Running Head: Sleep Quality in Vietnamese COPD Patients
Funding Support: None
Date of Acceptance: February 2, 2026 | Published Online Date: February 9, 2026
Abbreviations: BD=bronchodilator; CAT=COPD Assessment Test; CI=confidence interval; COPD=chronic obstructive pulmonary disease; FEV1=forced expiratory volume in 1 second; GOLD=Global initiative for chronic Obstructive Lung Disease; mMRC=modified Medical Research Council dyspnea scale; OR=odds ratio; PSQI=Pittsburgh Sleep Quality Index; SD=standard deviation
Citation: Thuy PT, Thu NT, Khan NC. Silent nights, restless lungs: sleep quality and associated factors among COPD patients in Vietnam—a cross-sectional study. Chronic Obstr Pulm Dis. 2026; 13(2): 104-110. doi: http://doi.org/10.15326/jcopdf.2025.0664
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive respiratory condition characterized by persistent airflow limitation and respiratory symptoms that substantially impair daily functioning and quality of life. Worldwide, COPD is a leading cause of morbidity and mortality, particularly in aging populations. In Vietnam, the burden of COPD continues to rise alongside population aging and high tobacco exposure, posing significant challenges to the health care system. Sleep disturbances are frequently reported among individuals with COPD but remain underrecognized in routine clinical practice. Poor sleep quality is linked to higher exacerbation rates, worse dyspnea, impaired cardiovascular function, and heightened psychological distress.1 Sleep disturbances constitute a major but often under-recognized comorbidity in patients with COPD. Beyond impaired sleep quality, individuals frequently experience a spectrum of sleep disorders, including insomnia, restless legs syndrome, periodic limb movement disorder, and sleep-disordered breathing such as overlap syndrome (concomitant COPD and obstructive sleep apnea).2,3 Mechanistically, several COPD-related features—including nocturnal hypoxemia, dynamic hyperinflation, respiratory muscle loading, chronic cough, and nocturnal wheezing—directly disrupt sleep architecture and reduce both slow-wave and REM sleep.4 Inflammatory activation and systemic corticosteroid use may further contribute to sleep fragmentation.5
Poor sleep quality in COPD is not merely a symptom but a clinically meaningful prognostic marker. Large cohort studies show that sleep disturbances are independently associated with increased risk of acute exacerbations, accelerated decline in lung function, reduced physical activity, and higher all-cause mortality.1 According to the study by Li et al,6 age, gender, symptom burden (COPD Assessment Test [CAT] score), and self-efficacy are key determinants of sleep quality in patients with COPD. Their findings indicate that individuals with COPD—particularly women, older adults, those with lower self-efficacy, and those with more severe symptoms—are at higher risk of sleep disturbances and poorer sleep quality.6 Although international studies report sleep disturbances in 40%–70% of COPD patients,7,8 Vietnamese data remain limited, and evidence on behavioral factors, such as electronic device use before bedtime, remains particularly scarce. Therefore, this study sought to characterize sleep quality among COPD outpatients at Bach Mai Hospital, and to determine the demographic, clinical, and behavioral factors associated with poor sleep quality.
Methods
This cross-sectional analytical study was conducted at the COPD outpatient clinic of Bach Mai Hospital in Hanoi, Vietnam, between February 2025 and May 2025. Consecutive patients aged 40 years old or older with a physician-confirmed diagnosis of COPD, established according to the 2024 Global initiative for chronic Obstructive Lung Disease (GOLD)9 criteria (postbronchodilator forced expiratory volume in 1 second [FEV1] to forced vital capacity <0.70), who attended routine follow-up visits were recruited. Patients unable to communicate effectively or unwilling to provide informed consent were excluded. All participants were clinically stable outpatients not receiving long-term oxygen therapy, home noninvasive ventilation, or continuous positive airway pressure treatment, as these conditions were excluded to avoid confounding effects on sleep quality.
