Running Head: The APEX COPD Registry Delphi Study
Funding statement: APEX COPD is conducted by Optimum Patient Care Global Limited, and co-funded by Optimum Patient Care Global and Boehringer Ingelheim Pharmaceuticals, Inc.
Date of acceptance: July 13, 2020
Abbreviations: chronic obstructive pulmonary disease, COPD; Advancing the Patient Experience in COPD, APEX COPD; electronic health records, EHRs; patient-reported information, PRI; patient reported health outcomes, PROs; Global initiative for chronic Obstructive Lung Disease, GOLD; Round 1, R1; Round 2, R2; Round 3, R3; American Thoracic Society, ATS; European Respiratory Society, ERS; modified Medical Research Council, mMRC; Optimum Patient Care, OPC; COPD-X Australian and New Zealand Guidelines, COPDX; point of care, POC; APEX COPD Operational Management Group, AOMG; inhaled corticosteroid, ICS; peak inspiratory flow rate, PIFR; forced expiratory volume in 1 second, FEV1; forced vital capacity, FVC; body mass index, BMI; alpha-1 antitrypsin deficiency, AATD; asthma/COPD overlap syndrome, ACOS; respiratory tract infection, RTI; antibiotic, Atb; oral corticosteroid, OCS; chest X-ray, CXR; computed tomography, CT; blood pressure, BP; coronary heart disease, CHD; congestive heart failure, CHF; gastroesophageal reflux disease, GERD; short-acting beta2-agonist, SABA; long-acting beta2-agoinist, LABA; short-acting muscarinic antagonist, SAMA; long-acting muscarinic antagonist, LAMA; phosphodiesterase, PDE; leukotriene receptor antagonist, LTRA; metered-dose inhaler, MDI; electronic cigarette, E-cigarette
Citation: Edwards CL, Kaplan AG, Yawn BP, et al. Development of the Advancing the Patient Experience in COPD registry: a modified Delphi study. Chronic Obstr Pulm Dis. 2021; 8(1): 135-151. doi: http://doi.org/10.15326/jcopdf.2020.0154
Online Supplemental Material: Read Online Supplemental Material (227KB)
Introduction
Chronic obstructive pulmonary disease (COPD) is managed predominantly by family physicians, but little is known about how this prevalent disease is managed in primary care, or how primary care management in the United States may be improved. Management of this disease is daunting when one considers the sheer size of the population (16 million U.S. adults and rising),1,2 the continuing rise in COPD-related mortality,3,4 the high symptom burden experienced by patients,5 and the cost to the U.S. economy (predicted $50 billion by the end of 2020).6 The prevalence and burden of COPD are predicted to increase over the coming decades due to continued exposure to COPD risk factors (tobacco smoking, air pollution) and aging of the population.7 Although the Global initiative for chronic Obstructive Lung Disease (GOLD) strategy provides clear strategies for COPD diagnosis and management,8 they are often not fully understood nor implemented in primary care practice.9 COPD remains a disease which is under- and mis-diagnosed, resulting in delayed and/or sub-optimal disease management.10-12 The question remains, how do we ensure optimum management of COPD patients in primary care?
Both primary care- and patient-related factors make this a difficult question to answer. The issues in primary care include a reticence to diagnose COPD in already multimorbid patients, the temptation to prescribe antibiotics for patients who present with chest infections/bronchitis rather than delving into a COPD diagnosis protocol, and a lack of understanding (or indeed, availability) of spirometry.12 The issue of COPD under-diagnosis may be further exacerbated by failure to recognize GOLD Group C patients. These patients are not particularly symptomatic but do experience a substantial number of chest infections.8 They, therefore, often remain under the care of their family physician and never receive specialist referral. These issues represent significant hurdles to optimized COPD management in primary care, since establishing and acting on an early diagnosis of COPD is a critical step in reducing the extensive morbidity and mortality of this disease. Large-scale efforts to promote awareness of COPD and encourage early diagnosis have been undertaken, to tackle these issues and others in COPD management (e.g., the National Lung Health Education Program in the United States.). Patient-related hurdles to optimized COPD management include disease denial (lack of understanding, under-estimation of disease impact), poor adherence, lack of patient engagement and empowerment, variable disease presenting patterns, and cost-related issues.12
In order to improve the management of COPD in primary care, it is first necessary to describe the patient population in a standardized way, using variables which are clinically relevant, and which can be practically collected and monitored longitudinally. This information should be relevant to both physicians and patients to encourage therapeutic, shared decision-making and ultimately better adherence. A COPD registry is one way to achieve these aims. Registries are well-established tools for tracking and reporting epidemiological disease trends, treatment benefits and risks that can be longitudinally monitored. They are also useful to track the natural progression of disease, which may be particularly relevant in COPD where progression is slow, and patterns can be difficult to spot. They have the potential to improve diagnostics and be used to inform treatment algorithms.13 Although both national and regional COPD registries and patient cohorts do already exist in the United States, (e.g., the COPD Genetic Epidemiology study 14 the COPD Patient-Powered Research Network,15 and others hosted by universities and health care networks), none are based in primary care. Those based in secondary care focus on patients with more severe disease, missing the milder and moderate severity patients. None have captured information on how patients are managed in primary care in real life.
