Running Head: Hyperglycemia, COPD and Readmissions
Funding Support: none.
Date of acceptance: October 22, 2020 ǀ Published online: November 19, 2020
Abbreviations: chronic obstructive pulmonary disease, COPD; standard deviation, SD; body mass index, BMI; analysis of variance, ANOVA; odds ratio, OR
Citation: McGraw M, White HD, Boethel C, Zolfaghari K, Hochhalter A, Arroliga A.Corticosteroid dosing and glucose levels in COPD patients are not associated with increased readmissions. Chronic Obstr Pulm Dis. 2021; 8(1): 117-123. doi: http://doi.org/10.15326/jcopdf.2020.0172
Online Supplemental Material: Read Online Supplemental Material (122KB)
Note: These findings were partially presented at the American Thoracic Society International Conference in 2018
Introduction
Acute exacerbations of chronic obstructive pulmonary disease (COPD) lead to increased mortality and morbidity, and frequently require hospitalization.1 The greatest risk factor for future hospitalization for COPD exacerbation is a history of hospitalization for exacerbation,1 so optimization of care during the hospital stay is critical to help reduce the burden of readmissions.
One of the mainstay treatments used in patients admitted to the hospital is systemic corticosteroids, both oral and parenteral. Early investigations into corticosteroid use for COPD exacerbation supported using corticosteroids over placebo to shorten a hospital stay as well as reduce rate of treatment failure (death, readmission, intubation) at 30 and 90 days, though it was noted that the patients who received glucocorticoid therapy were more likely to have hyperglycemia requiring therapy than those who received the placebo.2,3 In recent years, there has been a trend toward reducing the amount of corticosteroids administered to patients, with the finding that lower doses can still adequately treat the patient. The Reduction in the Use of Corticosteroids in Exacerbated COPD (REDUCE) trial found that a 5- day course of oral corticosteroids was noninferior to a 7-14 day course, and that a 5- day course of treatment was associated with overall shorter hospital length of stay.4
Providing the lowest corticosteroid dose can decrease the risk of complications including hyperglycemia, osteoporosis, fractures, obesity, and susceptibility to infections, among others.5 Moreover, it is estimated that 28% of patients admitted for COPD exacerbation suffer from diabetes.6 Although the current literature recommends prednisone 40 mg per day for a 5-day period,4 actual dose given and length of treatment are highly variable.
Given the known risk of hyperglycemia with corticosteroid use, we hypothesized that higher doses of corticosteroids will lead to increased readmissions, due to complications of hyperglycemia. The objective of this study was to assess rates of corticosteroid overuse in hospitalized COPD exacerbation patients admitted to a tertiary care referral center in central Texas, and to determine if corticosteroid exposure and average blood glucose had any effect on hospital readmission.
Methods
Study Design and Data Collection
A retrospective study of patients above 18 years old admitted for COPD exacerbation was conducted at a tertriary care referral center in central Texas between February 2014 and July 2016. A COPD exacerbation was defined in 2 ways based on Center for Medicare and Medicaid Services guidelines (Supplement A in the online supplement). Data collection included demographics, age, sex, body mass index (BMI), daily corticosteroid dosing, average daily blood glucose level, emergency department admission, length of hospitalization, and discharge disposition. The primary outcomes studied were readmission rates at 30 and 31-90 days. Prednisone equivalent dosage was calculated for each different type of corticosteroid administered per individual during hospitalization.7 The average daily glucose was determined as the mean of glucose levels per day during hospitalization and high maximum glucose was recorded as an indicator of whether the patient had any glucose level reading over 180 mg/dL during hospitalization. Discharge disposition was grouped into 4 categories: home, home with home health, post-hospital facility, and other (court/law enforcement, home with hospice, inpatient hospice, and left against medical advice).
