Running Head: Effect of Chronic Bronchitis on COPD Burden
Funding Support: The study was funded by Novartis Pharma AG.
Date of Acceptance: February 8, 2025 │ Publication Online Date: February 28, 2025
Abbreviations: % pred=percentage predicted; BMI=body mass index; CB=chronic bronchitis; CFTR=cystic fibrosis transmembrane conductance regulator; COPD=chronic obstructive pulmonary disease; EHR=electronic health record; FEV1=forced expiratory volume in 1 second; FVC=forced vital capacity; HCRU=health care resource utilization; HIPAA=Health Insurance Portability and Accountability Act; ICD-10-CM=International Classification of Diseases, Tenth Revision, Clinical Modification; NHLBI=National Heart, Lung, and Blood Institute; SPIROMICS=SubPopulations and InteRmediate Outcome Measures in COPD
Citation: Krishnan JK, Criner GJ, Lashari BH, et al. Disease onset and burden in patients with chronic bronchitis and COPD: a real-world evidence study. Chronic Obstr Pulm Dis. 2025; 12(2): 127-136. doi: http://doi.org/10.15326/jcopdf.2024.0565
Online Supplemental Material: Read Online Supplemental Material (807KB)
Introduction
Chronic bronchitis (CB) is classically defined as having cough and sputum production for at least 3 months per year for 2 consecutive years and is commonly associated with a history of smoking.1,2 CB occurs in up to three-quarters of patients with chronic obstructive pulmonary disease (COPD)3 and also in individuals with normal lung function (nonobstructive CB, hereafter termed CB), with prevalence estimates varying widely among both categories.4 CB has a higher prevalence generally among those with smoking exposure.3 Due to variability in CB definitions, estimation of the true burden of CB remains challenging. Compared to patients who do not have CB, those with CB have a higher risk of developing incident COPD, respiratory mortality, and all-cause mortality.4 The COPD Genetic Epidemiology observational study revealed that among patients with COPD, those with CB have worse respiratory symptoms, worse quality of life, and higher exacerbation risk than those without CB.5
Despite the existing literature describing the impact of CB on prognosis, the prevalence of patients with CB or COPD alone, compared to those who have both conditions and their respective health care resource utilization (HCRU) has not been estimated before in a real-world population. In this analysis, our primary aim was to identify patients with CB, COPD, and both CB and COPD through the application of the classical definition of CB to real-world clinical notes in electronic health records (EHRs) and to compare the characteristics of these populations as well as their HCRU. We also examined medication use patterns in these populations and the distribution of the timing of CB versus COPD onset for patients who eventually have both. Additionally, patients who develop COPD are at risk for several other comorbidities due to complex genetic and environmental exposure interactions. These comorbidities have been shown to cluster, potentially due to common underlying mechanisms.6 One potential underlying mechanism that may contribute to comorbidity clustering is smoking-related acquired cystic fibrosis transmembrane conductance regulator (CFTR).7,8 Acquired systemic CFTR dysfunction is associated with CB, gastroesophageal reflux disease, and osteoporosis. 6,9,10 Therefore, the final aim of this analysis was to determine if comorbidities that are associated with CFTR dysfunction are more prevalent in patients with both COPD and CB using real-world data.
Methods
Study Design
This was a retrospective cohort study based on the secondary use of data from the Optum® de-identified EHR data set. Optum® EHR is a longitudinal EHR repository derived from dozens of health care provider organizations in the United States. Administrative medical data is obtained from both inpatient and ambulatory EHRs, practice management systems, and other internal systems and is processed, normalized, and standardized across the continuum of care from both acute inpatient stays and outpatient visits. The Optum data has been de-identified pursuant to the Health Insurance Portability and Accountability Act (HIPAA) and Optum, using the expert determination method, and has documented the methods and results of the analysis that justify such determination as set forth in 45 C.F.R. 164.514 and the “Guidance Regarding Methods for De-identification of Protected Health Information in Accordance with the HIPAA Privacy Rule.”11,12 As this study is a secondary analysis of fully anonymized data, an institutional review board or additional informed consent was not required.13
The study period was from January 1, 2007, to September 30, 2020. Patients were studied from 2 years prior to first diagnosis with a post-diagnosis follow-up period for each patient of 5 years.