The Advancing the Patient Experience in COPD (APEX COPD) registry (https://www.apexcopd.org/) will be the first U.S. primary care health system-based registry, designed to follow these patients longitudinally, investigate how they are managed in real-life settings and the consequence(s) of various management strategies. The overall aim is to improve primary care for patients with a diagnosis of COPD by capturing clinically-relevant and high-quality data using a standardized set of variables, from multiple sources, in sufficient numbers of patients to ensure representativeness to the wider COPD population, and to answer key research questions relating to COPD in primary care. The registry plans to bring together information captured in electronic health records (EHRs), and information provided by patients themselves (i.e., from questionnaires and during office visits). This will be achieved using standardized data collection, guided by COPD clinicians both in primary and specialist care. Further, the registry may identify patterns of health care before a diagnosis,16 and has the potential to identify new COPD phenotypes.
The aim of the Delphi exercise described in this article was to gain expert consensus on a standardized list of variables on demographic, disease monitoring and treatment variables to establish the APEX COPD registry. Selection of these variables was dictated not only by clinical relevance; it was also important that variables were already known to family physicians and that it was practical and feasible to collect them in primary care.
Methods
Design
This study used a modified, 3-round Delphi process to achieve consensus on the core variables to be collected in the APEX COPD registry.17 Variables were initially selected from relevant COPD guidelines and recommendations to give all potentially clinically relevant options, and subsequently refined by the panel to the items desired for inclusion in the registry.
Panel Selection
The APEX COPD Delphi panel consisted of appropriately qualified and experienced individuals in the field of COPD and primary care, capable of providing critical and informed input. This panel included 14 experts in primary and specialist care from the United States and internationally – 5 family physicians, 3 pulmonologists, 6 respiratory researchers (5 of whom had substantial prior experience as family physicians), with >70% of panel members based in the United States. (Table E1 in the online supplement). The panel members met 2 or more of the following criteria:
- Evidence of relevant COPD research published in high-ranking peer-reviewed journals (e.g., high number of citations and research items).
- A history of participation in the development and/or management of 1 or more respiratory registries or cohorts, epidemiological databases, and scientific congress committees in a country and/or internationally.
- Experience as a medical clinician (e.g., physician or nurse) with an interest in advancing COPD management in clinical practice.
Modified Delphi Process
A modified Delphi process was used to reach consensus on which data to collect into the registry from EHRs, patient-reported information/patient-reported outcomes (PRI/PROs), and at consultation.18 The process consisted of 3 iterative rounds (Round 1 [R1], Round 2 [R2] and Round 3 [R3]; Figure 1).
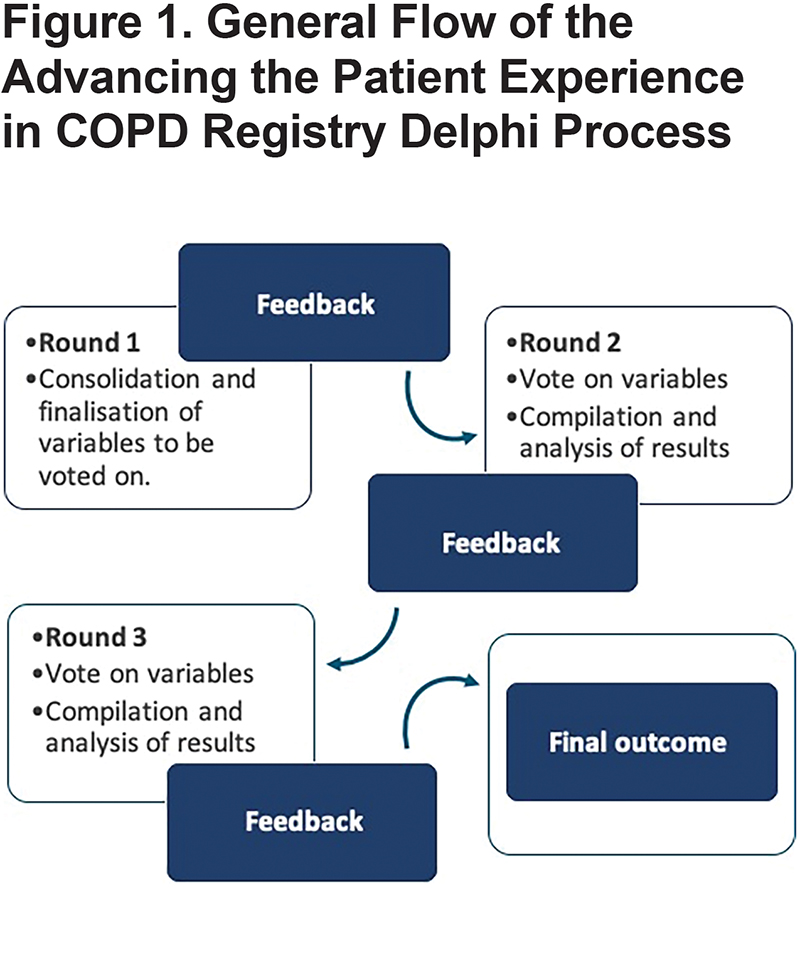
Each Delphi panel member was issued an electronic APEX COPD Excel workbook to review, provide suggestions and vote, to select core variables. Members then returned the completed Delphi workbooks to the APEX COPD administrator within a 4-week time period. The Delphi administrator directly corresponded with all panel members individually to ensure anonymity of replies and was responsible for disseminating workbooks and result summaries for each round.