Statistical Analysis
Sample characteristics are described using descriptive statistics. Frequencies and percentages are used to describe categorical variables. Means and standard deviations (or medians and ranges where appropriate) are used to describe continuous variables. A chi-square test (or Fisher’s exact test where appropriate) and student’s t-test (or their non-parametric equivalents) were used to assess outcomes among different patient groups. An analysis of variance (ANOVA) model (or Kruskal-Wallis test when appropriate) was used to test for differences in continuous variables between the 3+ groups. The post-hoc Tukey’s test was used for pairwise comparisons when the ANOVA test showed a statistically significant result. Multivariable logistic regression assessed the association between readmission rate, glucose, and prednisone equivalent dosage adjusting for age, sex, BMI, history of diabetes, emergency department admission, and discharge disposition. The regression model variables were selected to control for potential confounding factors that were of interest. The average daily glucose and high maximum glucose were used in separate regression models due to the high correlation. A subgroup analysis of this study was conducted which considered only patients carrying a diagnosis of diabetes in the year prior to their first COPD admission.
All analyses were performed using SAS, Version 9.4 (SAS Institute, Cary, North Carolina). Research was approved by the local institutional review board (protocol number 160323), and the need for consent was waived, due to the retrospective nature of the study.
Results
Patient Characteristics
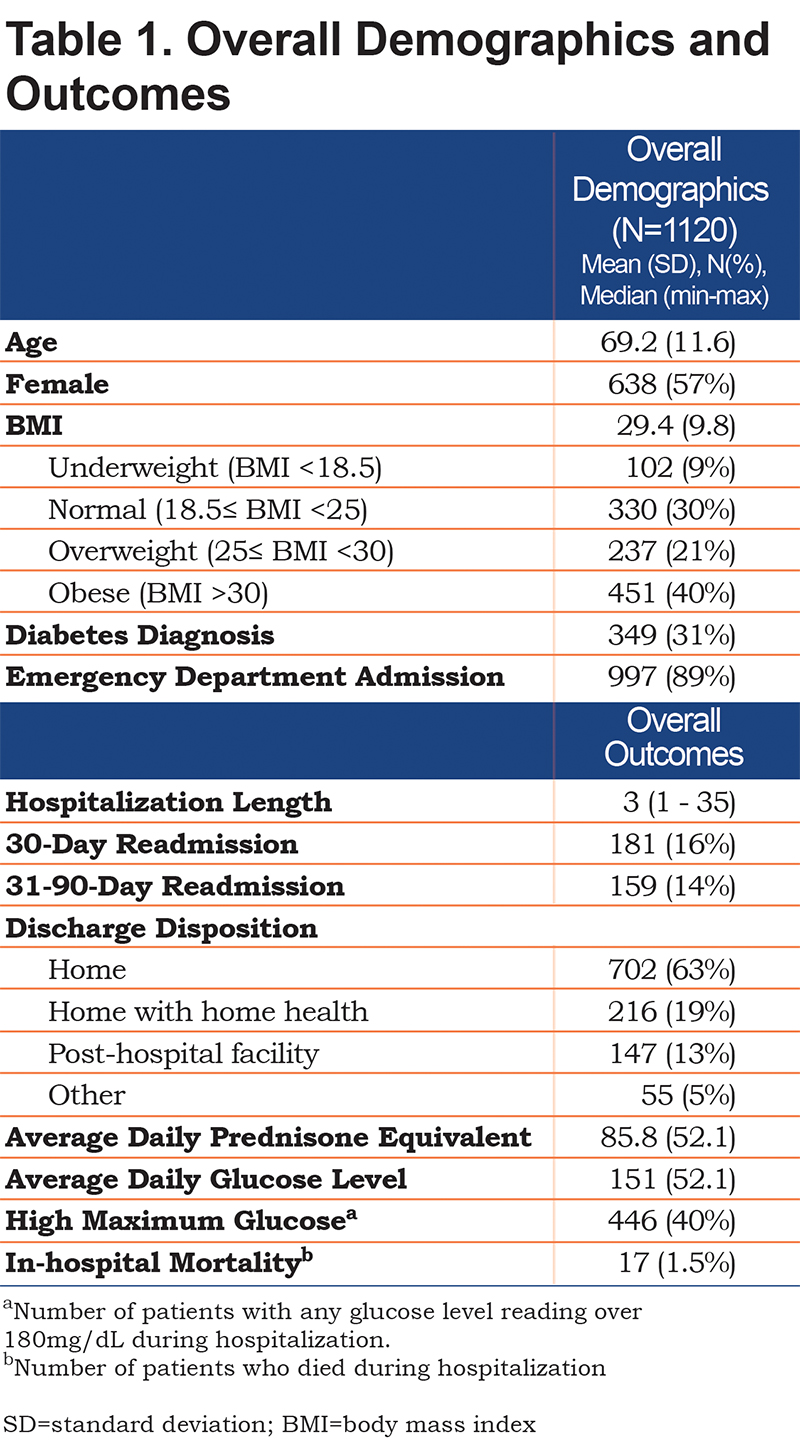
A total of 1120 inidividuals were included in this study. The mean age was 69.2 years old (SD 11.6), average BMI was 29.4 (SD 9.8), and the majority were female (57%). The median for hospital length of stay was 3 days (range 1-35) and 89% were admitted through the emergency department. Of the total, 349 (31%) had diabetes prior to admission. The average prednisone equivalent dose per day was 86.0 mg (SD 52.1). Guideline-based dosing of prednisone equivalent 40 mg per day was seen in 201 patients (18%).
