPD participants from the SPIROMICS cohort, we demonstrate that reduced plasma cathelicidin is independently associated with lower % predicted FEV1 at baseline. There was a non-statistical trend toward lower % predicted FEV1 at 1 year in individuals with cathelicidin < 50 ng/ml. Similarly, low cathelicidin was associated with a trend in increased reported AECOPD events in the year prior to study enrollment. To our knowledge, this is the first description of associations between reduced plasma cathelicidin and lung function decrements in a COPD cohort. These results are strengthened by analyzing data from one of the largest, most comprehensively-phenotyped U.S. COPD cohorts using mixed-model analyses. These findings demonstrate that low cathelicidin identifies those COPD patients with lower lung function and may inform adverse longitudinal lung outcomes.
We have previously shown a relationship between low cathelicidin and decreased longitudinal lung function in smokers without COPD.3 In that study, plasma cathelicidin < 35 ng/ml (representing the lowest quartile in the study cohort) was associated with a 72 ml lower FEV1 at 6 months and a 103 ml lower FEV1 at 18 months from baseline. The findings of this analysis extend the previously described associations between lower plasma cathelicidin and lung function impairments to a population with established COPD. Opposite from the associations in smokers without COPD, we observed an association between baseline FEV1 and low cathelicidin, with a non-statistically significant trend in longitudinal FEV1 decline. These findings may reflect that reduced cathelicidin is a stronger marker in smokers with preserved lung function due to the propensity for those with greater lung function to be more susceptible to lung function declines than those with established, more severe COPD.27 In established COPD, the association between low cathelicidin and reduced baseline lung function could reflect the longitudinal losses that occurred earlier in life. While our findings expand on the potential role of using cathelicidin as a marker of COPD patients at a high risk for lung function impairments, they also suggest that these measurements may be more valuable in at-risk smokers and those with relatively mild obstruction.
Identifying blood-based markers contributing to the development and progression of COPD is a key research goal. Some blood-based markers have been associated with disease progression in various combinations,28 while others have shown variable consistency in associations across large COPD cohorts.21,29-32 In this analysis, plasma cathelicidin levels were weakly correlated with CRP and WBC count. If externally validated in other COPD cohorts, the association between reduced cathelicidin and adverse pulmonary outcomes raises the possibility that this peptide could serve as a potential biomarker to inform risk stratification in COPD.
Low plasma cathelicidin levels may reflect a deficiency in the innate immune system. Studies in cathelicidin gene knock-out mice and cultured epithelium have described decreased viral14 and bacterial clearance,33-35 increased bacterial biofilm formation,36,37 differential mucociliary cell activity,38 and decreased epithelial integrity39 in cathelicidin-deficient states. Host-microbiome interactions and the response to environmental irritants may be altered by less robust innate immunity40 leading to colonization of more pathologic bacteria in the airways, further propagating the dysbiosis and inflammatory cycle characteristic of COPD.41 This potential mechanism is supported by previous observations of increased incident pneumonia in cathelicidin-deficient states.2 In COPD, infection risk can manifest through COPD exacerbations. We did not observe an independent association between reduced cathelicidin level and increased COPD exacerbation risk. This lack of an association may be due to underpowering given the few COPD exacerbation events in SPIROMICS.42 Alternatively, cathelicidin may not be implicated in the causal pathway for exacerbations.
Cathelicidin is regulated by vitamin D.9 In our analysis, plasma cathelicidin was not correlated with serum vitamin D. The addition of vitamin D attenuated the relationship between baseline lung function and categorical cathelicidin. Associations between blood vitamin D and cathelicidin levels have been inconsistent.2,3,17 Mechanistically, vitamin D interacts with the immune system through multiple pathways.43-46 Investigations have shown increased ASL cathelicidin levels and antimicrobial activity after supplementation with vitamin D, suggesting biological plausibility of an in vivovitamin D-cathelicidin relationship.47 Randomized controlled trials assessing vitamin D supplementation to prevent infectious episodes have enrolled patients who were at or near vitamin D sufficiency,48,49 potentially selecting for cohorts less likely to see beneficial response from supplementation. Cathelicidin levels were not measured in these trials. Our study suggests that a relationship between cathelicidin and lung function may not be independent of vitamin D and may inform future studies using plasma cathelicidin levels or antimicrobial activity as a marker for response to vitamin D supplementation in trials including participants with COPD.