Delphi Round 1
The Delphi workbook (APEX COPD Workbook R1) was developed initially by consolidating variables from current guidelines and recommendations: the American Thoracic Society (ATS) and European Respiratory Society (ERS) joint guidelines, the COPD Foundation guidelines, and GOLD.19-26 Variables under consideration included:
- Patient demographics.
- Medical history, symptoms (COPD-relevant), prior exacerbations, exposure, and comorbidities.
- COPD treatment and management, including medications and side effects (such as those related to steroid exposure and/or biologics), adherence data when available, vaccinations, referrals, surgery, rehabilitation, smoking cessation, and other non-pharmacological strategies.
- Patient-reported information and outcomes including health status scores (COPD Assessment Test, modified Medical Research Council dyspnea scale [mMRC]), and questionnaires (e.g., inhaler satisfaction questionnaire, and the Test of Adherence to Inhalers questionnaire) to measure respiratory inhaler device satisfaction and adherence.
- Medical test/investigations, including spirometry, electrocardiogram, and biomarkers (blood eosinophils, immunoglobulin E, and fractional exhaled nitric oxide where possible).
The workbook comprised a 2-tab Excel spreadsheet:
- On tab one, displaying the potential core list (Figure 2), panel members were required to select an option (“Yes” or “No”) via a drop-down menu for each variable, indicating whether or not they concurred that the variable would be part of the APEX COPD registry core variables list.
- On tab 2, panel members were encouraged to nominate variables from the “Additional” variables list (Figure 3) and/or propose new variables (“Suggested”). During this round, experts were also encouraged to provide comments for excluding or including variables.
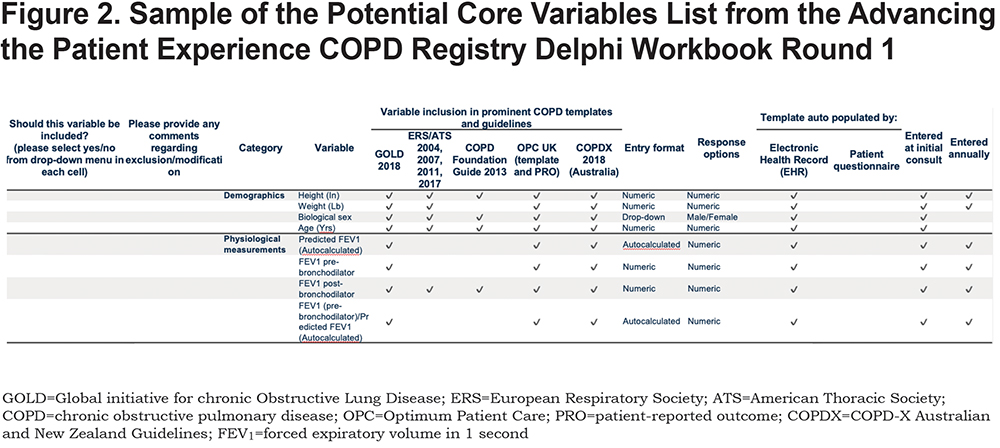
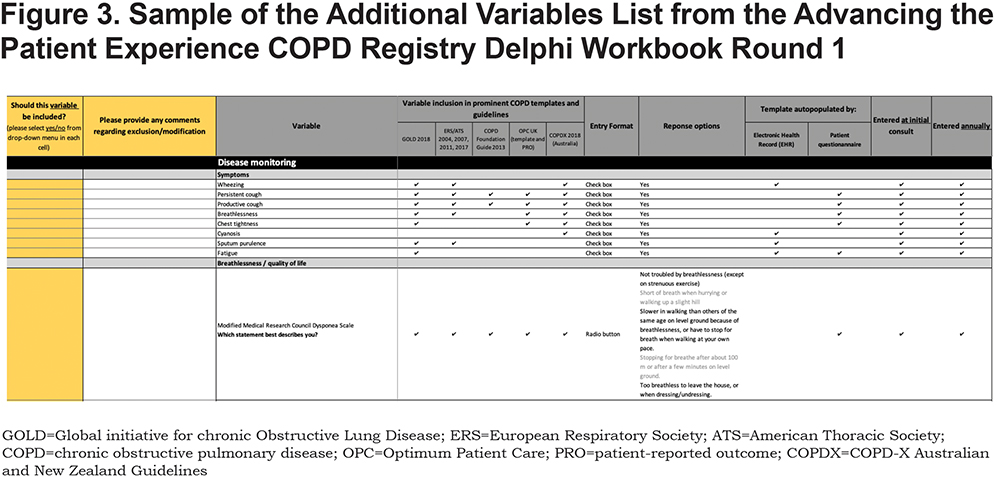
At round closure, the Delphi administrator anonymized all returned workbooks and compiled all replies to tabulate frequency of responses, “Yes” and “No,” for each variable on the lists. Variable consensus was evaluated using summary statistics (frequency counts) generated with the Microsoft Excel V16.27 statistical package. Delphi R1 consensus rules for each variable assessed by the panel were as follows: Keep (>66% “yes”); undecided (≥50% to ≤66% “yes”); exclude (<50% “yes”).