The average daily glucose level was 151.0 mg/dL (SD 52.1). Only 38% had an average glucose of less than 120mg/dL during their stay. Another 42% had averages between 120mg/dL and 180mg/dL, and the rest had an average above 180mg/dL. There were 446 patients (40%) with high maximum glucose (>180mg/dL on any reading) (Table 1). Patients with diabetes prior to admission were the majority of the high maximum glucose group at 76% (Table 2). Patients with an average daily glucose<120mg/dL received less prednisone equivalent compared to those >120mg/dL (77.4mg versus 90.9mg, p<0.001).
Outcomes
The 30-day and 31-90-day readmission rates were 16% and 14% respectively and in-hospital mortality rate was 1.5%. In decreasing order, patients were discharged to home (63%), home with home health (19%), post-hospital facility (13%), and other locations (5%) (Table 1).
Prednisone equivalent dosage was similar for those discharged to a post-hospital facility (86.5 mg daily) and those discharged to home with or without home health (84.0 mg daily). Patients discharged to home with home health or a post-hospital facility were more likely to be readmitted within 30 days in comparison to those discharged home without home health (unadjusted odds ratio [OR]=1.60, 95% CI [1.15, 2.23], p<0.01).
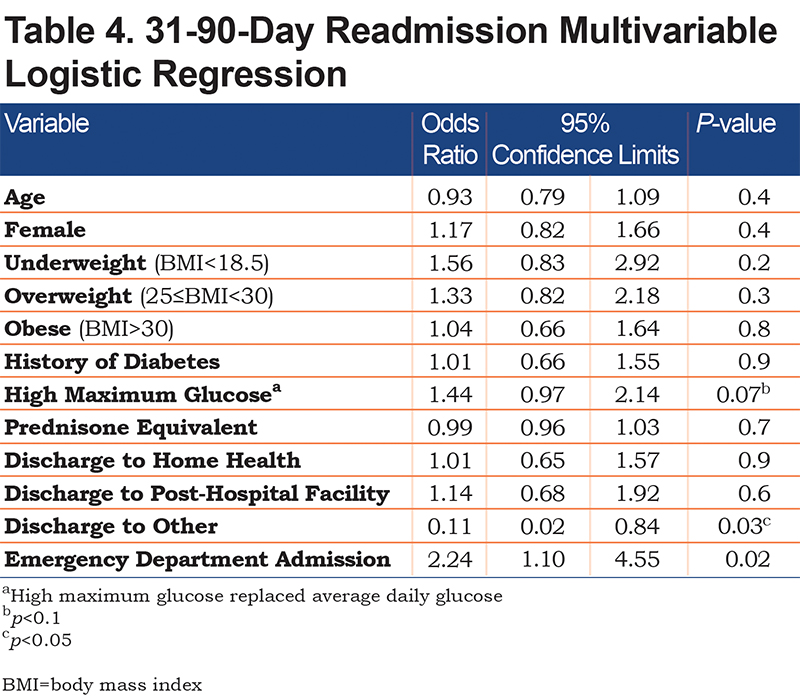
Average daily glucose and guideline-based dosing of prednisone equivalent 40 mg per day were not found to be associated with 30-day or 31-90-day readmission rates. However, 31-90- day readmission had a borderline significant association with high maximum glucose (unadjusted OR=1.37, 95% CI [0.98, 1.93], p=0.07).