Based on a previously reported 74.8% prevalence of poor sleep quality,10 a 95% confidence interval (CI) level, and a 5% margin of precision, the minimum sample size required was 289, which was achieved. Sleep quality was assessed using the validated Vietnamese version of the Pittsburgh Sleep Quality Index (PSQI) (Cronbach’s α = 0.79). Dyspnea severity was evaluated with the modified Medical Research Council (mMRC) scale, overall symptom burden with the CAT, and airflow limitation with postbronchodilator FEV1.
Descriptive statistics, including means, standard deviations, and frequency distributions, were used to summarize patient characteristics. Comparisons between patients with poor versus good sleep quality (PSQI >5 versus ≤5) were performed using Chi-square tests as appropriate. Univariate associations between sleep quality (poor versus good, based on PSQI) and clinical characteristics were examined using Chi-square analyses, and unadjusted odds ratios (ORs) with 95% CIs were calculated from 2×2 contingency tables. The study protocol was approved by the Ethics Committee of Bach Mai Hospital, and all participants provided written informed consent.
Results
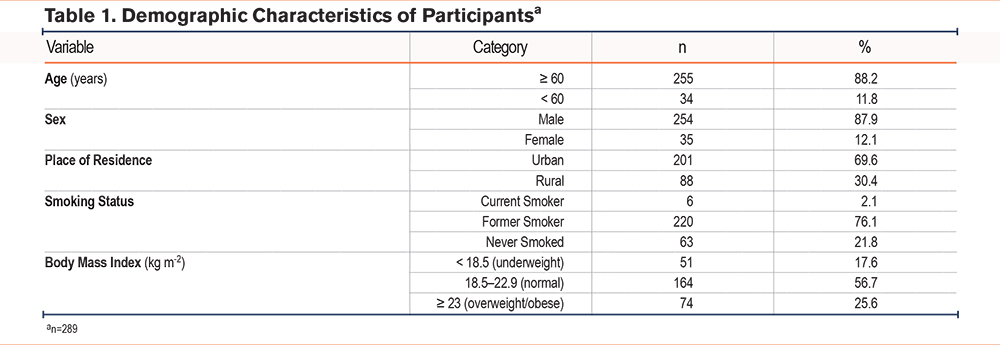
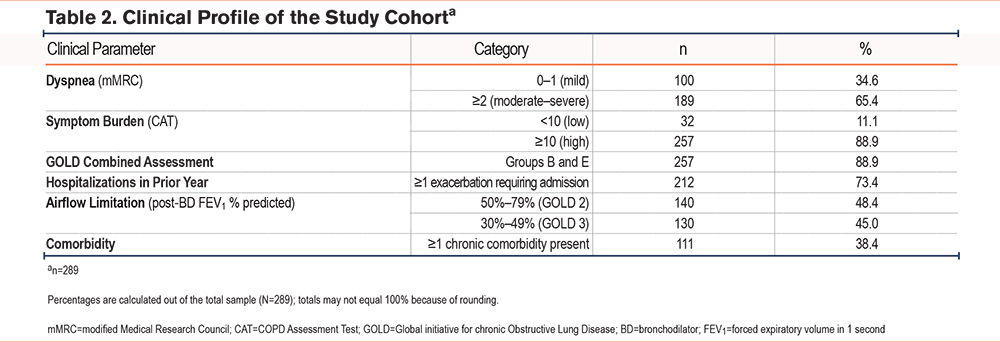
The cohort consisted predominantly of older male patients, with 88.2% aged ≥ 60 years and 87.9% being men (Table 1). Although only 2.1% were current smokers, a substantial proportion (76.1%) were former smokers, reflecting a considerable cumulative tobacco exposure. Nutritional status varied, with 17.6% classified as underweight and 25.6% as overweight or obese (Table 1). Symptom burden was notable: 65.4% reported moderate-to-severe dyspnea (mMRC grade ≥ 2), and 88.9% had CAT scores ≥ 10, placing the majority in GOLD groups B or E regardless of spirometric severity (Table 2). In line with this clinical profile, 73.4% had experienced at least one COPD-related hospitalization in the previous year (Table 2).