Electronic Health Record Review Process
Patients’ EHRs were electronically reviewed using an algorithm that “read” each record, searching for particular terms used to define CB. The rules for searching within the EHRs are outlined in more detail in the online data supplement. Manual chart reviews were not performed.
Definitions of Chronic Bronchitis and COPD
Cohorts were defined based on the contents of Optum® EHR using 2 sources: clinical notes for CB, and the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes for COPD. Both sources were then combined to derive a definition of the COPD and CB (COPD-CB) cohort (Figure 1).
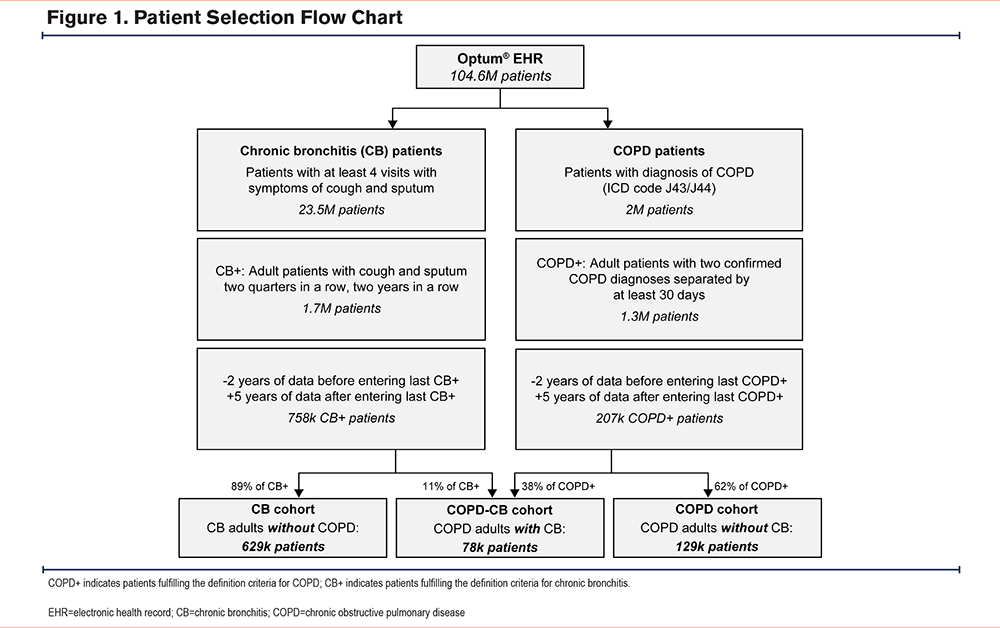
Definition of Patients With Chronic Bronchitis Cohort Using Optum® Electronic Health Record Clinical Notes (CB+)
CB is defined clinically as a cough with sputum production for at least 3 months a year during a period of 2 consecutive years.1,2 To apply this definition, records in Optum® EHR were scanned for documentation of both sputum and cough in the medical encounter notes, within 2 consecutive 3-month periods, 2 years in a row. For these analyses, "symptoms for 2 consecutive 3-month periods" was interpreted as the equivalent of symptoms reported in the clinical notes during 2 quarters in a row, i.e., at least 90 days apart and no more than 180 days apart. These 3-month periods had to be identified in each of 2 consecutive years to meet the definition of CB. See the online data supplement for further details on symptom scanning in clinical notes.
