Running Head: Tobacco Use and COPD Quality of Life
Funding Support: This manuscript is supported with federal funds from the Center for Tobacco Products (CTP), the Food and Drug Administration (FDA), and the Department of Health and Human Services, through an interagency agreement between the FDA CTP and the National Institute on Drug Abuse (NIDA) at the National Institutes of Health (NIH). The PATH Study is supported with federal funds from NIH NIDA and FDA CTP, under contract to Westat (contract nos. HHSN27101100027C and HHSN27120160001C). Carlos Blanco, Wilson Compton, and Heather L. Kimmel were substantially involved in the scientific management of and providing scientific expertise for contract nos. HHSN27101100027C and HHSN27120160001C. Disclaimer: The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the U.S. Department of Health and Human Services or any of its affiliated institutions or agencies.
Date of Acceptance: December 6, 2023 │ Published Online Date: December 19, 2023
Abbreviations: 6MWD=6-minute walk distance; BMI=body mass index; CI=confidence interval; CNP=civilian noninstitutionalized population; COPD=chronic obstructive pulmonary disease; ENDS=electronic nicotine delivery systems; GEE=generalized estimating equations; HRQoL=health-related quality of life; PD30=past 30-days; PATH=Population Assessment of Tobacco and Health; PROMIS=Patient-Reported Outcomes Measurement Information System; SE=standard error
Citation: Paulin LM, Halenar MJ, Edwards KC, et al. Relationship between tobacco product use and health-related quality of life among individuals with COPD in waves 1–5 (2013–2019) of the Population Assessment of Tobacco and Health Study. Chronic Obstr Pulm Dis. 2024; 11(1): 68-82. doi: http://doi.org/10.15326/jcopdf.2023.0422
Online Supplemental Material: Read Online Supplemental Material (142KB)
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic, progressive respiratory disease most commonly caused by cigarette smoking and is a leading cause of morbidity in the United States.1,2 Importantly, once COPD develops, continued exposure to cigarette smoke is associated with worse outcomes, including a greater rate of lung function decline, increased exacerbations, and mortality.3,4 However, less is known about the impact of noncigarette tobacco product use, including e-cigarettes, on respiratory outcomes in those with established disease. Given that individuals with COPD may be less likely to succeed in smoking cessation than those without COPD,5 it is important to quantify the relationship between ongoing noncigarette tobacco product use on COPD morbidity compared to cigarette use.
While mortality, hospitalizations, and disease severity are important measures of COPD burden, assessment of the quality of life of individuals with COPD is an accepted major outcome in observational trials of COPD as it readily captures the impact of disease on daily life and is a reliable predictor of disease progression and mortality.6,7 8,9 The Patient-Reported Outcomes Measurement Information System (PROMIS) is one such tool used to capture health-related quality of life (HRQoL).10 Prior research using PROMIS has shown important associations with clinical outcomes, including lung function and 6-minute walk distance (6MWD) in individuals with COPD,11,12 and HRQoL has been shown to vary with tobacco use in the general population.13,14 However, the relative impact of different tobacco products on HRQoL among individuals with COPD is largely unknown. Identifying the impact of noncigarette tobacco exposures on HRQoL may provide insight into targeted interventions and education surrounding tobacco product use in those with COPD.
In this study, we aim to identify the impact of various tobacco products on HRQoL using data from Waves 1 to 5 of the Population Assessment of Tobacco and Health (PATH) Study, an ongoing, nationally representative, longitudinal cohort study that collects self-reported information on tobacco use patterns, health behaviors, and medical history.
Methods
Study Design, Setting, and Participants
The PATH Study is a nationally representative longitudinal cohort study that collected data annually for the first 4 waves (Wave 1 to Wave 4) of data collection (Wave 1; data were collected from September 12, 2013, through December 14, 2014; Wave 4 data were collected from December 1, 2016, through January 3, 2018) and biennially between Wave 4 and Wave 5 (Wave 5 data were collected from December 1, 2018, through November 30, 2019). The PATH Study design employed a stratified address-based, area probability sampling design at Wave 1 that oversampled adult tobacco users, young adults (aged 18–24), and African American adults (i.e., this comprises the Wave 1 cohort). A probability replenishment sample was selected from the U.S. civilian noninstitutionalized population (CNP) at the time of Wave 4 (including persons who were not in the CNP at the time of Wave 1). The replenishment sample was combined with the respondents from the Wave 1 cohort to represent the CNP at the time of Wave 4 (i.e., this comprises the Wave 4 cohort) Cross-sectional analyses use the Restricted Use File Wave 5 data from the Wave 4 cohort, and longitudinal analyses use the Restricted Use File data from Waves 1–5 from the Wave 1 cohort.15 The respondents were interviewed in person and questionnaires were administered via audio computer-assisted self-interviews by trained interviewers for the PATH Study. The complete PATH study design and methods have previously been published.16-18 All adult respondents provided informed consent. The study was conducted by Westat and approved by the Westat Institutional Review Board.