Delphi Round 2
All variables from R1 as well as “suggested” variables were included in a single tab in the R2 workbook and the expert panel was requested to engage in a similar voting process for Delphi R2. The Delphi R1 summary results and panel member comments (“Comments”) were anonymized and provided in the R2 workbook to facilitate an informed decision. Delphi R2 was divided into 2 parts:
- Round 2a: Each variable that received a 66% or more consensus from the Delphi panel in R2 was moved to a second phase of analysis (R2b). Other variables were excluded from the APEX COPD registry core variable list.
- Round 2b: Variables moved to R2b were analyzed more specifically to determine which data sources they should be collected from (EHRs, PRI/PRO, or at the doctor’s office [i.e., point of care (PoC)]).
Delphi R2 consensus rules for each variable assessed by the panel were as follows: Keep (≥66% consensus); undecided (40% to 65% consensus); exclude (<40% consensus). Additionally, all excluded variables from R2 were vetted by the APEX COPD Operational Management Group (AOMG) (Table E2 in the online supplement). If excluded variables were considered key to COPD primary care by the AOMG, they were re-included for review in R3.
Delphi Round 3
The Delphi panel also took part in a similar voting process for Delphi R3 via a third electronically distributed workbook (The APEX COPD Delphi Workbook R3). The Delphi R2 summary results and panel member comments (“Comments”) were anonymized and provided in the R3 workbook to facilitate an informed decision. R2 “Undecided” and additional AOMG vetted variables were included in the R3 workbook. Delphi R3 consensus rules for each variable assessed by the panel were as follows: keep (≥66% consensus); undecided (40% to 65% consensus); exclude (<40% consensus). All undecided and excluded variables from R3 were vetted by the AOMG. If these variables were considered key to COPD primary care by the AOMG, they were included in the final core variable list.
Results
Delphi Round 1
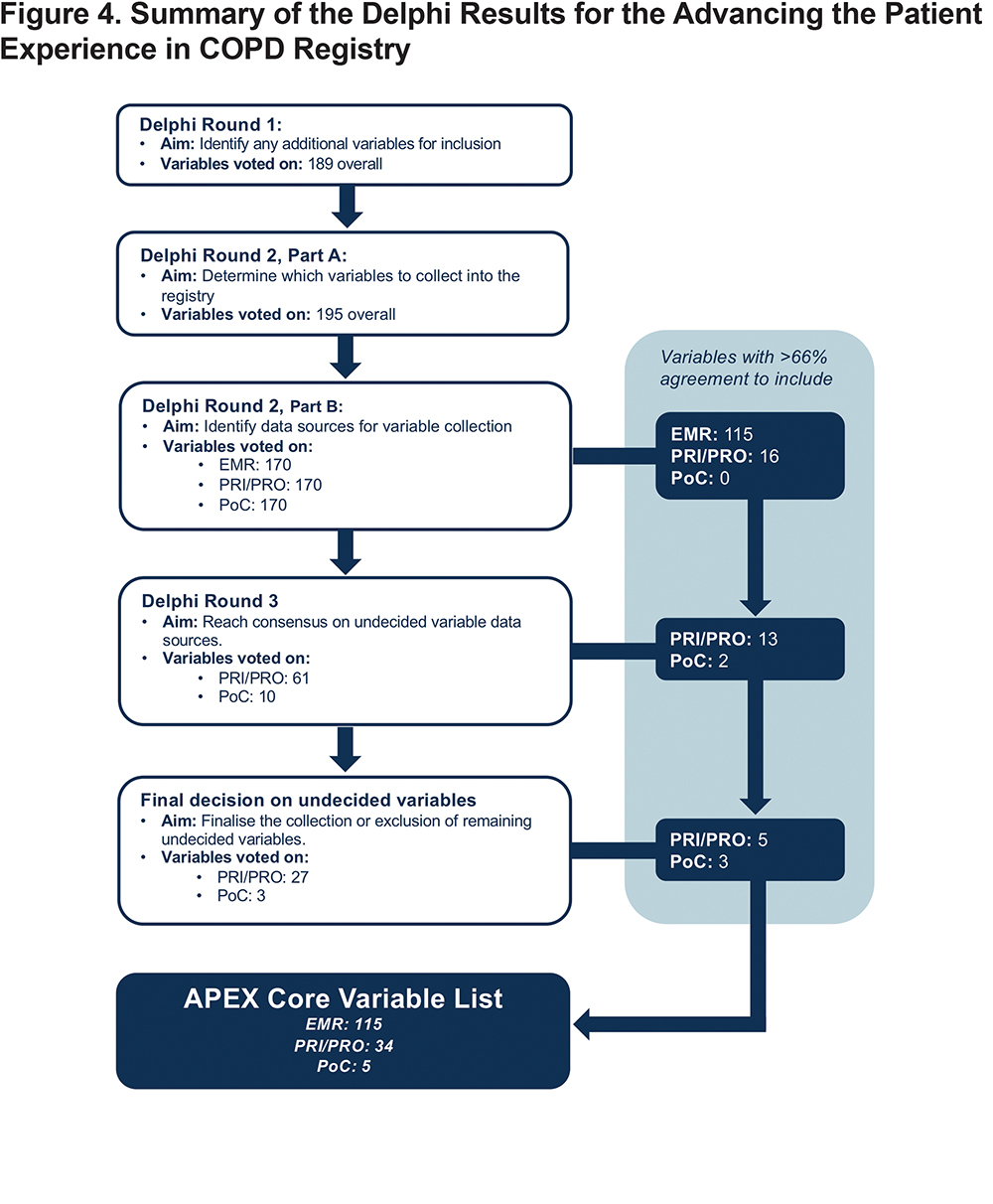
In R1, the expert panel voted on 189 clinical COPD variables belonging to the categories of demographics, disease monitoring, and treatment (Table E3 in the online supplement). Overall, 149 of the variables received >66% consensus to keep, 25 were undecided (50% to 66% consensus), and 15 were recommended to exclude (receiving <50% consensus) (Table E4 in the online supplement). All 189 variables were entered into voting R2. Six “suggested” variables recommended by the panel were also added bringing the total to 195 variables to proceed to R2.
Delphi Round 2