Definition of Patients With COPD Cohort Using International Classification of Diseases, Tenth Revision, Clinical Modification Codes (COPD+)
The ICD-10-CM codes J43 (emphysema) and J44 (other chronic obstructive pulmonary disease) were used to identify patients with COPD. Patients had to have at least 2 diagnoses of COPD with these codes in their EHRs, at least 30 days apart. Patients with ICD-10-CM codes J41 (simple and mucopurulent CB) and J42 (unspecified CB), which are frequently included for COPD, in their EHR were not used to identify patients in this cohort as these codes are used for CB.
Definition of Chronic Bronchitis, COPD, and COPD-Chronic Bronchitis Cohorts
The CB cohort was defined as patients who had CB (as per the above definition) only without COPD. Index day for this cohort was defined as the date of their first record of “cough and sputum” symptoms. Patients were included in the cohort if they had 2 years of Optum® EHR data prior to the index day and 5 years of follow-up Optum® EHR data after the index day, all within the study period defined above. The COPD cohort was defined as patients who had COPD only without CB. Index day for this cohort was defined as the date of their first record of COPD diagnosis. Patients were included in the cohort if they had 2 years of Optum® EHR data prior to index day and 5 years of follow-up Optum® EHR data after the index day, all included within the study period. The COPD-CB cohort was identified as patients with both CB and COPD as defined above at any time during the study period. For the COPD-CB cohort, the index day was the later of the index days for CB and COPD, i.e., the first time a patient qualified for inclusion in both cohorts. Note that due to the large sample size, all results with numerical differences between different cohorts had p-values<0.05.
Definition of Smoking Status
Clinical notes on smoking status recorded in Optum® EHR were reviewed to determine smoking status as outlined in the online data supplement.
Spirometry
Forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) percentage predicted were computed from the FEV1 and FVC measurements recorded in the Optum® EHR, and where available, in the follow-up window as per previously published calculations.14 Averages over the 5-year period were calculated for patients who had more than one measurement in their EHR.
Time to Diagnosis Within the COPD-Chronic Bronchitis Cohort
For patients in the COPD-CB cohort, the time between their first CB diagnosis and their first diagnosis with COPD was assessed. Patients diagnosed with COPD first followed by CB had a negative value.
Health Care Resource Utilization
Data were used to assess the clinical characteristics of the cohorts and their HCRU. For the purpose of the study, HCRU encompasses the following data: the number of medical encounters, the number of pulmonologist visits, the number of imaging referrals, the number of hospitalizations, and the number of days in the hospital. Medical encounters are defined by the following activities recorded in the EHRs: visits to the hospital and specialists, and in-person, written, or telephone contact with a doctor. The mean number of medical encounters in each cohort was calculated for each year. All medical encounters are counted individually regardless of whether they relate to the same issue on the same day. Encounters are not restricted to those related to COPD or CB only.
For each patient, lists of admission dates of distinct hospital visits in the follow-up period were extracted and grouped by the year of the follow-up window. The frequency for each patient was calculated by dividing the total number of hospital visits in the follow-up window by the number of years of the follow-up window. The number of days spent in the hospital was computed using the visit start and end dates from the EHRs. Specialist visits and oxygen therapy were computed using the definitions in the online data supplement.
Medication Use
The number of drug prescriptions for the following medications in the follow-up period was extracted from the EHR: mucolytics and phosphodiesterase type 4 inhibitors, macrolides, long-acting muscarinic antagonists, long-acting beta2-agonists, and inhaled corticosteroids. See the online data supplement for further details.
Analysis of Common COPD Comorbidities
The prevalence of gastroesophageal reflux disease, diabetes, osteoporosis, bronchiectasis, lung cancer, and chronic rhinosinusitis, which are potentially associated with CFTR dysfunction,10,15-17 was evaluated in all 3 cohorts. These comorbidities were identified using ICD-10-CM codes, which is further described in the online data supplement.