Primary Outcomes – PROMIS
PROMIS includes over 300 measures of physical, mental, and social health for use with the general population and with individuals living with chronic conditions. To assess HRQoL among those diagnosed with COPD, we used the PROMIS Global-10 sub-questionnaire, which is used to measure generic health, rather than disease-specific health.19 The PATH Study includes all 10 questions from the Global-10 questionnaire, which allows for the creation of a physical health subscale (scored 4–20), a mental health subscale (scored 4–20), and 2 individual items (each scored 1–5) that assess overall health and overall ability to carry out social activities and roles (social health) (Supplemental Table 1 in the online supplement). Higher scores indicate a higher HRQoL.
COPD Diagnosis
At Wave 1 of the PATH Study, participants were asked: “Has a doctor, nurse, or other health professional EVER told you that you had any of the following lung or respiratory conditions? Choose all that apply: COPD, chronic bronchitis, emphysema, asthma, some other lung or respiratory condition, none of the above, don’t know, refused.” At subsequent waves, participants were asked about respiratory disease diagnoses over the past 12 months. For these analyses, COPD, chronic bronchitis, and emphysema diagnoses were combined to create one COPD measure and are referred to as COPD moving forward, similar to prior COPD prevalence studies using National Health and Nutrition Examination Survey data.20,21 Individuals with a history of “some other lung or respiratory condition” were excluded. Given that COPD diagnoses are rarely made before age 40, and consistent with national COPD cohorts,21-23 we limited our sample to adults aged ≥40 years who have ever had a COPD diagnosis at Wave 5 for the cross-sectional analyses (N=1670). For our longitudinal analyses we limited our sample to adults aged ≥40 years at Wave 1 who have ever had a COPD diagnosis at Wave 1 (N=686).
Tobacco Exposures of Interest
Participants reported lifetime and past 30-day (P30D) use of combusted tobacco products (cigarettes, traditional cigars, cigarillos, filtered cigars, pipes, and hookah) and noncombusted tobacco products (snus pouches, other smokeless tobacco [loose snus, moist snuff, dip, spit, chewing tobacco], and e-cigarettes) at each wave. After Wave 1, additional electronic nicotine delivery systems (ENDS) were also asked about, but hereafter will be referred to as e-cigarettes.
Thirteen mutually exclusive categories defined all past and current tobacco use possibilities:
- never or former experimental use [e.g., lifetime use of <100 cigarettes or never used other products fairly regularly; hereafter referred to as never];
- former established use [e.g., lifetime use of ≥100 cigarettes or ever used other products fairly regularly, and with no use of any tobacco products in the past 30 days; hereafter referred to as former];
- exclusive P30D use of: cigarettes, e-cigarettes, cigars [traditional, cigarillos, filtered cigars], smokeless tobacco products [smokeless tobacco, snus], hookah, pipes;
- P30D use of combinations: cigarettes and e-cigarettes, polycombusted tobacco products, polycombusted and noncombusted tobacco products, and e-cigarettes and smokeless tobacco.
Based on distribution of years since quitting cigarette smoking (Supplemental Figure 1 in the online supplement) and prior literature,24,25 former tobacco users (99% of whom were former cigarette users) were split into 2 groups: (1) those who had quit cigarette smoking <5 years ago (<5-year cigarette cessation; mean [standard error (SE)] years since quitting of 2.1 [0.2]), and (2) those who had quit cigarettes ≥5 years ago (≥5-year cigarette cessation, mean [SE] years since quit of 24.4 [1.1]).
Covariates
Covariates included sociodemographic variables of age, sex, body mass index (BMI), race/ethnicity (categories of non-Hispanic White, non-Hispanic Black, non-Hispanic other race or multiple races, and Hispanic), and education (categories of less than high school or GED, high school graduation, some college or associate degree, bachelor’s degree or more). Other potential smoke exposure-related confounders included pack years of cigarette smoking,26,27 past week second-hand smoke exposure (per 5 hours),28 and cannabis use.29 A disease comorbidity index score that has been used in prior studies to capture important contributors to morbidity in COPD patients (range 0–9) was also included.30 Missing data on age, gender, race, Hispanic ethnicity, and adult education were imputed as described in the PATH Study User Guide.31 All covariates used in cross-sectional analyses were from Wave 5, all covariates used in longitudinal analyses are from the baseline of the wave pair.
Statistical Analysis
For the cross-sectional analyses, we examined the associations between Wave 5 PROMIS scores and covariates using weighted comparisons of means. We estimated the association of PROMIS score with Wave 5 tobacco exposure categories while adjusting for covariates using multivariable linear regression. We employed 2 separate analytical approaches to better understand the relationship between tobacco product use and PROMIS outcomes, each with a different reference category: (1) individuals who quit cigarette smoking ≥5 years ago (≥5-year cigarette cessation), and (2) individuals who exclusively smoked cigarettes in P30D. The cross-sectional analyses were weighted using the Wave 5 single-wave full-sample and replicate weights for the Wave 4 cohort.
