Running Head: Severe Obesity in AATD-Associated COPD
Funding Support: This study was supported by an unrestricted research grant from AlphaNet.
Date of Acceptance: January 6, 2026 | Published Online Date: February 9, 2026
Abbreviations: AATD=alpha-1 antitrypsin deficiency; BMI=body mass index; CCI=Charlson comorbidity index; CDC=Centers for Disease Control and Prevention; COPD=chronic obstructive pulmonary disease; HRQoL=health-related quality of life; MCID=minimum clinically important difference; MCS=Mental Component Summary; mMRC=modified Medical Research Council; PCS=Physical Component Summary; SD=standard deviation; SE=standard error; SF-36=36-Item Short-Form Health Survey; SGRQ=St George's Respiratory Questionnaire
Citation: Choate R, Holm KE, Sandhaus RA, Mannino DM, Strange C. Severe obesity and alpha-1 antitrypsin deficiency-associated COPD: a dual burden on health and quality of life. Chronic Obstr Pulm Dis. 2026; 13(2): 177-183. doi: http://doi.org/10.15326/jcopdf.2025.0672
Note: Preliminary results from this study were presented in abstract format at the 2025 American Thoracic Society International Conference in San Francisco, California in May 2025.
Introduction
Chronic obstructive pulmonary disease (COPD) and obesity are 2 major public health challenges, each contributing substantially to morbidity, health care utilization, and mortality. In COPD, obesity is commonly associated with greater symptom burden and impaired health status.1 As COPD progresses to more advanced stages, underweight status becomes more prevalent and is independently associated with worsened outcomes, including increased mortality risk.2 These trends underscore the complex and dynamic relationship between body weight and COPD progression.
Although the clinical implications of weight extremes in COPD are increasingly recognized, key knowledge gaps persist regarding how body mass index (BMI) influences symptoms and health-related quality of life (HRQoL). These gaps are particularly pronounced in the context of alpha-1 antitrypsin deficiency (AATD)-associated COPD, a genetically defined type of COPD characterized by earlier onset.
Campos et al recently reported high rates of overweight and obese individuals in a historical cohort with AATD, associating elevated BMI with poorer clinical outcomes and reduced HRQoL.3 The current study expands on this work, examining the associations between BMI and clinical, demographic, and behavioral characteristics in a large cohort of individuals with AATD-associated COPD. We aimed to determine whether the findings reported by Campos et al are replicated in this expanded and current dataset and to further explain the role of body weight in shaping disease presentation and patient-centered outcomes in AATD-related COPD.
Methods
We performed a cross-sectional analysis of data from AlphaNet participants enrolled between 2008 and 2019. AlphaNet is a U.S.-based health management organization supporting individuals with AATD and lung disease through regular telephone calls to provide education and support and collect research data.4 Participants with lung disease and available self-reported weight and height data were included in these analyses. BMI was classified per the Centers for Disease Control and Prevention guidelines5 into underweight (<18.5kg/m2), healthy weight (18.5≤25kg/m2), overweight (25≤30kg/m2), obese (30≤40kg/m2), and severely obese (≥40kg/m2). Demographic and clinical characteristics (exacerbations, oxygen use, dyspnea severity), HRQoL (measured using St George’s Respiratory Questionnaire [SGRQ] and the 36-item Short Form Health Survey [SF-36]), and self-reported exercise behavior were examined. Charlson comorbidity index (CCI) scores were calculated using the established weights6 and categorized as 1, 2, and 3 or more. Since the study population has COPD, which contributes 1 point to the index, the 0 category was not applicable and, therefore, omitted.
Continuous variables were summarized as means (standard deviation) and compared across BMI groups using analysis of variance with Bonferroni-adjusted pairwise comparisons. Categorical variables were analyzed via Chi-square tests with false discovery rate adjustments (Benjamini-Hochberg).7 A multivariable linear regression model was fit with SGRQ Total score as the outcome, adjusted for covariates selected a priori based on clinical relevance and prior research, including age, sex, smoking history, CCI, and exacerbation frequency.
Analyses were conducted using SAS version 9.4 (SAS; Cary, North Carolina). The study was approved by the University of Kentucky Institutional Review Board (#43435).
Results
Of 8154 participants (mean age 57.6±11.2 years), BMI distribution was: underweight 4.7%, healthy weight 35.6%, overweight 30.1%, obese 23.9%, and severely obese 5.8% (Table 1). Overall, 56.8% reported ≥2 exacerbations in the past year, and 28% had modified Medical Research Council (mMRC) grade 4 dyspnea. Less than 1% (n=87) had lung transplants and 7 individuals had liver transplants.