Results
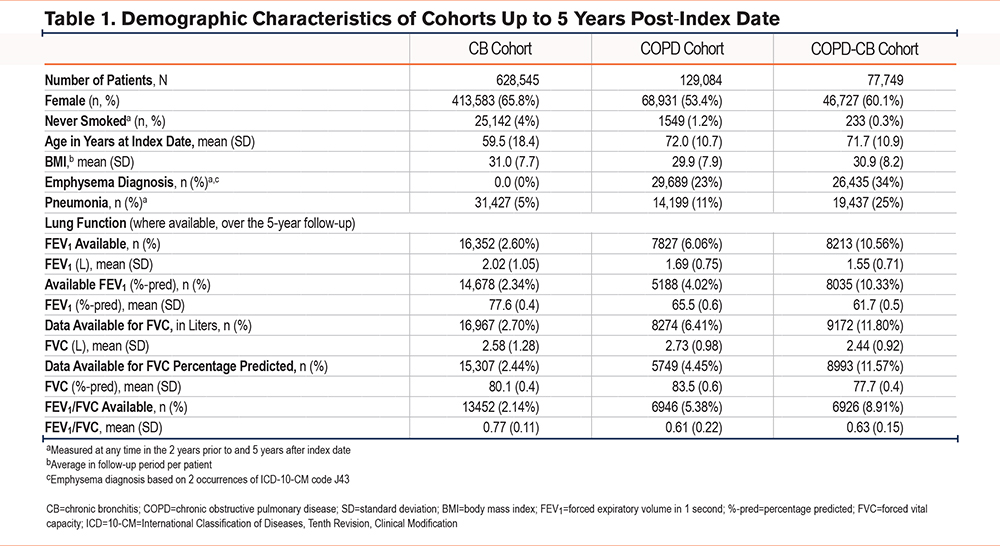
The total population of the Optum® EHR for the study period was 104,633,876 individual patients. The analysis revealed 628,545 patients in the CB cohort, 129,084 in the COPD cohort, and 77,749 in the COPD-CB cohort (Figure 1). Of the 3 cohorts, the COPD-CB cohort had the lowest proportion of patients who had never smoked, as expected since both COPD and CB are associated with smoking (Table 1). Where spirometry data were available, the CB cohort patients had a higher mean FEV1 and mean FEV1/FVC ratio compared with the COPD and COPD-CB cohorts, with the latter 2 having broadly similar spirometry results to each other (Table 1). A greater proportion of patients with CB only or with both CB and COPD were women, whereas the numbers of men and women were almost balanced across patients with COPD only (Table 1).
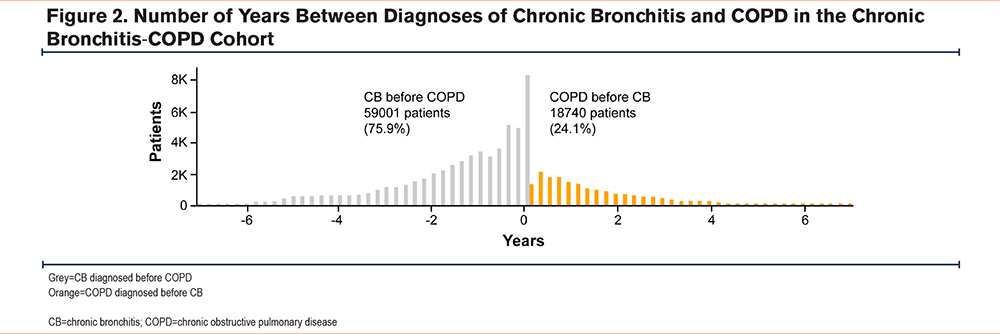
Time to Diagnosis Within the COPD-Chronic Bronchitis Cohort
More patients fulfilled the criteria for CB diagnosis before their first diagnosis with COPD (59,009 patients [75.9%]), than had COPD before being diagnosed with CB (18,740 patients [24.1%], Figure 2).