Generalized estimating equations (GEE) were used to evaluate the longitudinal association between tobacco use assessed at each baseline wave and PROMIS scores at each follow-up wave over four 2-wave periods (Wave 1 to Wave 2, Wave 2 to Wave 3, Wave 3 to Wave 4, and Wave 4 to Wave 5). GEE allows for the inclusion of associations from all 4 periods in a single analysis while statistically controlling for interdependence among observations contributed by the same individuals.32,33 The GEE analyses were weighted using the Wave 5 longitudinal (all-waves) full-sample and replicate weights for the Wave 1 cohort. Weighted estimates from Waves 1–5 of the PATH Study Wave 1 cohort represent the resident population of the United States aged ≥12 years at the time the specific data were collected who were in the CNP at Wave 1.
SEs were derived using the balanced repeated replication method34 with Fay’s adjustment set to 0.3 to increase estimate stability.35 Variables for pack years of cigarette smoking among ever cigarette users and secondhand smoke exposure were Winsorized at the 95th percentile (e.g., 88.5 pack years at Wave 5) and 98th percentile (100 hours at Wave 5, respectively, to limit the influence of outliers.27 Participants who were missing PROMIS data, tobacco product use data, or covariate data were omitted from analyses. All analyses were conducted using Stata survey data procedures,36 version 17.0.
Results
At Wave 5, 1670 adults aged ≥40 years had self-reported physician-diagnosed COPD (Table 1). A total of 38.3% were male, 75.8% were White, 8.1% were of Hispanic ethnicity, and 29.2% completed less than a high school education. Mean BMI was 29.8, and mean comorbidity index score was 2.9. Most (79.4%) had ever smoked ≥100 cigarettes and among those, pack years averaged 30.9. In P30D measures, 35.1% of participants with COPD used cigarettes, 5.7% smoked cigars, 0.3% used hookah, 7.2% used e-cigarettes, 2.8% used smokeless tobacco products, 11.5% used cannabis, and 53.6% were exposed to secondhand smoke.
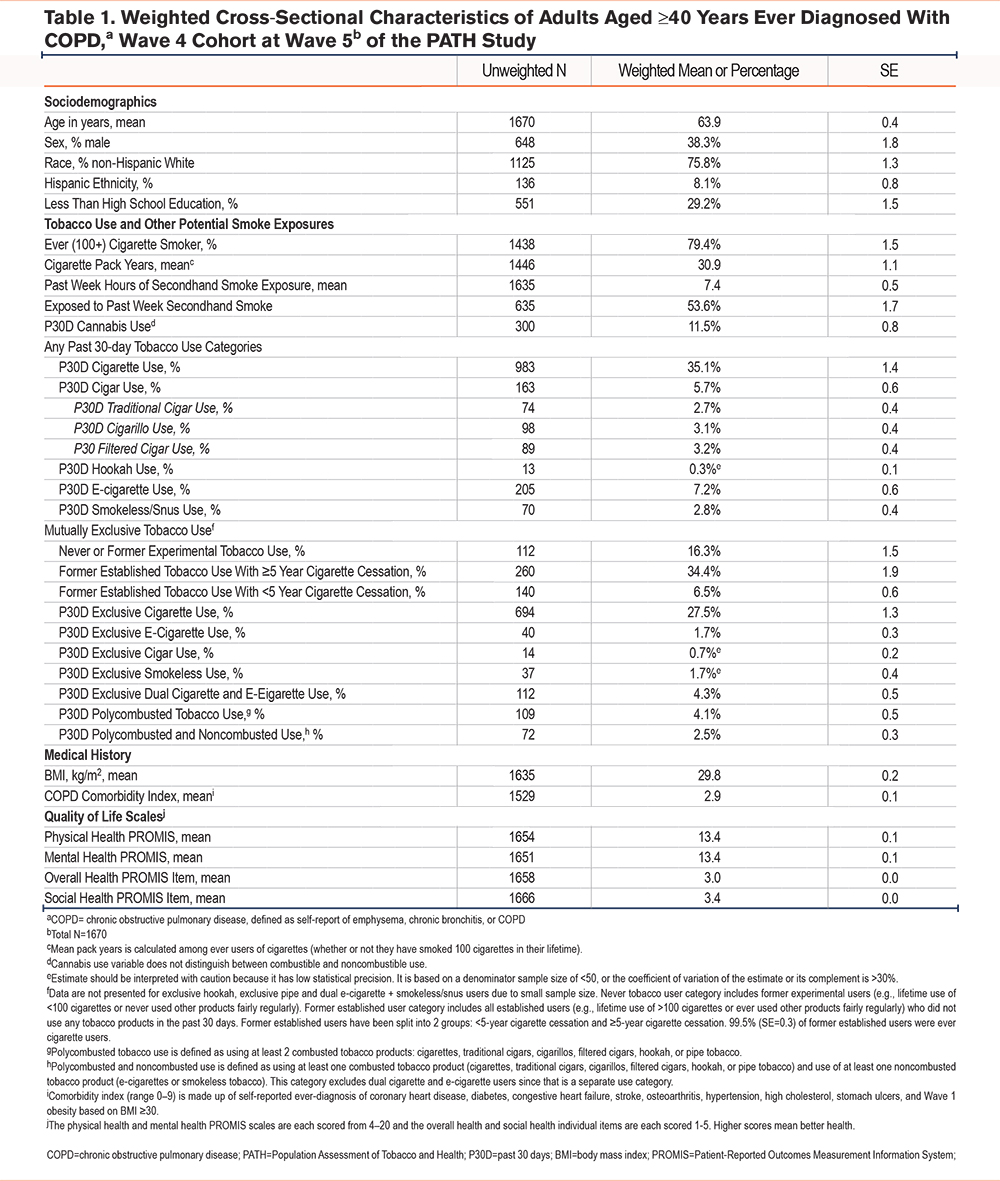
Product Use and Cigarette Smoking History Among Select Tobacco Use Categories
Median years of product use was approximately 5 years in the e-cigarette group in contrast to >30 years for all other product groups (i.e., median of smokeless tobacco presented for exclusive smokeless tobacco group, median cigarette use presented for all polytobacco use groups; Figure 1 Panel A). Median cigarette pack years was high in all groups (>25 years), including those who formerly smoked cigarettes and those who had currently switched to exclusive use of e-cigarettes or smokeless tobacco products (Figure 1 Panel B).