After voting R2, 25 of the 195 variables were excluded from collection into the registry and 170 were confirmed for collection (Table E5, Part A in the online supplement). The Delphi panel recommended that of the 170 confirmed variables, 115 be collected from EHRs, and 16 via PRI/PRO. At this stage, no variables were confirmed for collection by clinicians during a visit. (Table E5, Part B in the online supplement). Undecided variables were entered into Delphi R3. Four undecided PRI/PRO variables from R2a were re-included for review in R3. These were:
- Poor appetite: an important factor used in conjunction with other cancer indicators which received consensus votes to be included for collection.
- Easy bruising: specific types of physiological side effects were not specified for voting; bruising is a common and important side effect to assess in patients receiving inhaled corticosteroids (ICSs).
- Pain (headache and muscle): specific types of pain as side effects were not specified for voting; muscle pain and headaches are common and preventable effects to assess in patients receiving inhaled medications.
- Low birth weight: an important childhood risk factor which may not be recorded in patients’ EHRs.
One excluded PoC variable from R2a was re-included for review in R3. This was:
- Inhaler technique assessment: important for interpretation of peak inspiratory flow rate (PIFR) which received a consensus vote to be included for collection from the EHR and an undecided vote to be collected during the office visit.
Delphi Round 3
A final round of voting (R3) was undertaken to vote on “undecided” PRI/PRO and PoC variables from R2. A total of 13 PRI/PRO and 2 PoC variables were kept on consensus. Of the remaining 27 undecided PRI/PRO variables at R3, a total of 3 were confirmed for collection by the AOMG (Table E6 in the online supplement). The reasons were:
- Pulmonary rehabilitation: critical for prevention of disease progression and management.
- Influenza vaccine: can be administered by an external provider and therefore may not be collected in patients’ EHRs. This information is critical for informing preventative care.
- Oral treatment side effect (candidiasis): specific types of oral side effects not specified for voting; oral candidiasis is a common and important side effect to assess in patients receiving ICSs.
Two additional PRI/PRO variables excluded in R2b were vetted and included in the final core variable list. These were:
- Asthma diagnosis (age of onset): decision to collect via PRI/PRO in addition to the EHR to identify age of onset where this information is unavailable in the EHR.
- Physiological treatment side effect (easy bruising): specific types of physiological side effects were not specified for voting; bruising is a common and important side effect to assess in patients receiving ICSs.