This study has limitations. As an observational study, our results are insufficient to establish a causal relationship between low cathelicidin and lung function decline. This study would benefit from external validation in additional COPD cohorts. Given the relatively low frequency of AECOPD events in the SPIROMICS cohort,42 we may be underpowered to detect a true association between AECOPD and cathelicidin. Similarly, 1 year is a short period to determine longitudinal changes in FEV1. However, this limitation illustrates the need for determining the stability and meaningfulness of cathelicidin levels and relationships with lung function decline over time. Studies have reported serum16 and sputum16-19 cathelicidin measurements in COPD. Whether cathelicidin concentration in the blood is reflective of the levels and activity in the airways is not fully determined,10 but blood levels have been associated with meaningful COPD outcome2,3 and increase under the stress of microbial infections or noxious stimuli similar to sputum levels.16 Similarly, we do not have microbiome data as part of this analysis. Medication usage (e.g., azithromycin or inhaled corticosteroids) was not included in our models and could potentially introduce residual confounding, although literature on these agents and plasma cathelicidin is not available.
In conclusion, we report an independent association between lower plasma cathelicidin and reduced lung function at baseline. Low cathelicidin was associated with trends toward lower lung function at 1 year that did not reach statistical significance. There was no robust association between cathelicidin and COPD exacerbations. Our findings extend the observations of reduced cathelicidin and impaired lung function from at-risk individuals to those with established COPD. This study implicates cathelicidin as a measurable component of the innate immune system potentially informative of the risk of lung function impairments in COPD. If validated in other COPD cohorts, plasma cathelicidin may serve as a potential marker to identify COPD patients at increased risk for adverse lung outcomes. Ultimately, mechanisms to augment cathelicidin may serve as a pharmacological target to improve the health of individuals at risk or with COPD.
Acknowledgements:
The authors thank the SPIROMICS participants and participating physicians, investigators and staff for making this research possible. More information about the study and how to access SPIROMICS data is at www.spiromics.org. We would like to acknowledge the following current and former investigators of the SPIROMICS sites and reading centers: Neil E. Alexis, PhD; Wayne H. Anderson, PhD; R. Graham Barr, MD, DrPH; Eugene R. Bleecker, MD; Richard C. Boucher, MD; Russell P. Bowler, MD, PhD; Elizabeth E. Carretta, MPH; Stephanie A. Christenson, MD; Alejandro P. Comellas, MD; Christopher B. Cooper, MD, PhD; David J. Couper, PhD; Gerard J. Criner, MD; Ronald G. Crystal, MD; Jeffrey L. Curtis, MD; Claire M. Doerschuk, MD; Mark T. Dransfield, MD; Christine M. Freeman, PhD; MeiLan K. Han, MD, MS; Nadia N. Hansel, MD, MPH; Annette T. Hastie, PhD; Eric A. Hoffman, PhD; Robert J. Kaner, MD; Richard E. Kanner, MD; Eric C. Kleerup, MD; Jerry A. Krishnan, MD, PhD; Lisa M. LaVange, PhD; Stephen C. Lazarus, MD; Fernando J. Martinez, MD, MS; Deborah A. Meyers, PhD; John D. Newell, Jr., MD; Elizabeth C. Oelsner, MD, MPH; Wanda K. O’Neal, PhD; Robert Paine, III, MD; Nirupama Putcha, MD, MHS; Stephen I. Rennard, MD; Donald P. Tashkin, MD; Mary Beth Scholand, MD; J. Michael Wells, MD; Robert A. Wise, MD; and Prescott G. Woodruff, MD, MPH. The project officers from the Lung Division of the National Heart, Lung, and Blood Institute were Lisa Postow, PhD, and Thomas Croxton, PhD, MD.
Author contributions: RMB, ASC, MBD conceptualized the study. ASC and RMB performed data analysis. RMB and MBD drafted the original manuscript. All coauthors were involved in collection of data, interpretation of of data, and critical review and revision of this manuscript.
Declaration of Interest
RMB is supported through grants from the National Institutes of Health. APC receives grants from the National Institutes of Health and receives consulting fees from VIDA. JMW receives grants from the National Institutes of Health. GJC receives grants from the National Institutes of Health and receives consulting fees from Novartis, AstraZeneca, Respironics, MedImmune, Actelion, Forest, and Pearl. REK receives contracts and grants from the National Institutes of Health. RP receives grants from the National Institutes of Health and research monies from the COPD Foundation. SAC receives grants from the National Institutes of Health and consulting fees from GlaxoSmithKline, Amgen, Glenmark, Sunovion, Genetech and MedImmune. CBC receives grants from the National Institutes of Health and contracts from the National Institutes of Health Foundation. IZB has consulting fees from Amgen, Theravance, GE Healthcare, AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim and Verona Pharma. JAK receives grants from the National Institutes of Health and the Patient-Centered Outcomes Research Institute and consulting fees from Inogen. MKH receives grants from the National Institutes of Health. JLC receives grants from the National Institutes of Health. NNH receives grants from the National Institutes of Health and the COPD Foundation and has contracts from AstraZeneca, Boehringer Ingelheim and Mylan. RAW receives consulting fees from AstraZeneca, Boehringer Ingelheim, Contrafect, Pulmonx, Roche, Spiration and Sunovion. MBD receives grants from the National Institutes of Health. ASC, DJC, SPP, and WWL have nothing to declare.