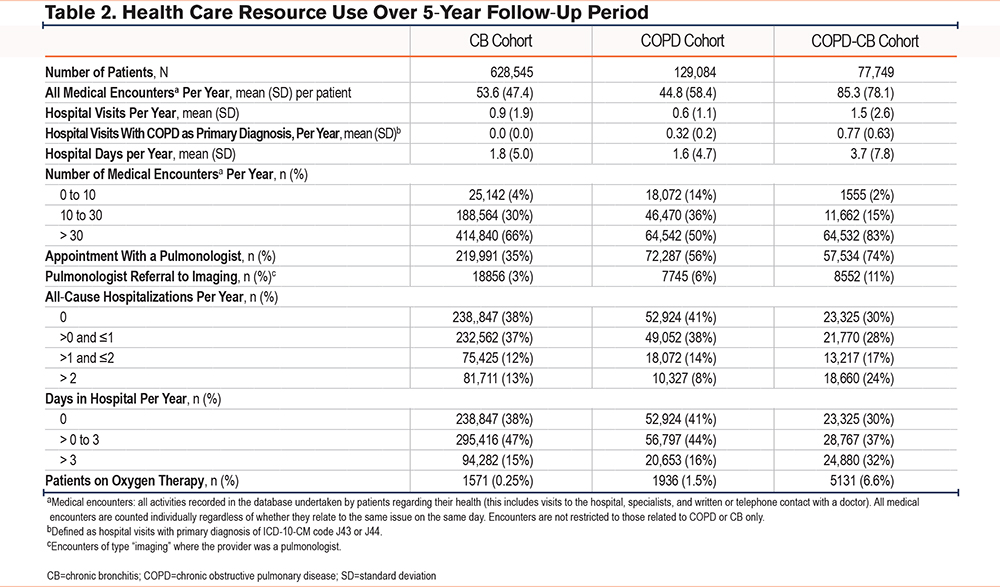
Health Care Resource Utilization
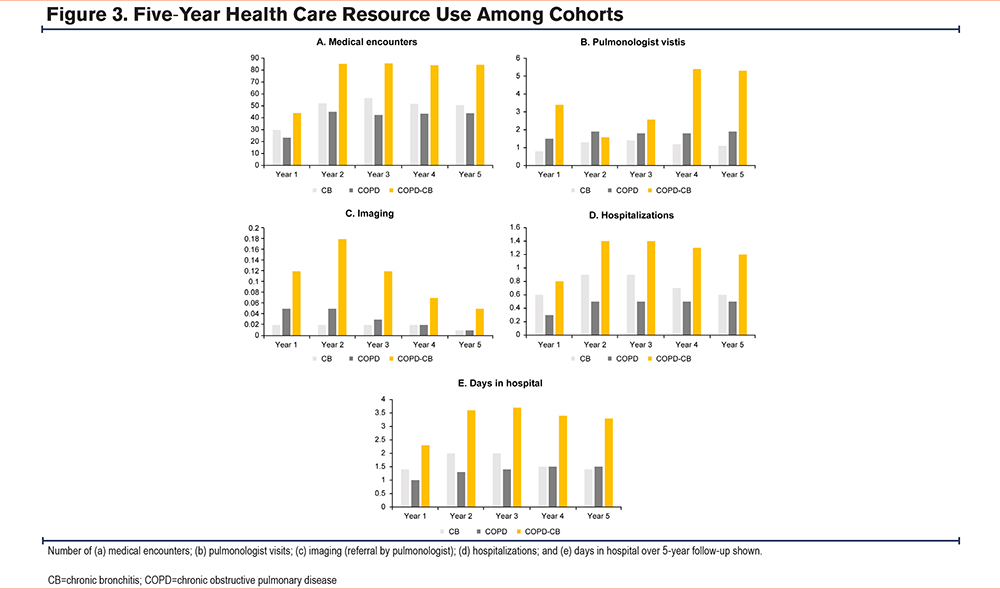
The COPD-CB cohort had the greatest number of interactions with health care (Table 1). Further, the COPD-CB cohort had the greatest HCRU in terms of the 5 metrics analyzed (medical encounters, interaction with a pulmonologist, referral to imaging, all-cause hospitalizations, and days in hospital, Table 2, Figure 3) and received the highest number of drug classes (Table S1 in the online supplement) among the 3 cohorts. Among patients with COPD or CB only, HCRU (represented by the 5 metrics described above) was similar over 5 years.