Severely obese participants were the youngest group (mean age 54.0±11.1 years) and had the highest prevalence of self-reported asthma (47.5%) and ≥2 exacerbations (71.4%). Nearly half (49.5%) of all participants used oxygen, and 44.4% reported productive cough, with no significant differences across BMI categories.
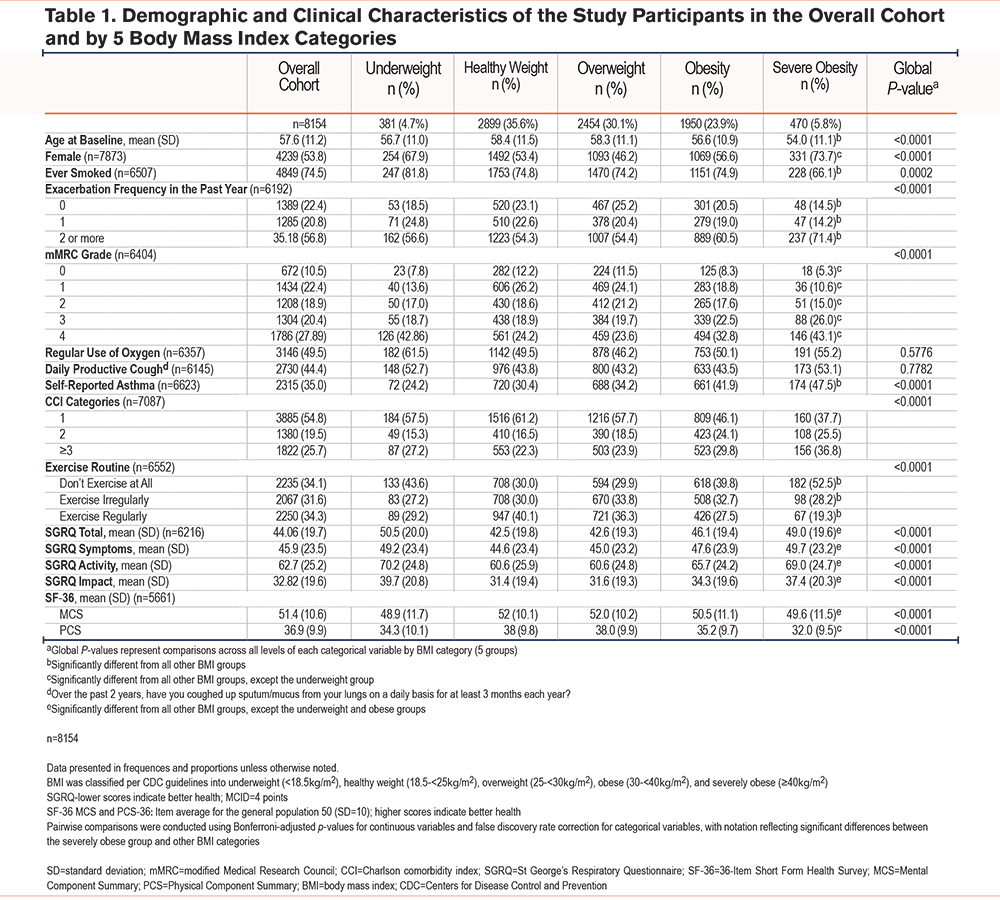
Dyspnea severity was greatest among underweight and severely obese individuals. Only 19% of those in the severely obese group reported regular exercise, while over half (52.5%) reported no exercise at all, significantly worse than other groups (Table 1 and Figure 1).
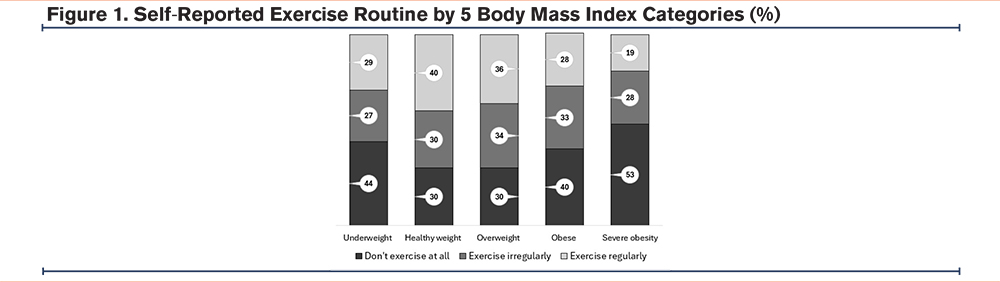
HRQoL measures were significantly impaired in the severely obese group. SF-36 Physical Component Summary scores were lower than in all other groups except the underweight, indicating poor physical health in these groups. Mental Component Summary scores were also impaired. SGRQ total scores were high in the severely obese group indicating poor HRQoL, however, not significantly different from the underweight or obese groups (Figures 2A and 2B). SGRQ subscale scores were similarly poor among underweight, obese, and severely obese individuals, with the greatest impairments in the activity domain.