Of the 3 undecided variables for collection during the office visit at R3, all were confirmed for collection by the AOMG. These were:
- Number of severe exacerbations in the past year,
- Forced expiratory volume in one second (FEV1) post-bronchodilator, and
- Forced vital capacity (FVC) post-bronchodilator
These were all considered critical for COPD management and will be collected at PoC only if missing from the EHR and PRI/PRO.
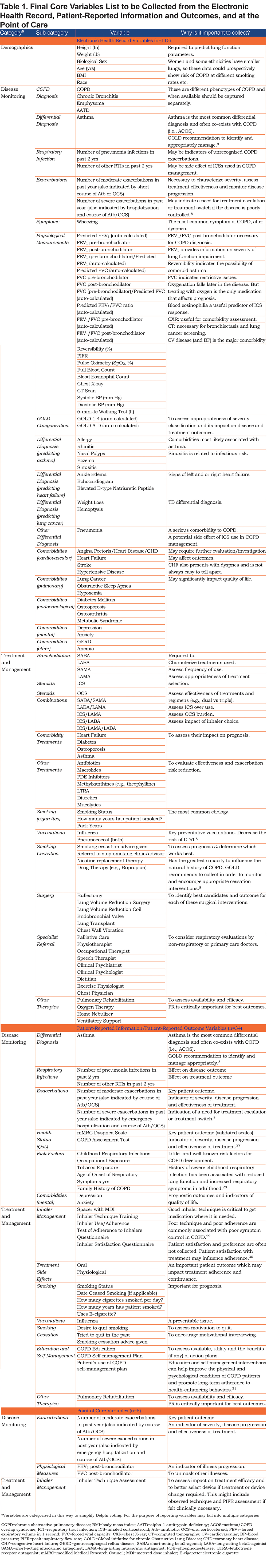
The core variables that achieved consensus via the closely guided 3 rounds of Delphi were included in the final core variable list and their importance described (Table 1).8,22,27-31
Discussion
Using the knowledge and experience of an international panel of COPD experts, workable criteria for registry purposes, a standardized core set of variables, and a potential method to unify data for COPD in the United States were generated and agreed to by consensus. All potential variables underwent a rigorous, stepwise consensus process to ensure the collection of the minimum required information to effectively and practically study the diagnosis and management of patients with COPD. Of the initially circulated "potential core" and "suggested" variables, 115 variables from existing EHRs, 34 PRI/PRO variables and 5 variables to be collected during office visits, were selected for integration into the APEX COPD registry (Figure 4). These selected variables fall into 3 broad categories (i.e., demographics, disease monitoring, and treatment), and should include information on diagnosis, exacerbations, symptoms, lung function and quality of life, comorbidities, smoking history, treatment specifics (including side effects), inhaler management (including inhaler technique) and education/self-management. They have been selected not only due to their clinical relevance and usefulness to family doctors and patients (Table 1) but also with feasibility, familiarity and practicality of collection in mind. This will ensure that the APEX COPD registry will be an asset to family doctors; a tool to identify how patients with COPD are managed in real life, in a population rarely included in randomized controlled trials. Following ratification of data collection, the registry plans to integrate information from multiple sources with maximal efficiency and present it to clinicians and patients in a structured and clinically useful format, with the aim of improving primary care for patients diagnosed with COPD. Data from the registry will also be used to answer key research questions relating to COPD in primary care, facilitating insight into this prevalent chronic disease. The outcomes of such research and any new research proposals will be continuously updated via the APEX COPD website (https://www.apexcopd.org/).
The panel-approved APEX COPD registry variables were chosen to ensure a comprehensive description of patients diagnosed with COPD and managed in real-life clinical practice among family physicians in the United States. Collection of baseline information on diagnosis, infection, exacerbations, severity classification, health status, and treatment-/comorbidity-patterns will provide a snapshot of clinical phenotypes of COPD, a better understanding of how patients are diagnosed and managed in primary care (e.g., use of spirometry), an estimation of the burden of disease (including the corticosteroid burden), and an assessment of whether diagnoses and severity classifications are correct and treatment is appropriate (compared to guideline recommendations).8 Appropriate variables will be assessed longitudinally to examine their impact on disease progression and treatment outcomes. For example, data may be assessed to: (1) compare the clinical, safety and cost-effectiveness of current COPD treatments; (2) describe treatment changes over time (and the reasons for those changes); (3) assess the impact of inhaler technique and inhaler type on key outcomes; (4) analyze risk factors associated with disease progression and health care utilization; and (5) predict response to treatment (e.g., biomarkers).