Medication Use
Macrolide prescriptions were more common in patients in the CB and COPD-CB cohorts than in the COPD cohort (Table S1 in the online supplement). In particular, individuals in the COPD-CB cohort experienced more frequent changes in classes of inhaled therapy, compared to patients with COPD alone, who did not have CB symptoms. This is illustrated in the more complex flow pattern observed for the COPD-CB cohort compared to the COPD-only cohort in Figure S2 in the online supplement.
Analysis of Common COPD Comorbidities
The COPD-CB cohort had a greater percentage of common COPD comorbidities than the COPD cohort and the CB cohort (Table S2 in the online supplement).
Discussion
Real-world data on the prevalence of the CB phenotype in patients with COPD are scarce.18 Similarly, clinical trials frequently focus on COPD as a whole rather than different clinical phenotypes.19 Additionally, the definition of CB varies significantly in epidemiologic studies.3 While the use of algorithms to interrogate EHRs is becoming more common,20,21 this study used a novel method of applying the classical definition of CB directly to physician notes in EHRs to determine the proportions of patients with COPD-CB or CB alone within the Optum® EHR. In clinical practice, physicians are unlikely to separate COPD and CB, therefore, the application of the CB definition to patient EHRs is a valuable tool to differentiate patients.
The proportion of patients with CB in our analysis is aligned with previously reported studies19,22 and the spirometry results for the patients at baseline also reflect previously reported distributions of phenotypes based on pulmonary function.23 A greater proportion of patients with CB only or with both CB and COPD were women, whereas, men and women were of almost equal proportion in patients with COPD only. Although it is frequently reported that CB is more common in men than women,24 some studies have shown that CB predominantly affects women, in line with our results.25,26
Treatment and HCRU burden are important in COPD, particularly as disease severity increases.27-32 Patients with COPD often have multiple comorbidities, which can also affect the HCRU burden.15 Compared to both the COPD cohort and the CB cohort, the COPD-CB cohort exhibited greater HCRU, including specialist visits. In addition, the COPD-CB cohort was exposed to a greater number of drug classes than the other cohorts (Table S2 and Figure S2 in the online supplement), suggesting that multiple medications were tried to improve control of burdensome patient symptoms. The frequent medication class changes in the COPD-CB cohort suggest that a clear treatment pathway was not available. Most patients in the COPD-CB cohort fulfilled the criteria for CB diagnosis before COPD diagnosis, and while persistent cough and sputum symptoms may have driven health care contact and diagnosis more rapidly than other COPD symptoms, it also suggests that halting the progression of CB to COPD-CB could reduce future HCRU burden. This reflects what has been previously described in other studies on a smaller scale33,34 and in a recent systematic review,35 where CB increased all-cause and respiratory disease-related mortality; and was associated with additional decline in FEV1.
Our results demonstrate the large HCRU burden for COPD-CB patients and confirm that most patients in this cohort initially had CB only, experiencing symptoms of cough and sputum which were reported in health care encounters, highlighting the importance of finding efficacious early interventions. This may also reflect the presence of cough and sputum symptoms driving patients to seek medical attention which then leads to the diagnosis of COPD. Interestingly, the HCRU of patients with CB alone was comparable or higher than in patients with COPD alone. Currently, there is uncertainty for clinicians and investigators about how to manage patients who present with CB and/or COPD-like symptoms and normal spirometric measurements.36 There have been suggestions to reintroduce the term “pre-COPD” to identify patients in whom spirometry is unable to detect airflow obstruction but who are at risk of subsequently developing COPD with a reduced FEV1/FVC ratio.37 Our study demonstrates that this severe progression path can be identified in the real world by asking patients whether they persistently experience symptoms of cough and sputum.