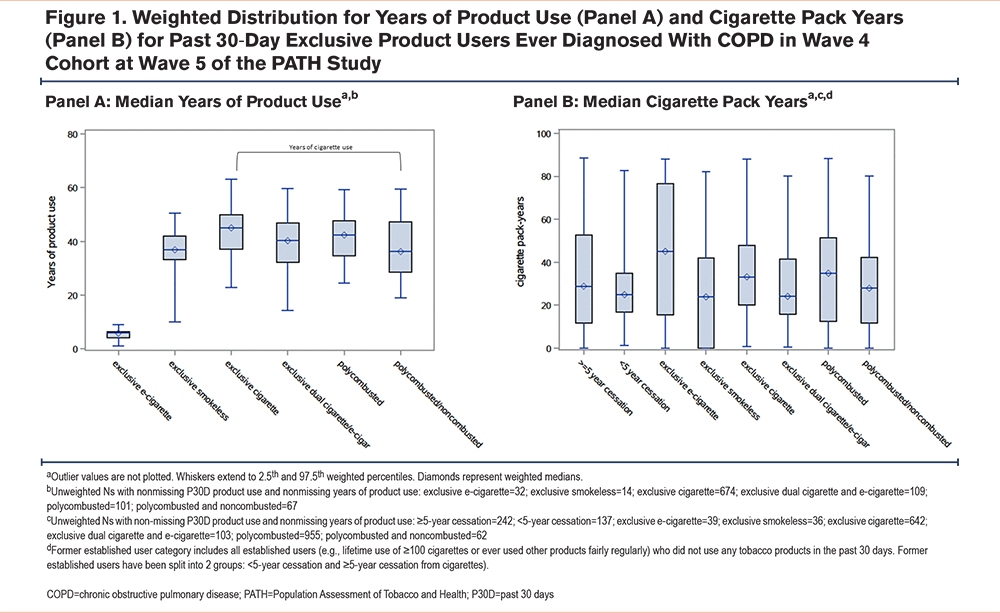
Cross-Sectional Association of Wave 5 Tobacco Product Use With Health-Related Quality of Life at Wave 5
Reference group: P30D Exclusive Cigarette Use at Wave 5: When compared to P30D exclusive cigarette use, never use was associated with significantly better HRQoL as measured by all PROMIS components (physical health beta=1.20, confidence interval [CI] [0.44, 1.97]); mental health beta=1.68, CI [0.81, 2.54]; overall health beta=0.32, CI [0.08, 0.55]; and social health beta=0.32, CI [0.05, 0.58]). Greater than or equal to (≥) 5-year cigarette cessation was also associated with better physical, mental, and overall health QoL, but not social health, compared to current exclusive cigarette use (physical health beta=0.70, CI [0.09, 1.31]; mental health beta=0.98, CI [0.28, 1.67]; overall health beta=0.22, CI [0.02, 0.42]). Compared to P30D exclusive cigarette use, cigarette and e-cigarette dual use was associated with worse social health (beta=-0.41, CI [-0.70, -0.13]), but not associated with worse physical health, mental health, or overall health. Those who used polycombusted and noncombusted tobacco products had better physical health (physical health beta=1.04, CI [0.16, 1.91]) than those who exclusively smoked cigarettes, but this was not the case with mental, overall, or social health. Less than (<) 5-year cigarette cessation and P30D exclusive e-cigarette use, smokeless tobacco use, and polycombusted tobacco product use were not significantly associated with HRQoL (Table 2).