As well as the collection of key COPD variables from multiple sources, the APEX COPD registry has numerous other assets, including: (1) its size and scope; (2) innovative use of technology to collect high quality data; (3) inclusion of clinical and database management expertise; (4) inclusion of expertise on gathering patient-reported information; (5) an integrated communication strategy; and (6) the organizational structure to oversee the initiative and ensure its continuance. Currently, it is planned to capture information from 3000+ patients diagnosed with COPD, with a wide geographic coverage throughout the United States, benefiting from both scale and generalizability to the wider COPD population. Patients included will have a diagnostic, monitoring, or review code for COPD prior to or at consultation and be aged ≥35 years at COPD diagnosis. Data collected by the APEX COPD registry will be maintained as a limited dataset in the APEX COPD database. Data will be completely de-identified, at the individual level, and anonymized when providing subsets of data for research purposes. Electronic data capture systems will be utilized to capture data directly from EHRs, which may already include valuable information on symptoms, lung function, COPD staging, pharmacologic treatment, comorbidities, and exacerbations. Use of an existing data resource to populate the APEX COPD registry precludes the need for lengthy additional data collection at the PoC, which will improve efficiency, reduce workload, time, and cost, and enhance the quality of data collected.
Expertise is embedded into the initiative, including the panel of 14 COPD experts on the APEX COPD Steering Committee, recruitment of primary care consultants experienced in COPD management, incorporation of a dedicated communications team to disseminate key research findings and partnership with experts in PRI and PRO (the COPD Foundation and the American Academy of Family Physicians), and database management and registry delivery (DARTNet Institute, Aurora, Colorado). Communication of APEX COPD registry research findings will also be facilitated via regular publication in peer-reviewed journals and dissemination of findings at international and regional scientific meetings. The APEX COPD registry is overseen by 5 bodies (Optimum Patient Care Global, the Respiratory Effectiveness Group, the Anonymized Data Ethics and Protocol Transparency Committee, the American Academy of Family Physicians, and the APEX COPD Steering Committee) safeguarding continuance of the registry into the future, and ensuring APEX COPD research is ethical, clinically appropriate and continues to bring genuine value to physicians who manage COPD in real-life clinical practice, and to patients who live with COPD.
Strengths and Weaknesses
Fourteen Delphi panel members from 4 countries (>70% U.S.-based experts) participated in 1 or more Delphi rounds, to allow for broad consensus to be obtained, and to ensure recommendations were pertinent not only to the United States, but also maintained applicability beyond U.S. borders. This approach dilutes the opinion of a single expert, so bias is decreased and diversity within the expert panel is maximized. Panel members were chosen for their expertise in the research field, and relevant medical practice and experience. The anonymity of the survey ensured all opinions were given equal weight and consideration. The Delphi process was carried out online, to facilitate ease of yes/no voting for each variable, as well as rapid and accurate vote counting and classification (i.e., yes, no, undecided) at the end of each Round. It also facilitated rapid and open communication among the COPD experts. The results covered a wide range of areas where consensus was achieved. Although the study employed a relatively small Delphi panel, recent studies have found that reliable outcomes can be obtained with a relatively small number of Delphi experts.32 The Delphi panel was also not fully representative of the diversity of stakeholders involved in respiratory care at the primary care level. In particular, the opinions of payers and patients were not solicited. Another limitation of the study is that the response rate was not 100%; a total of 13 of 14 experts (93%) responded to all 3 Delphi rounds. However, there was consistency in the number of experts who participated in each round (R1=93%; R2=100%; R3=100%), which ensured that the possibility of reaching consensus was conserved.
In conclusion, COPD experts have agreed on core variables to collect in the APEX COPD registry. The majority of these variables will be extracted from EHRs but will also include PRI/PRO and PoC data from 3000+ patients diagnosed with COPD across the United States. Data will be integrated, standardized and stored in the APEX COPD database and made available for COPD-related research. It will be used to analyze COPD natural history as well as clinical, safety and cost-effectiveness of current COPD treatments in primary care across the United States.
Acknowledgments
Author contributions: CE, LB, VC, and CP contributed to the concept and/or design of this study. Acquisition and analysis of data was provided by CE, MK, CLL and DP. Interpretation of results was provided by CE, AK, BY, JWK, KLC, CF, GG, MH, CM BM, WP AS, NS and DP. All authors have revised each draft, contributed to the intellectual content of the articles and given final approval of the version to be published. All authors agree to be accountable for all aspects of the work, ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors.
We thank Alvaro Aranda, MD, (Hospital Auxilio Mutuo, San Juan, Puerto Rico) for his scientific and clinical contributions during the drafting of this manuscript. We also thank Audrey Ang for editorial assistance, Bronte Sawyer for project coordination, and Lisa Buttle, PhD, for assistance with drafting the article. Ruth B. Murray, PhD is acknowledged for her substantial contribution to the interpretation, summarization and presentation of data in this article and significant intellectual input to the manuscript. Dr. Murray is the founder and director of Medscript Ltd., a company that provides writing and editorial support for APEX COPD publications.