The longitudinal data presented here show that this patient population, across all cohorts, experiences a constant HCRU burden without clear improvement over time, and finding a treatment tailored to CB and/or COPD-CB would be instrumental to reducing this burden. There remains a need to improve treatment available, both to improve symptom control and to address underlying causes. New targets investigated for CB and COPD include the CFTR,7 interleukins (IL; IL-5, IL-4, IL-13, IL-33) or their receptors, and antithymic stromal lymphopoietin.38,39
A major strength of this study is that it is the first to report on CB and COPD in a broad, real-world U.S. population. The Optum® EHR database captures data from mostly commercially insured patients (44%), but also Medicaid, Medicare, and uninsured patients (12%, 12%, and 14%, respectively) at over 700 hospitals and 140,000 health care providers and contains data from over 113 million patients. These patients are distributed across the Midwest, Northeast, South, and West U.S. census regions, with the Midwest region over-represented as compared to U.S. census numbers. There are also some limitations in that patients >65 years of age are overrepresented compared to their proportion of the population. African American, Hispanic, and Asian patients are underrepresented when compared to their proportion of the U.S. population according to U.S. census data.40
The study has several limitations: HCRU and prescription data can be undercounted in Optum® EHR. However, as these data are likely missing at random, this limitation in the EHRs will affect all cohorts equally. The novel method of applying the classical definition of CB to EHRs to diagnose CB presents some limitations: first, the definition of CB was applied retrospectively, and second, because the CB cohort definition is a translation of the World Health Organization’s definition adapted to the EHR format (i.e., clinical notes), there is the possibility of false positive diagnoses, e.g., some of patients diagnosed with CB may have had bronchiectasis instead, which is common in patients with COPD and CB and is also associated with high HCRU.41,42 A review of symptoms in the Optum® EHR did not exclude negative descriptions (e.g., not detected, not confirmed, etc.). For a CB diagnosis, it was assumed that “symptoms for 2 consecutive 3-month periods” from a patient’s perspective were the equivalent of symptoms reported at least 90 days apart and no more than 180 days apart, but they could possibly constitute 2 episodes of acute bronchitis over a 3-month period. However, this spacing was necessary to minimize the risk of solely capturing one acute episode that was managed with 2 encounters. Data on smoking status were often missing, so only those patients where there was clear evidence of never smoking were categorized as “never smokers.” This group may, therefore, be underestimated, but this should affect all cohorts equally. Furthermore, the cause of CB remains unknown in those without a history of smoking or a diagnosis of COPD. Oxygen use was determined by a one-time listing of a Healthcare Common Procedure Coding System code and these data were limited in the dataset. Potential limitations also arise from the real-world nature of the clinical notes of the EHRs. For example, imaging may have been ordered but may not have been documented in the medical note as being ordered by a pulmonologist. Furthermore, any medical encounter on the same day is labeled as an individual medical encounter, i.e., there is no grouping in relation to a particular event or query, therefore, medical encounter data may appear higher than expected. More complete spirometric data could have provided more context for diagnosis within the COPD cohort and reduced the likelihood of misclassification. Furthermore, spirometry data would have enabled more detailed characterization of the CB cohort and especially the COPD-CB cohort which has the highest HCRU.
Notwithstanding the limitations of this study, these results demonstrate the prevalence of CB, both on its own and in patients with COPD. Furthermore, our study highlights the importance of a CB diagnosis, not only for patients with COPD where the disease may be more severe with worse clinical outcomes, but also for those patients with CB only, who experience a high burden of disease and have few treatment options. Patients with COPD, with or without CB, have many unmet needs. Besides the need to improve therapies available to patients, it is important to note that CB is not just a subtype of COPD but rather CB and COPD are 2 separate conditions that can overlap. There is a pressing requirement to detect CB at an earlier stage, before the onset of airway obstruction, to potentially alter the course of disease progression.