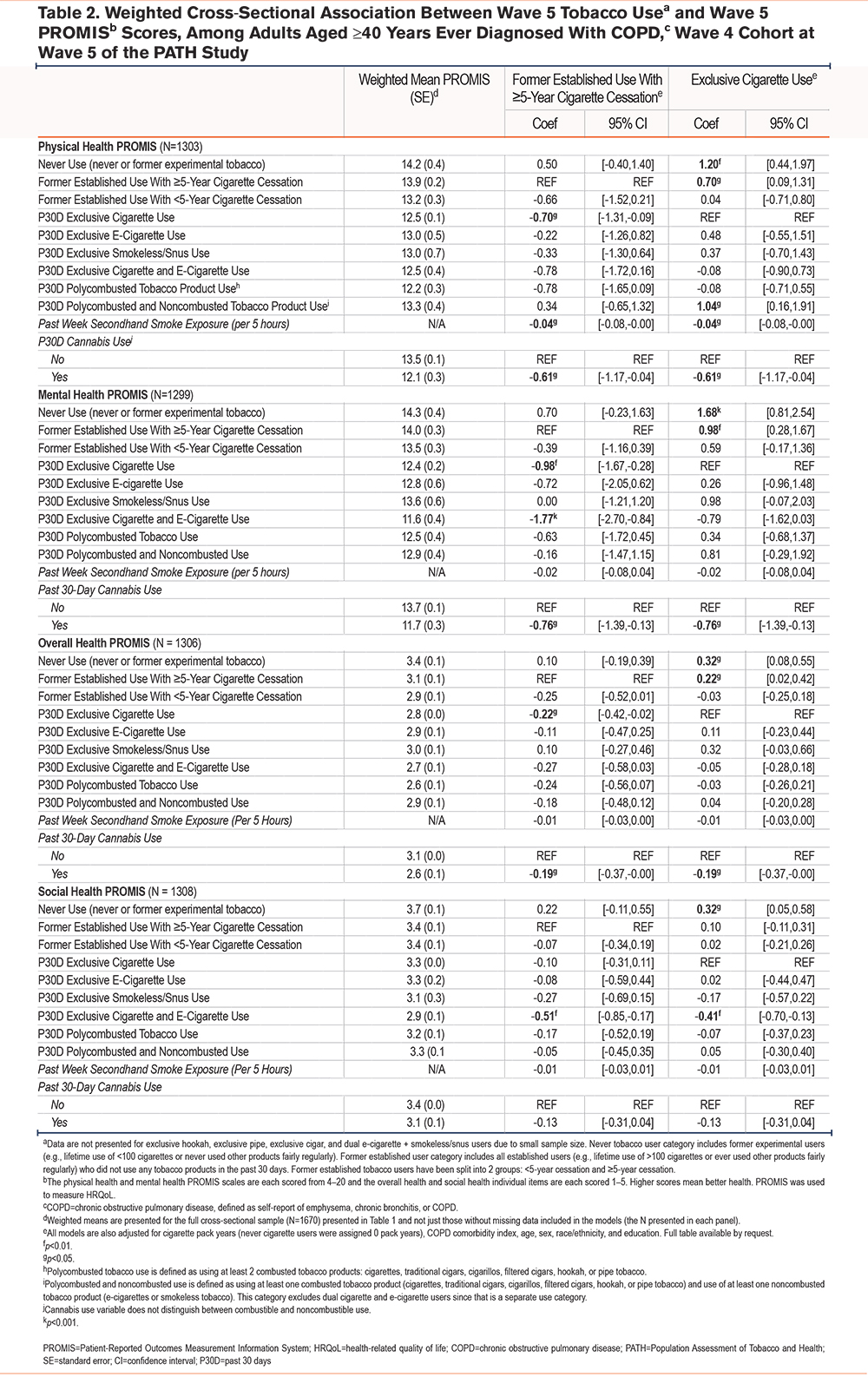
Reference Group: ≥5-year Cigarette Cessation: When compared to ≥5-year cigarette cessation, P30D exclusive cigarette use was negatively associated with physical, mental, and overall health, but not associated with social health (physical health beta=-0.70, CI [-1.31, -0.09]; mental health beta=-0.98, CI [-1.67, -0.28]; overall health beta=-0.22, CI [-0.42, -0.02]). P30D cigarette and e-cigarette dual use was negatively associated with mental health and social health but was not associated with physical health or overall health (mental health beta=-1.77, CI [-2.70, -0.84]; social health beta=-0.51, CI [-0.85, -0.17]). Other tobacco use categories were not associated with QoL when compared to ≥5-year cessation (Table 2).
Other Inhalational Exposures: For individuals exposed to secondhand smoke in the past week, the 10th percentile of hours exposed per week was 1 hour; the 99th percentile was 100 hours/week. In multivariable analysis, secondhand smoke exposure was associated with worse physical health (beta per 5 hours of secondhand smoke exposure=-0.04, CI [-0.08, -0.00]). For those exposed to 100 hours of secondhand smoke per week, this is equivalent to a nearly 1-point decrease in physical health score. There was no association between secondhand smoke exposure and mental, overall, or social health. P30D cannabis use was associated with worse physical, mental, and overall health (physical health beta=-0.61, CI [-1.17,-0.04]; mental health beta= -0.76, CI [-1.39,-0.13]; overall health beta=-0.19, CI [-0.37,-0.00]). There was no association between cannabis use and social health (Table 2).
Longitudinal Association of Tobacco Product Use with Health-Related Quality of Life From Waves 1-5
Reference Group: P30D Exclusive Cigarette Use: When evaluating the longitudinal relationship between tobacco product use and change in PROMIS scores over the four 2-wave pairs, cigarette and e-cigarette dual use at baseline wave was associated with worsening overall health over time (beta=-0.08, CI [-0.16, -0.01]) compared to exclusive cigarette use. Never tobacco users had improved social health (beta=0.18, CI [0.04, 0.33]) compared to exclusivecigarette users. There were no additional longitudinal associations between any other tobacco use category and HRQoL when compared to exclusive cigarette use (Table 3).