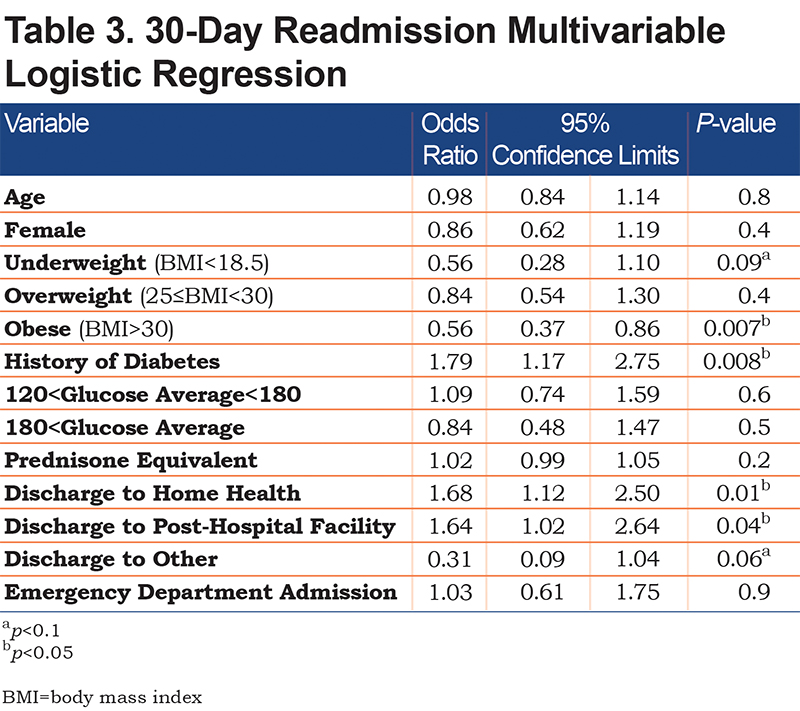
In the multivariable logistic regression (Table 3), the odds of a 30-day readmission for the obese group (BMI>30) was lower than the normal group (18.5≤BMI<25) (adjusted OR=0.56, 95% CI [0.37, 0.86], p<0.05) and the odds of readmission for patients discharged to home with home health (adjusted OR=1.68, 95% CI [.12, 2.50], p<0.05) or post-hospital facility (adjusted OR=1.64, 95% CI [1.02, 2.64], p<0.05) was higher than those discharged to home without help. The regression models did not show any significant associations between 30-day readmission and average daily glucose, high maximum glucose, or prednisone equivalents per day after controlling for potential confounding factors. In accordance with the unadjusted model, the risk of readmission at 31-90 days had a borderline significant association with high maximum glucose (adjusted OR=1.44, 95% CI [0.97, 2.14], p=0.07) as demonstrated in Table 4.
Subanalysis
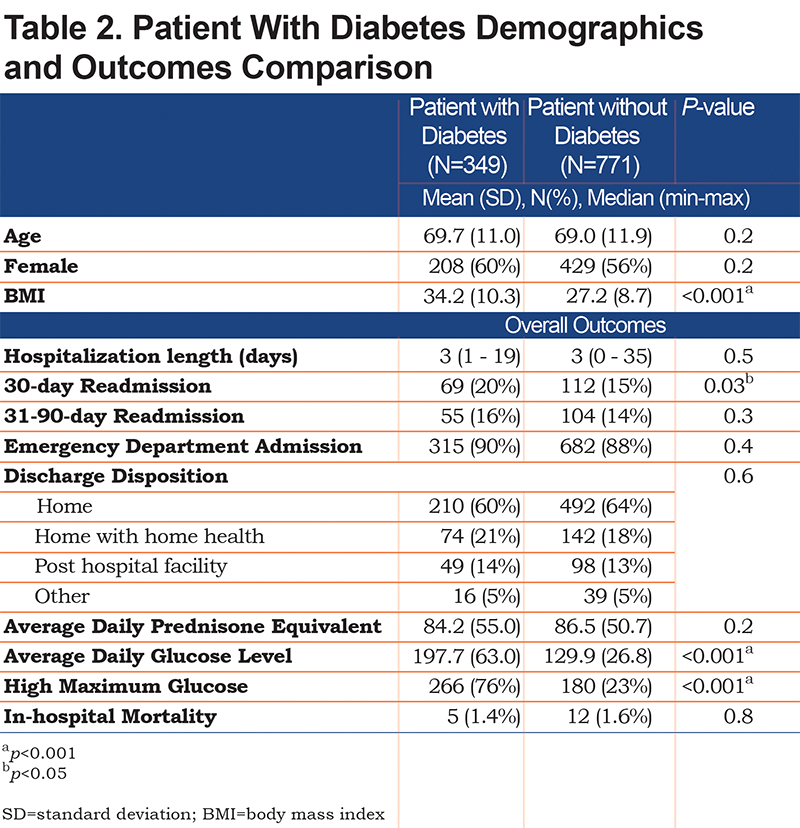
A subanalysis was conducted on 349 individuals with a diagnosis of diabetes. This subgroup of patients had a higher BMI (34.2 versus. 27.2, p<0.001) and average daily glucose level (197.7 versus 129.9, p<0.001) in comparison to the non-diabetic group. Also, high maximum glucose was prevailing (76%) in this subgroup of patients. The 30-day readmission rate was higher among patients with diabetes in comparison to others (20% versus 15%, p=0.02). The hospital length of stay, emergency department admission rate, average prednisone equivalent, 31-90-day readmission rate, and discharge disposition were similar to the non-diabetic group (Table 2). Both unadjusted and adjusted models did not show any association between 30-day or 31-90-day readmission rates and glucose levels or prednisone equivalent doses in this specific group of individuals.
Discussion
We found that guideline discordant dosing of corticosteroids in the treatment of COPD exacerbation did not impact 30-day or 31-90-day readmission rates after discharge from the hospital. Additionally, average glucose level during the initial hospital stay did not impact 30- day or 31-90-day readmission rates after discharge. These findings suggest that strict adherence to guideline-based corticosteroid dosing during hospitalization for COPD exacerbation may not impact the patient’s risk of readmission in the immediate or 3-month periods and this heterogeneity in steroid prescribing practice is consistent with other literature. We did find that patients discharged home with home health or a post hospital facility were more likely to be readmitted within 30 days compared to those discharged home without home health (21% versus 14%, p<0.01), which may be a major factor to total cost of care.