Acknowledgments
Author contributions: All authors meet the criteria for authorship in accordance with the International Committee of Medical Journal Editors. The authors take responsibility for the integrity of the data and analysis and the work as a whole. Editorial support to draft and revise the manuscript based on input from all authors was provided by Sorcha Mc Ginty, PhD. All authors contributed to revisions for critically important content. All authors approved this submitted version of the manuscript.
Data sharing: The data used were licensed from Optum and are not publicly available.
Financial support for medical editorial assistance was provided by Novartis Pharma AG, Basel, Switzerland. We thank Sorcha Mc Ginty, PhD, and Seónadh O’Leary, PhD, (Novartis Medical and Knowledge Solutions, Global Business Solutions, Dublin, Ireland) for their medical editorial assistance with this manuscript. We thank Raymond Przybysz, PhD, of Novartis Pharmaceuticals Corporation (New Jersey) for support with study design and statistical analysis.
Declaration of Interests
JK declares medical writing and editorial services funded by Novartis; grants to her institution from the American Thoracic Society Fellowship in Health Equity; research assistance for Primary Parents Award; COMMUNITY Center Investigator Development Core; NIH T32 HL134629; Weill Cornell Fund for the Future Award; consulting fee for 2023 Verona Pharma advisory board; medical writing support for a different manuscript from Novartis; medication samples delivered to her institution from Boehringer-Ingelheim and GlaxoSmithKline; a donor gift to her institution from Donna Redel Research Fund and Thomas King Fellowship. FJM declares Novartis as sponsor of the parent study; grants and consulting fees from AstraZeneca, GSK, Chiesi, Boehringer-Ingelheim, Novartis, Polarean, Sanofi/Regeneron, Sunovion, and TEVA Pharmaceuticals; consulting fees from CSL Behring, Pulmonx, Theravance/Viatris, and UpToDate; payment or honoraria for disease state presentation from AstraZeneca and GSK (partners of the SPIROMICS program and partners in the National Heart, Lung, and Blood Institute (NHLBI) CAPTURE validation study); participation in an event adjudication committee for Medtronic and participation in a data safety monitoring board for GSK. GJC declares funding for medical writing support from GSK; research grants and personal fees from AstraZeneca, Boehringer Ingelheim, GSK, PulmonX, Respironics Inc, and Respivant Sciences, research grants from ALung Technologies Inc, the American College of Radiology, the American Lung Association, BioScale Inc, BREATH Therapeutics Inc, the COPD Foundation, Coridea/ZIDAN, Corvus, Dr. Karen Burns of St. Michael’s Hospital, Fisher & Paykel Healthcare Ltd, Galapagos NV, Kinevent, Lungpacer Medical Inc, the NHLBI, Nurvaira Inc, the Patient-Centered Outcomes Research Institute, the Pulmonary Fibrosis Foundation, Spiration Inc, Steward St. Elizabeth’s Medical Center of Boston Inc, and Veracyte Inc; and personal fees from Amgen, AstraZeneca, Boehringer Ingelheim, Broncus Medical, CSA Medical, EOLO Medical, Gala Therapeutics, Helios Medical, Ion, Merck, Medtronic, Mereo BioPharma, NGM Biopharmaceuticals, Novartis, Olympus, the Implementation Group, and Verona Pharma. PA declares ownership of Novartis stock and was an employee of Novartis at the time of writing this article. EK and MS are Novartis employees and own Novartis stock. HK declares ownership of Novartis stock and was an employee of Novartis at the time of writing this article; honoraria from De Gruyter GmbH for role as Editor-in-Chief of the journal Epidemiologic Methods and her role as co-chair of the ISPOR Society Special Interest Group on Statistics in Health Economics and Outcomes Research. AL, VK, and BHL have no conflicts to declare.