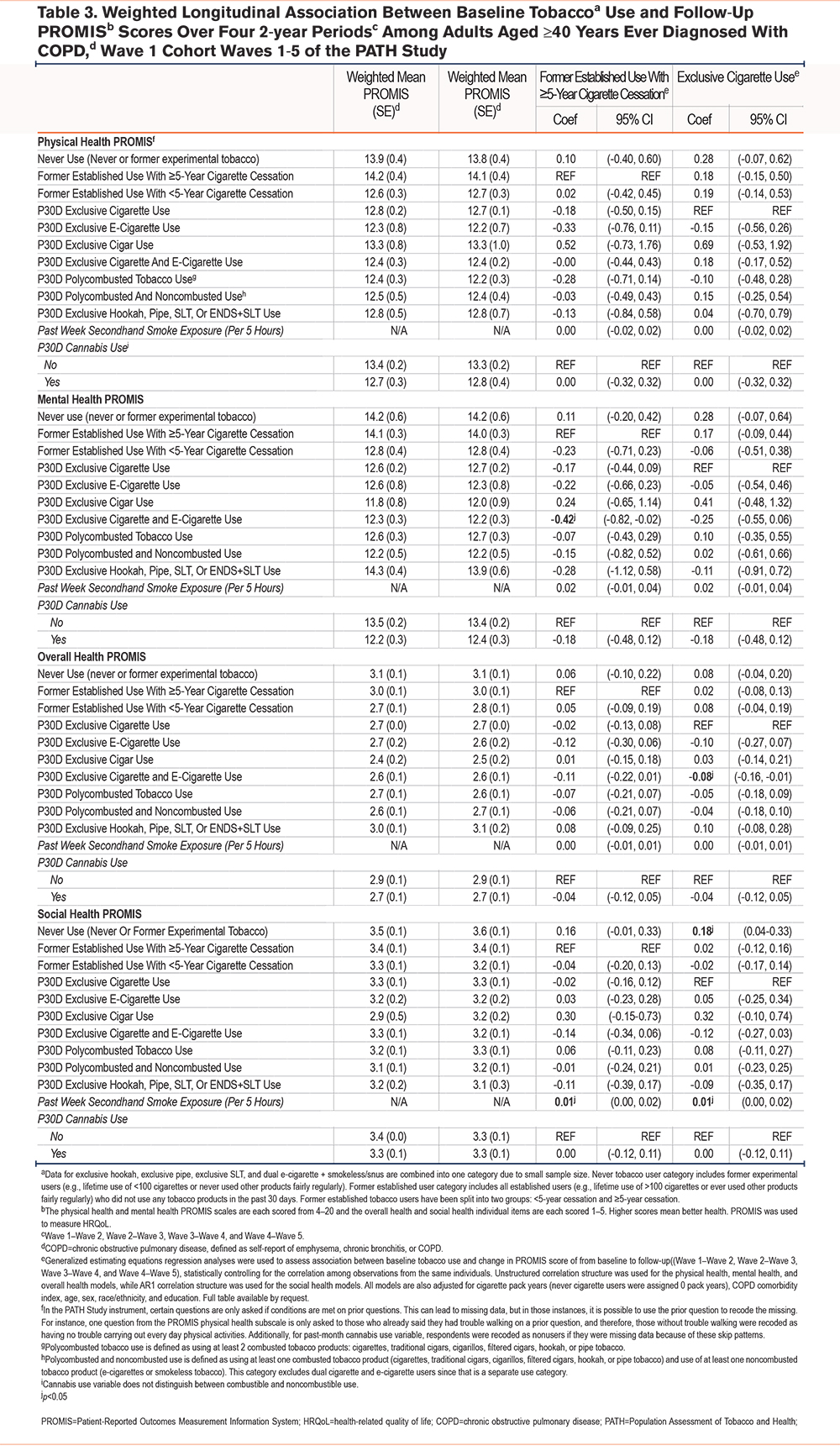
Reference Group: ≥5-year Cigarette Cessation: Compared to ≥5-year cigarette cessation, cigarette and e-cigarette dual use was associated with worsening mental health over the four 2-wave pairs (beta mental health=-0.42, CI [-0.82, -0.02]. There were no other significant associations between other tobacco use categories and HRQoL when compared to ≥5-year cessation (Table 3).
Other Inhalational Exposures: There were no meaningful longitudinal associations between secondhand smoke exposure or P30D cannabis use and change in HRQoL (Table 3).
Discussion
In this nationally representative study of tobacco product use, cross-sectional analyses indicated that current exclusive cigarette use was associated with worse HRQoL compared to ≥5-year cigarette cessation, emphasizing negative physical and mental effects of continued cigarette exposure among people with COPD. Never use and ≥5-year cigarette cessation were the only tobacco use categories that were consistently associated with higher HRQoL compared to exclusive cigarette use, highlighting the importance of complete and sustained smoking cessation for individuals with COPD. We did not find evidence that exclusive e-cigarette use presented a lower risk for HRQoL decrements. When compared to ≥5-year cigarette cessation, current cigarette and e-cigarette dual use was associated with worse mental and social health. When compared to the exclusive cigarette use group, cigarette and e-cigarette dual use was associated with worse social health. Longitudinally, compared to ≥5-year cigarette cessation, dual cigarette and e-cigarette use was associated with worse mental health and overall health when compared to current exclusive cigarette use.