Our findings do not give license to use of higher doses of steroids, as this study was not designed to evaluate the effectiveness of current corticosteroid recommendations. Risk of hyperglycemia from higher doses of corticosteroids should continue to be a consideration for clinicians. Umpierrez et al showed that patients with new onset hyperglycemia on admission had increased mortality and length of stay compared to those with normoglycemia on admission,8 though glucose levels at readmission was not studied in our data. Additionally, new onset diabetes has been observed to occur in up to 46% of patients after initiation of corticosteroid therapy.9 Furthermore, Blackburn et al found that the risk of diabetes was even higher in the elderly population when initiated on a corticosteroid.10
We did perform a subgroup analysis consisting of those carrying a diagnosis of diabetes prior to their initial admission, but the analysis did not show any significant trend regarding readmission rates. Breakey et al showed similar outcomes when they found that glucocorticoid use increased the risk of hyperglycemia in admitted patients with respiratory diseases except in those previously carrying a diagnosis of diabetes.11 This is somewhat counterintuitive, as one would expect those who already have insulin resistance and depletion would be more likely to develop hyperglycemia from a trigger. It is possible that glucose levels during admission are more aggressively managed in those who have been identified to clinicians as diabetics.
Our current study has design lmitations and the findings of this study should be interpreted within the appropriate context. The study was retrospective which should be considered before generalization. Another limitation is the lack of protocolized management of COPD exacerbation and variability in steroid administration, although order sets are available to encourage standardization of care. Finally, data regarding past history related to COPD and the severity were not available secondary to study design.
Additional research is needed to understand if, and how, in-hospital treatment of COPD exacerbation impacts functional status, and health, after discharge.
Conclusion
We conclude that guideline discordant dosing of corticosteroids and the average daily glucose level in the treatment of COPD exacerbation did not impact 30-day or 31-90-day readmission rates after discharge from the hospital.
Acknowledgements
Author contributions: Drs. McGraw, White, Hochhalter, Arroliga and Boethel contributed to the study design, manuscript writing, and editing. Mr. Zolfaghari contributed to the study design, manuscript writing and editing and data analysis.
Declaration of Interest
The authors have no conflicts of interest to disclose.