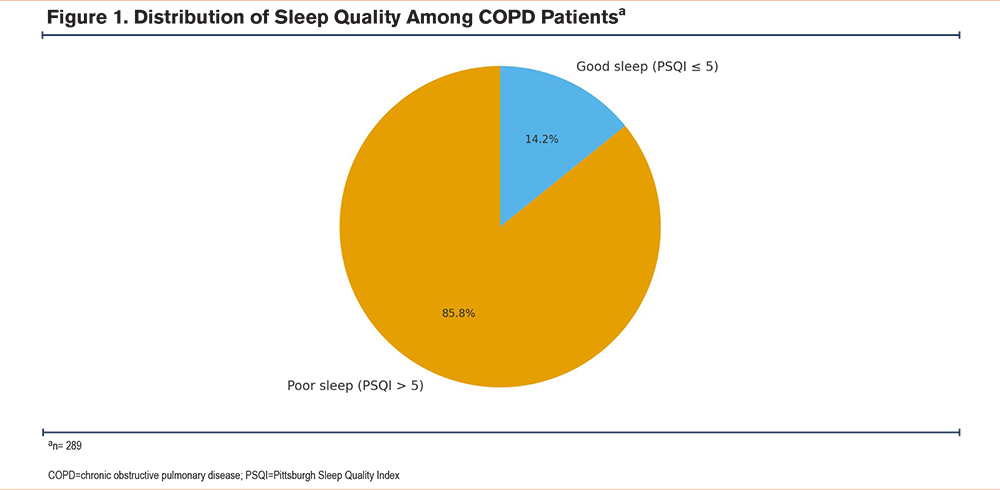
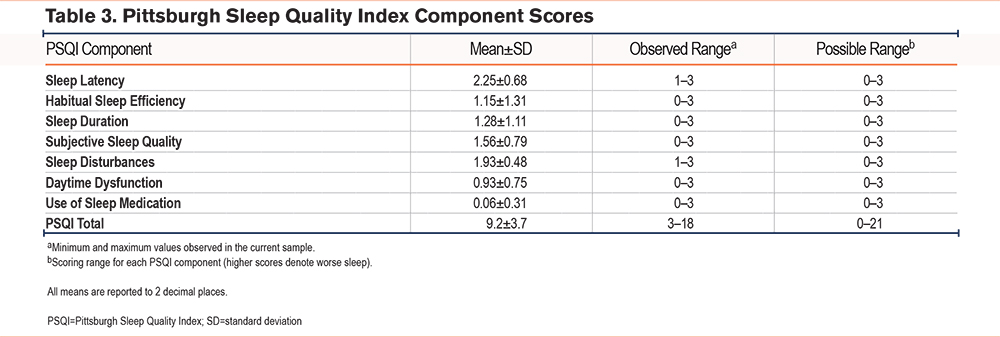
Sleep-related outcomes were similarly notable. The mean global PSQI score was 9.2±3.7, and 85.8% of participants exceeded the established cutoff for poor sleep (PSQI >5) (Figure 1; Table 3). Among the PSQI domains, the most affected were sleep latency (mean 2.25±0.68), nocturnal disturbances (1.93±0.48), and subjective sleep quality (1.56±0.79), whereas the use of hypnotic medications was minimal (0.06±0.31) (Table 3).
