Running Head: CRS and Pseudomonas aeruginosa in Bronchiectasis
Funding Support: None
Date of Acceptance: February 13, 2026 | Published Online Date: February 23, 2026
Abbreviations: AFB=acid-fast bacilli; BMI=body mass index; BRR=Bronchiectasis and NTM Research Registry; CF=cystic fibrosis; CI=confidence interval; COPD=chronic obstructive pulmonary disease; CRS=chronic rhinosinusitis; FEV1=forced expiratory volume in 1 second; FEV1 %pred=forced expiratory volume in 1 second percentage predicted; H influenzae=Haemophilus influenzae; IBD=inflammatory bowel disease; IQR=interquartile range; NO=nitric oxide; NTM=nontuberculous mycobacteria; OR=odds ratio; P aeruginosa=Pseudomonas aeruginosa; PCD=primary ciliary dyskinesia; S aureus=Staphylococcus aureus; SD=standard deviation
Citation: Grabauskas T, Brunton AE, Metersky ML, et al. Association of chronic rhinosinusitis and Pseudomonas aeruginosa in sputum of patients with non-cystic fibrosis bronchiectasis. Chronic Obstr Pulm Dis. 2026; 13(2): 158-166. doi: http://doi.org/10.15326/jcopdf.2025.0732
Introduction
Non-cystic fibrosis bronchiectasis (henceforth referred to as bronchiectasis) is a chronic pulmonary condition characterized by permanent bronchial dilatation, mucus plugging, and, often, chronic bacterial airway infection. Patients commonly have symptoms of cough, dyspnea, and fatigue.1 Several factors are linked to bronchiectasis exacerbations and progression, including age, smoking, treatment adherence, comorbid conditions such as chronic rhinosinusitis (CRS),2 and chronic lower airway infections—particularly those caused by Pseudomonas aeruginosa (P aeruginosa).1-3
CRS is defined as prolonged inflammation of the nasal and paranasal sinuses, causing nasal discharge, congestion, facial pain, and olfactory disturbances for at least 12 weeks and accompanied by endoscopic or radiographic evidence of mucosal inflammation.4 Its reported prevalence among people with bronchiectasis ranges from 7.1% to 62% depending on study population and the number of centers involved in data collection.5,6 In contrast, the estimated prevalence in the general population7,8 is between 3.0% and 8.7%. Importantly, CRS has been associated with increased bronchiectasis radiographic disease severity, reduced quality of life, and a shorter time to exacerbation.6
Bacterial infection of the lower airways is a key factor associated with the severity of bronchiectasis. For example, a positive pathogenic bacterial sputum culture in people with bronchiectasis was shown to be associated with lower percentage of forced vital capacity, forced expiratory volume in 1 second (FEV1) percentage, and longer symptom duration.9 P aeruginosa is the most commonly isolated pathogen in people with bronchiectasis,9,10 with estimates ranging from 21% to 33%. P aeruginosa has been associated with worsened bronchiectasis outcomes, including more frequent exacerbations and reduced FEV1 percentage predicted (FEV1 %pred).11 Other frequently cultured pathogens in people with bronchiectasis include Haemophilus influenzae (H influenzae) and Staphylococcus aureus (S aureus).10 Importantly, H influenzae has been associated with higher bronchiectasis severity index and exacerbation frequency.12
CRS is associated with bacterial infection of the paranasal sinuses,13 with S aureus being the most common pathogen in maxillary sinuses of people with CRS.14 Concordance between upper and lower airway sputum cultures has been observed in people with bronchiectasis.15 Given the high prevalence of CRS in individuals with bronchiectasis, CRS may act as a reservoir for pathogenic bacteria, and thereby, increase the risk of lower airway infections.16,17 Therefore, we aimed to investigate the relationship between a history of CRS and lower airway infection with P aeruginosa, S aureus, and H influenzae in patients enrolled in the U.S. Bronchiectasis and NTM Research Registry (BRR). We hypothesized that patients with bronchiectasis and a physician-reported history of CRS would have a higher prevalence of sputum culture positivity for one or more of these 3 pathogens.