Interestingly, we found that <5-year cigarette cessation was associated with outcomes more similar to those who currently smoked cigarettes than to ≥5-year cigarette cessation, suggesting that duration of abstinence is important to consider when evaluating HRQoL in individuals with COPD. Prior work has focused on the timing of health benefits following smoking cessation. While cardiovascular outcomes such as blood pressure and heart rate normalize rapidly after smoking cessation, quantifiable respiratory improvements (e.g., decreased rate of decline in lung function) may require a sustained period of smoking cessation.37 In a nationally representative study of the French general population, improvements in HRQoL were seen beginning at 2 years following smoking cessation, and continued to increase through 5 years.38 Moreover, in a study of 117 COPD patients, Hassan et al found that, individuals who quit smoking for ≥5 years were less likely to have respiratory symptoms than those who quit smoking more recently (<5 years).25 Findings in our study support clinical recommendations1 for people diagnosed with COPD to quit smoking cigarettes without delay; these individuals should be counseled that the benefits of smoking cessation may occur over several years.39,40
We observed better mental health among individuals who formerly smoked cigarettes compared to those who currently smoked cigarettes. This is of particular interest given that many individuals who currently smoke cigarettes name stress relief as a main driver of continued smoking, and report concern that smoking cessation would negatively impact their stress management abilities.41 Findings in the study are similar to a previous study by Hajek et al, which found a decrease in perceived stress of individuals hospitalized with cardiac disease who were successful in quitting smoking compared to those who continued to smoke.14 Moreover, Cavazos-Rehg et al found that smoking cessation was associated with a decreased risk of persistent or recurrent mood disorder in adults with a prior history of a mood or anxiety disorder,13 similar to findings by Blanco et al which found that smoking remission was not associated with increased risk of psychiatric disorders among adults with existing mood, anxiety, or substance abuse disorder.42 These data support clinical efforts to assure patients who smoke that worsening of mood is not an anticipated outcome following cessation.
We found that exclusive e-cigarette use was not associated with any benefits or harms to HRQoL. Our findings differ from a previous study of 39 patients with COPD by Palosa et al, which found that cigarette smokers who switched to e-cigarette use had improved markers of COPD morbidity, including COPD Assessment Test score and 6MWD after 5 years of follow-up.43 Future work that examines a longer duration of e-cigarette use would be of value to better characterize the impact of these products on COPD outcomes. We found that dual use of e-cigarettes and cigarettes had both cross-sectional and longitudinal associations with worse HRQoL when compared to ≥5-year cigarette cessation and exclusive cigarette use. While desire to quit cigarette smoking is one of common reasons for using e-cigarettes among adults,44 a previous analysis of PATH Study data found that individuals who smoked cigarettes and used e-cigarettes as aids to help quit smoking were less likely to be nicotine abstinent at long-term follow-up compared to a matched sample of those who did not use e-cigarettes as cessation aids.45 The findings that dual users of cigarettes and e-cigarettes have worse HRQoL than ≥5-year cessation may reflect the fact that these persons tend to be heavier, more dependent smokers46 compared to ≥5-year cessation, many of whom may not have been heavy cigarette smokers to begin with. Thus, the cigarette exposure may be the main driver of the observed associations with HRQoL. While we accounted for pack years of cigarette smoking in our models, we recognize the potential for unmeasured confounding that is not captured by pack years, which has been shown to be an imperfect measure of overall cigarette smoking history.47
Of interest is the cross-sectional association between secondhand smoke exposure and worse HRQoL even after adjusting for current and past cigarette smoking. The magnitude of the effect size for individuals with high secondhand smoke exposure (the upper range of the interquartile range is an amount of time that one might expect when living with a parent or spouse who smokes) was among the highest for physical health. These findings were similar to results from a previous study that showed that individuals who never smoked and had higher secondhand smoke exposure had significantly lower scores on physical and social functioning and bodily pain compared to never smokers without secondhand smoke exposure.48 Another study also found that secondhand smoke exposure is associated with worse COPD-related morbidity among COPD patients who continue to smoke.3 These findings highlight the harmful relationship between noxious particles and fume exposure and worse outcomes in COPD.
Findings in the study suggest that P30D cannabis use was negatively associated with physical, mental, and overall health, even after accounting for current tobacco use and pack years of smoking history. Following greater legal availability of cannabis and a general increase in cultural acceptance of use of cannabis and its derivatives,49 cannabis is often seen as a cure for various physical and mental ailments.50 While limited industry-sponsored survey research may show benefit among some respondents,51 our findings provide some information that cannabis use negatively influences HRQoL.52 Tan et al found that concurrent cannabis and cigarette smoking synergistically increased the risk of respiratory symptoms and COPD when compared to those who only smoke cigarettes and nonsmokers.53 While we did not find evidence of a longitudinal relationship between cannabis use and HRQoL, clinicians should consider advising against regular cannabis use among people with COPD given the strong cross-sectional associations.