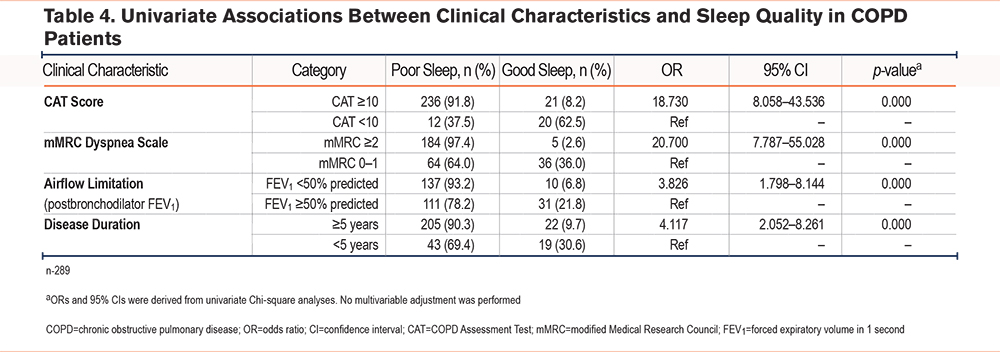
Significant differences in sleep quality were observed across multiple clinical characteristics. Patients with higher symptom burden (CAT score ≥ 10) demonstrated markedly poorer sleep, with 91.8% reporting poor sleep compared with 37.5% in the CAT score <10 group. These individuals had substantially increased odds of poor sleep (OR = 18.73; 95% CI: 8.06–43.54; p <0.001). Similarly, dyspnea severity was strongly associated with impaired sleep. Among patients with an mMRC grade ≥ 2, 97.4% experienced poor sleep, compared with 64.0% in those with and an mMRC grade of 0–1 (OR = 20.70; 95% CI: 7.79–55.03; p <0.001). Airflow limitation also showed a significant relationship with sleep quality. Poor sleep occurred in 93.2% of patients with a postbronchodilator FEV1 <50%, versus 78.2% in those with an FEV1 ≥ 50% (OR = 3.83; 95% CI: 1.80–8.14; p <0.001). In addition, disease duration of ≥ 5 years was associated with a higher prevalence of poor sleep (90.3% versus 69.4%), corresponding to an increased likelihood of sleep impairment (OR = 4.12; 95% CI: 2.05–8.26; p <0.001) (Table 4). Overall, these findings indicate that poorer sleep quality was consistently observed in patients with more severe symptoms, greater dyspnea, worse airflow limitation, and longer disease duration.
Discussion
In our cohort, 73% of participants had experienced a COPD-related hospitalization within the preceding year—a proportion substantially higher than that reported in many international COPD cohorts. This pattern reflects the clinical context of Bach Mai Hospital, one of the largest respiratory care centers in Northern Vietnam. The COPD management clinic typically receives patients with more severe disease, higher symptom burden, and frequent exacerbations, many of whom fall into GOLD Group E. This case-mix likely contributed to the elevated hospitalization rate and should be considered when interpreting the prevalence and severity of sleep disturbances observed in this study.
The present study confirms that sleep disturbance is a significant problem in COPD. The prevalence of poor sleep quality in our study (85.8%) is similar to the findings of a U.S. study by Scharf et al1 (86%) and an Indian study by Kushwaha et al10 (71.2%). Thus, it is clear that sleep disturbance needs to be given more attention in the multidisciplinary management of COPD patients, which is often overlooked by clinicians. The mean PSQI score in our cohort (9.2±3.7) closely mirrors the findings of Li et al6 who reported a comparable level of sleep impairment (10.07±4.23). This consistency across studies suggests that poor sleep quality is a pervasive issue among COPD patients regardless of geographic or health care setting. Such convergence further reinforces the need for routine assessment and targeted management of sleep disturbances in this population.
Among the PSQI domains, sleep latency showed the greatest decline in our cohort (mean 2.25±0.68), while hypnotic use remained insignificant (0.06±0.31). These findings are consistent with those of Kushwaha et al10 who reported the highest mean score for sleep latency (1.76±0.8) and the lowest for hypnotic use (0.08±0.3). This consistency across studies suggests that delayed sleep onset is a common and prominent concern in COPD patients, while reliance on pharmacological sleep aids remains low. Such results suggest that there is a real need for further research and professional society guidelines in behavioral, nonpharmacological, and even pharmacological management to address sleep initiation difficulties in this patient population.