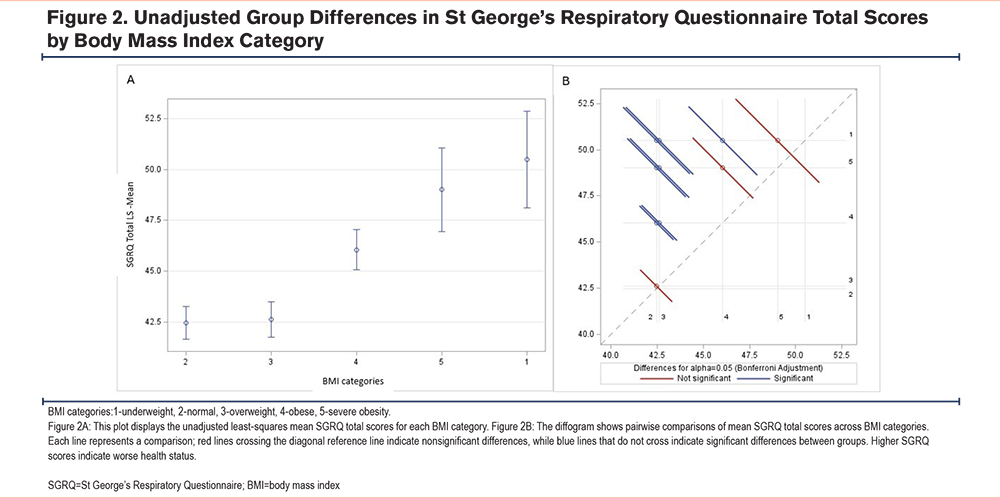
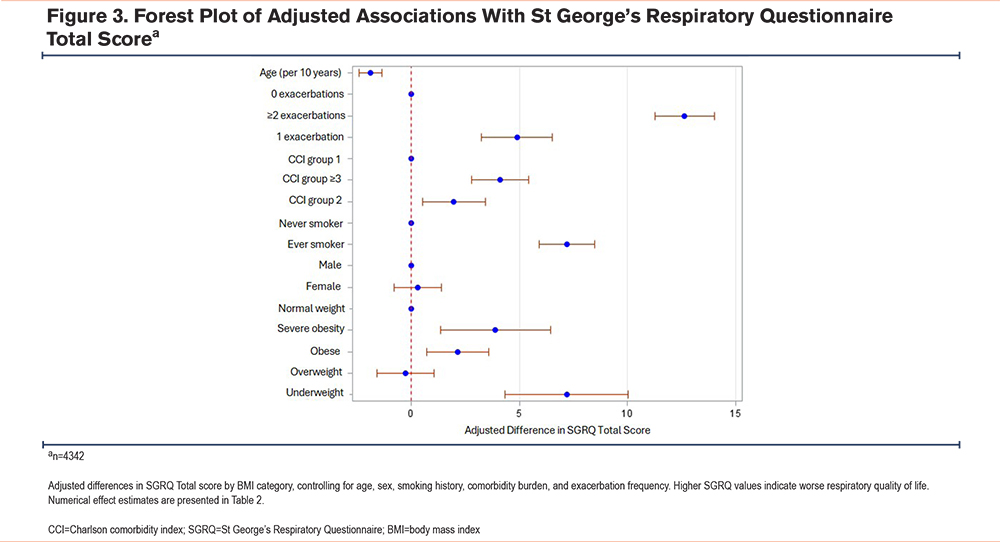
In the multivariable model adjusting for age, sex, smoking, comorbidity burden (CCI), and exacerbation frequency, both underweight and severe obesity remained independently associated with worse SGRQ Total scores (p<0.001), indicating that the relationship between BMI extremes and impaired respiratory health is not fully attributable to studied comorbidities or measured clinical confounders (Table 2, Figure 3).
Discussion
In this large cohort of individuals with AATD-associated COPD, severe obesity was associated with worse clinical outcomes and poorer HRQoL, extending the findings of prior studies in a broader sample.3 Notably, severely obese individuals were younger on average, suggesting that excess weight may contribute to earlier or more aggressive disease manifestation in this population.