Our study has several strengths. Analyses were limited to adults aged ≥40 years, a timeline consistent with the development of airflow obstruction following years of exposure to noxious particles and gases.1 The current study benefits from the repeated outcome measures afforded by the PATH Study, allowing for both a cross-sectional and longitudinal examination of the association between multiple tobacco products and HRQoL. This study explicitly accounted for pack years of smoking and additional confounders that are often not captured by other studies. The use of current exclusive cigarette smoking and ≥5-year cessation as reference categories provides 2 scenarios to highlight the unique impact of individual tobacco products on the HRQoL of people with COPD.
Several limitations warrant comment. The PATH Study relies on participant self-report of physician diagnosis and not spirometry to define COPD. While spirometric evidence of airflow obstruction is required to make a COPD diagnosis, spirometry is underutilized in clinical settings,20,54,55 and the lack of spirometry in the PATH Study along with the potential of diagnostic and recall bias associated with self-report may introduce imprecision in the COPD diagnosis. Accounting for self-report of asthma as well as multiple other diseases that may share symptoms with COPD in our adjusted analysis helps to address concerns for misclassification of self-report of physician-diagnosed COPD, a potential limitation of survey research in general. We examined baseline tobacco product use as a predictor of HRQoL, and while prior PATH Study research has shown product-specific persistent use over time in adults, use over time was not incorporated into the analyses.56 We did not consider additional details on frequency of use within the P30D (i.e., regular daily versus intermittent use). In addition, despite controlling for cigarette exposure, residual confounding may have influenced our results. For example, while we emphasize the importance of considering pack years to estimate cigarette smoke exposure, we recognize the potential for unmeasured confounding that is not captured by pack years, which has been shown to be an imperfect measure of overall cigarette smoking history.47 Knowing that COPD symptoms and HRQoL may influence an individual’s ability and choice to inhale tobacco products, the potential remains for reverse causality, especially among those who smoke cigarettes and use e-cigarettes in an attempt to improve health outcomes.45 Although this was a large national survey, the sample size of individuals with COPD was somewhat limited for several of the tobacco product categories (e.g., exclusive hookah, exclusive pipe, and dual e-cigarette + smokeless/snus use). Given the dynamic nature of the e-cigarette industry, including additional details on device type, and e-liquid composition and flavorings in future studies would be of value as nicotine delivery and subsequent health outcomes may vary with device characteristics.57 Future studies should extend this analysis with more waves of data to garner a larger sample.
In summary, our analysis of PATH Study data from Wave 1 to Wave 5 partially supports the known harms of continued cigarette smoking among those with existing COPD and supports the known benefits of smoking cessation. Given that COPD is a progressive and incurable illness, the mainstay of therapy is focused on preventing further lung function decline, avoiding exacerbations, and improving HRQoL.1 These results support continued focus on prevention of tobacco product initiation as well as effective behavioral and pharmacologic tobacco cessation therapies to decrease the negative impact of cigarette smoking on HRQoL among those with COPD. Additional research studies on secondhand smoke exposure and cannabis use could help inform clinical recommendations on avoidance of these additional inhalational exposures. Future studies that examine a longer duration of exclusive e-cigarette use and dual cigarette and e-cigarette use would be informative to further quantify the impact of these products on HRQoL.
Acknowledgements
Author contributions: LMP and JDS were involved with the initial conception of the research questions. All authors were involved with the planning of the data analyses and interpretation of the data analyses. MJH analyzed the data. LMP wrote the manuscript. All authors were involved with reviewing, editing, and final approval of the manuscript. This manuscript was prepared while author LDG was employed at the Food and Drug Administration, Center for Tobacco Products.
Data sharing statement: Details on accessing the PATH Study data are described in the PATH Study Restricted Use Files website located at https://doi.org/10.3886/ICPSR36231.v29. Access to this data is restricted. Users interested in obtaining this data must complete a Restricted Data Use Agreement.
Declaration of Interests
K. Michael Cummings provides expert testimony on the health effects of smoking and tobacco industry tactics in lawsuits filed against the tobacco industry. He has also received payment as a consultant to Pfizer, Inc., for services on an external advisory panel to assess ways to improve smoking cessation delivery in health care settings. Martin C. Mahoney has provided expert testimony on the health effects of smoking in lawsuits filed against the tobacco industry. He has also received research support from Pfizer, Inc., for an on-going clinical trial of smoking cessation, and has previously served on external advisory panels sponsored by Pfizer to promote smoking cessation in clinical settings. Raymond Niaura receives funding from the Food and Drug Administration Center for Tobacco Products via contractual mechanisms with Westat and the National Institutes of Health. Within the past 3 years, he has served as a paid consultant to the Government of Canada via a contract with Industrial Economics Inc., and has received an honorarium for a virtual meeting from Pfizer Inc. Dr. Niaura was an unpaid grant reviewer for the Foundation for a Smoke Free World. Between mid-2015 and 2020, Dr. Niaura frequently communicated with Juul Labs personnel, for which there was no compensation, and received hospitality in the form of meals at some meetings. All other authors have nothing to declare.