Our findings demonstrate a clear and consistent relationship between disease severity and impaired sleep quality in patients with COPD. Symptom burden, as reflected by CAT scores, showed one of the strongest associations, with individuals reporting more severe symptoms exhibiting nearly 19-fold higher odds of poor sleep. This is highly concordant with the study by Li et al which similarly identified CAT score as an independent determinant of sleep quality, emphasizing the central role of symptom perception in shaping sleep outcomes.6 Dyspnea severity, captured by the mMRC scale, was an equally powerful determinant; patients with an mMRC grade ≥ 2 had markedly higher rates of poor sleep, supporting the well-established observation that breathlessness, particularly at night, induces frequent awakenings, increases sleep latency, and heightens nocturnal sympathetic activation.11
Airflow limitation also contributed significantly to sleep impairment. Patients with an FEV1 <50% predicted were substantially more likely to experience poor sleep, suggesting that reduced ventilatory reserve and nocturnal hypoventilation may play a role in sleep fragmentation. Furthermore, longer disease duration was associated with a greater likelihood of poor sleep, possibly reflecting the cumulative impact of progressive respiratory symptoms, chronic inflammation, and comorbidities on long-term sleep health. Unlike several previous studies that found no association between FEV1 and sleep quality,11,12 our cohort demonstrated a clear relationship between more severe airflow limitation and poorer sleep. This discrepancy may be explained by the higher burden of symptoms and dyspnea in our population, in whom reduced ventilatory reserve and greater nocturnal respiratory instability are more likely to disrupt sleep. In settings where disease severity is greater, as in our sample, FEV1 may, therefore, play a more prominent role in shaping sleep quality.
Taken together, these results underscore that both symptom severity and physiological impairment are key drivers of sleep disturbance in COPD. They highlight the need for clinicians to routinely evaluate sleep quality—particularly in patients with high CAT scores or mMRC grades, marked airflow limitation, or longstanding disease—so that targeted symptom control, dyspnea management, and behavioral interventions can be integrated into comprehensive COPD care.
Future Research Directions
Future research should further explore the bidirectional relationship between sleep quality and COPD severity, including whether poor sleep acts as a trigger for exacerbations or represents a component of the broader COPD vicious cycle. Longitudinal studies will be essential to clarify causal pathways and to determine whether improving sleep quality can modify disease progression or reduce exacerbation risk.
Limitations
This study has several limitations that should be considered when interpreting the findings. First, the single-center design may limit the representativeness of the sample and reduce the generalizability to broader COPD populations across Vietnam. Second, the assessment of sleep quality relied on self-reported measures, which are subject to recall bias and may not fully capture objective sleep disturbances. Third, the cross-sectional design precludes any inference of causality between clinical characteristics and sleep outcomes. Despite these limitations, the study provides important and timely evidence on sleep quality among patients with COPD in Vietnam, where sleep medicine remains in an early stage of development and local data are scarce.
Conclusion
This study confirms that poor sleep quality is a common and clinically significant characteristic in Vietnamese outpatients with COPD. Poor sleep quality is strongly associated with dyspnea and respiratory function. In clinical practice, attention should be paid to sleep disorders, especially in symptomatic patients. Further studies are needed to evaluate the bidirectional relationship between sleep quality and respiratory function in patients with COPD.
Acknowledgements
Author contributions: PTT contributed to the study methodology, performed the data analysis, interpreted the results, drafted the original manuscript, and revised the manuscript for important intellectual content. NTT was responsible for data collection and participated in data analysis. NCK contributed to the study design and methodology and critically reviewed and revised the manuscript for intellectual content. All authors reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Other acknowledgements: We sincerely acknowledge the valuable support of the Vietnam Society of Sleep Medicine, whose expertise and collaborative efforts have greatly facilitated the development and implementation of sleep medicine-related research at Bach Mai Hospital. Their guidance, academic contributions, and continuous encouragement have played an important role in advancing our studies and strengthening the capacity for clinical and scientific work in this field.
AI policy compliance: In accordance with this policy, the authors disclose that ChatGPT (OpenAI o3, July 2025 release) was employed solely to refine English phrasing and to standardize table layout. All AI-generated suggestions were critically reviewed and edited by the authors, who assume full responsibility for the final content of the manuscript. No AI tool was used to generate, analyse, or interpret study data.
Declaration of Interest
The authors have no conflicts of interest to declare.