Our findings suggest a dose-response relationship between BMI and exacerbation frequency, with the highest burden observed among the severely obese, paralleling observations in non-AATD COPD populations.8 Severely obese individuals also reported the most severe dyspnea and the lowest levels of physical activity, which likely contributes to a cycle of deconditioning, inactivity, and worsening respiratory symptoms.9
HRQoL was substantially impaired in both severely obese and underweight participants. Differences in SGRQ total scores between these groups and normal/overweight participants exceeded the minimum clinically important difference of 4 points, indicating a meaningful impact on perceived health status. Similarly, both physical and mental HRQoL assessed via SF-36 were significantly reduced in these extreme weight groups, reinforcing the detrimental effects of both obesity and underweight in AATD-associated COPD. Our multivariable analyses adjusting for age, sex, smoking history, CCI, and exacerbation frequency demonstrate that both underweight and severe obesity were significantly associated with worse SGRQ Total scores. These findings suggest that low or high BMI contributes to poor respiratory quality of life in AATD, independent of comorbidities or demographic factors.
Self-reported exercise patterns provide additional insight into behavioral contributors to disease burden. Only 1 in 5 severely obese individuals in our study reported regular exercise, while more than half reported no exercise at all. Barriers such as dyspnea, fatigue, and musculoskeletal limitations likely play a role. Nonetheless, this finding highlights the need for tailored, feasible physical activity interventions. Sedentary behavior is a known risk factor for adverse metabolic and cardiovascular outcomes,10 and growing evidence suggests that obesity may accelerate aging processes through chronic inflammation and physiologic stress.11
These results underscore the importance of a comprehensive, individualized approach to weight and activity management in AATD-associated COPD. While nutritional and exercise interventions are central to non-AATD COPD care, specific guidance for the AATD population remains limited.12 However, our previous work and that of others support the effectiveness of tailored interventions in improving BMI and exercise tolerance in individuals with AATD.13,14
It is important to note that the literature on obesity and COPD remains mixed.15 Some studies have found that higher BMI is associated with worse outcomes, including greater dyspnea, reduced HRQoL, and increased hospitalizations.8 Conversely, other reports suggest that higher BMI may be linked to lower exacerbation rates.16 This conflicting evidence has been described as the “obesity paradox,”15 highlighting the complexity of BMI as a surrogate for adiposity and health risk, particularly in individuals with high muscle mass. These nuances underscore the need for more precise and individualized measures of body composition in future research and clinical care.
Strengths of our study include the large, well-characterized cohort of individuals with AATD-associated COPD and the inclusion of both disease-specific and generic HRQoL measures. The availability of exercise behaviors data also adds a valuable patient-centered dimension. However, several limitations should be noted. All data were self-reported, which may introduce recall or reporting bias, particularly related to physical activity. Additionally, the cross-sectional design precludes causal inference and limits the ability to assess temporal changes in BMI or outcomes. BMI also does not account for differences in body composition or fat distribution, which may affect the interpretation of obesity-related risk. Although liver involvement is an important aspect of AATD, the small number of participants with liver disease or liver transplantation precluded meaningful interpretation. Finally, information on potential treatment-related confounders, including pulmonary rehabilitation, airway clearance, statins, GLP1 inhibitors, or surgical procedures for weight control, was not routinely collected in the AlphaNet dataset and could not be incorporated into the analyses.
Conclusion
In this large cohort of individuals with AATD-associated COPD, severe obesity was associated with younger age, higher exacerbation frequency, greater dyspnea, and worse HRQoL, mirroring the clinical burden observed in underweight individuals. These findings underscore the dual vulnerability posed by both obesity and underweight in this population and highlight the importance of addressing weight and activity through individualized clinical and behavioral interventions.
Acknowledgements
Author contributions: RC performed the data analysis and drafted the manuscript. CS, RAS, KEH, and DMM contributed substantially to the study design, data interpretation, and critical revision of the manuscript. All authors reviewed and approved the final version.
Other acknowledgements: The authors would like to thank the AlphaNet Coordinators and participants for their contributions to this study.
Declaration of Interest
RC reports research support from AlphaNet. KEH receives consulting income from AlphaNet. CS is a paid AlphaNet medical director. DMM reports consulting income from AstraZeneca, GlaxoSmithKline, AmGen, Genentech, Roche, Regeneron, Sanofi, Lilly, Chiesi, Up to Date, and the COPD Foundation. RAS is a paid AlphaNet medical director.