The authors received no direct compensation related to the development of the manuscript. Writing, editorial support, and/or formatting assistance was provided by Audrey Ang of the Observational and Pragmatic Research Institute, Singapore, and Dr. Buttle of Medscript Ltd, Ireland, which was funded by Boehringer Ingelheim Pharmaceuticals, Inc.
Boehringer Ingelheim was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Declaration of Interest
Chelsea Edwards is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Alan Kaplan is a member of the advisory board of, or speakers bureau for, Astra Zeneca, Boehringer Ingelheim, Grifols, GlaxoSmithKline, Merck Frosst, Novo Nordisk, Novartis, Paladdin, Pfizer, Purdue, Sanofi, Teva, and Trudel. Barbara Yawn has served on COPD-related advisory boards for GlaxoSmithKline, AstraZeneca, Novartis, and Boehringer Ingelheim, and received COPD-related investigator-initiated research funds from GlaxoSmithKline, Boehringer Ingelheim, AstraZeneca, and Novartis. Janwillem W. H. Kocks declares grants and personal fees from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, and Novartis, and grants from Chiesi, Mundipharma and Teva. Lakmini Bulathsinhala is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Victoria Carter is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Ku-Lang Chang and Chester Fox declare no conflicts of interest. Gokul Gopalan is a former employee of Boehringer Ingelheim, which is a co-founder of the APEX COPD initiative and current employee of Vertex Pharmaceuticals. MeiLan Han reports consulting for Boehringer Ingelheim, GlaxoSmithKline and AstraZeneca, and research support from Novartis and Sunovion. Maja Kruszyk is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Chantal Le Lievre is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Cathy Mahle is an employee of the company Boehringer Ingelheim, which is a co-founder of the APEX COPD initiative. Barry Make reports funding from the National Heart, Lung and Blood Institute for the COPD Genetic Epidemiology study; grants and medical advisory boards from Boehringer Ingelheim, GlaxoSmithKline, AstraZeneca, and Sunovian; personal fees for DSMB from Spiration and Shire/Baxalta; CME personal fees from WebMD, National Jewish Health, American College of Chest Physicians, Projects in Knowledge, Hybrid Communications, SPIRE Learning, Ultimate Medical Academy, Catamount Medical, Eastern Pulmonary Society, Catamount Medical Communications Medscape, Eastern VA Medical Center, Academy Continued Healthcare Learning, and Mt. Sinai Medical Center; royalites from Up-To-Date; medical advisory boards from Novartis, Phillips, Third Pole, Science 24/7, and Vernoa; and grants from Pearl outside the submitted work. Wilson Pace is on the advisory board for Mylan and has stock from Novo Nordisk, Pfizer, Novartis, Johnson and Johnson, Stryker, Amgen, Gilead, and Sanofi. Chris Price is an employee of the company Optimum Patient Care, which is a co-founder of the APEX COPD initiative. Asif Shaikh is an employee of the company Boehringer Ingelheim, which is a co-founder of the APEX COPD initiative. Neil Skolnik is on advisory boards for AstraZeneca, Teva, Lilly, Boehringer Ingelheim, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline and has received payment for lectures/speaking engagements from AstraZeneca and Boehringer Ingelheim and research support from Sanofi, AstraZeneca, Boehringer Ingelheim, and GlaxoSmithKline. David Price has board membership with Amgen, AstraZeneca, Boehringer Ingelheim, Chiesi, Circassia, Mylan, Mundipharma, Novartis, Regeneron Pharmaceuticals, Sanofi Genzyme, Teva Pharmaceuticals, Thermofisher; consultancy agreements with Amgen, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Mylan, Mundipharma, Novartis, Pfizer, Teva Pharmaceuticals, Theravance; grants and unrestricted funding for investigator-initiated studies (conducted through Observational and Pragmatic Research Institute Pte Ltd) from AstraZeneca, Boehringer Ingelheim, Chiesi, Circassia, Mylan, Mundipharma, Novartis, Pfizer, Regeneron Pharmaceuticals, Respiratory Effectiveness Group, Sanofi Genzyme, Teva Pharmaceuticals, Theravance, UK National Health Service; payment for lectures/speaking engagements from AstraZeneca, Boehringer Ingelheim, Chiesi, Cipla, GlaxoSmithKline, Kyorin, Mylan, Mundipharma, Novartis, Regeneron Pharmaceuticals, Sanofi Genzyme, Teva Pharmaceuticals; payment for the development of educational materials from Mundipharma, Novartis; payment for travel/accommodation/meeting expenses from AstraZeneca, Boehringer Ingelheim, Mundipharma, Mylan, Novartis, Thermofisher; funding for patient enrollment or completion of research from Novartis; stock/stock options from AKL Research and Development Ltd which produces phytopharmaceuticals; owns 74% of the social enterprise Optimum Patient Care Ltd (Australia and UK) and 74% of Observational and Pragmatic Research Institute Pte Ltd (Singapore); and is a peer reviewer for grant committees of the Efficacy and Mechanism Evaluation programme, and Health Technology Assessment.