Study Design and Methods
Data Source
The BRR is a centralized database of physician-reported adult patients diagnosed with bronchiectasis and/or nontuberculous mycobacteria (NTM) from 29 sites across the United States. Following patient consent, data is abstracted from electronic medical records from the baseline period, defined as the 2-year look-back period up to and including the date of consent. Follow-up data is collected annually thereafter. Standardized data collection forms include demographics, medical history and clinical characteristics, respiratory symptoms, microbiology, treatment, and imaging. The data coordinating center and each participating site received institutional review board approval.10
Study Population and Design
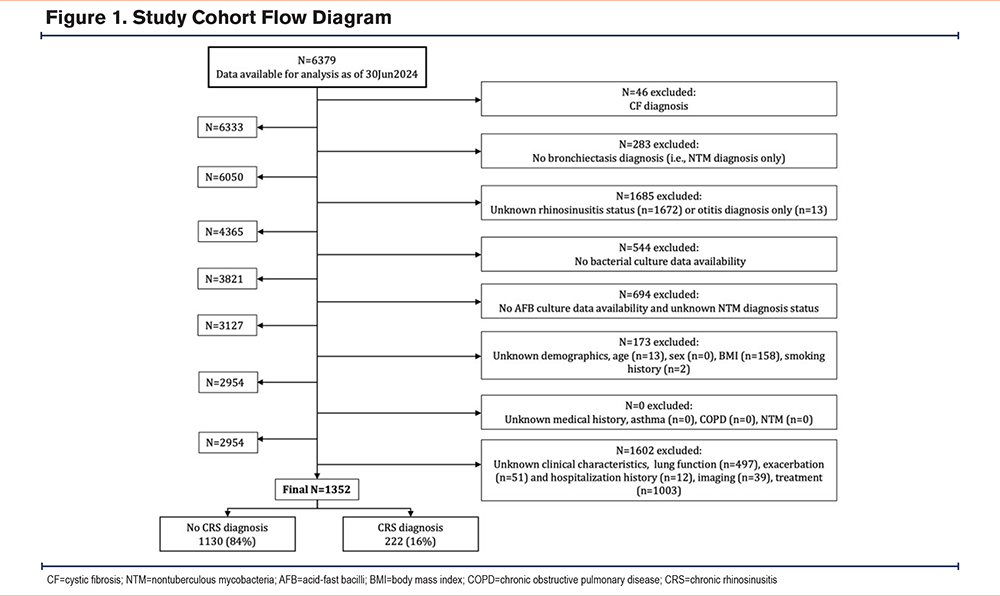
People with bronchiectasis and known CRS disease status at baseline were included. Patients with unknown demographics (age, sex, body mass index [BMI]), medical history (smoking history, asthma, chronic obstructive pulmonary disease [COPD]), NTM, clinical characteristics (lung function, exacerbation and hospitalization history, imaging, treatment), and microbiology (bacterial and acid-fast bacilli [AFB] cultures) were excluded, as were patients with a diagnosis of cystic fibrosis (CF). This was a cross-sectional study, with all data evaluated from the baseline period only.
Study Variables and Outcomes
Patients were defined as either having or not having a CRS diagnosis, as reported in the BRR. CRS in the BRR is based on physician-reported diagnosis from each participating site. Specifically, the investigators were asked to select all co-existing conditions from the list of 37 possibilities, including rhinosinusitis, that a patient has ever been diagnosed with. Sputum positivity for the study outcomes, P aeruginosa, S aureus, and H influenzae were defined as at least 1 positive culture during the baseline period. Covariates included patient age, sex, and several factors known or stipulated to be associated with respiratory P aeruginosa infection such as BMI,18 smoking history19 (current or former versus never), asthma, COPD,20 NTM,21 prebronchodilator FEV1%pred,20 history of at least 1 exacerbation during the baseline period,22 history of at least 1 exacerbation- or pulmonary-related hospitalization during the baseline period,22 total number of lobes involved on the patient’s computed tomography scan,23 chronic macrolide therapy,24 and inhaled corticosteroid use.25 NTM diagnosis was defined as those with a physician-reported diagnosis meeting the international consensus disease criteria26,27 plus documentation of at least 1 positive AFB culture during the baseline period.
Statistical Analysis
Descriptive statistics were computed overall and by CRS diagnosis status for the demographics, comorbidities, clinical characteristics, and treatment history of the study population. Chi-square, independent 2-sample t-test, and Wilcoxon tests were used to determine the relationship between categorical variables and the mean or median difference between continuous variables, respectively.
We used a multivariable logistic regression model to determine the odds of at least 1 positive P aeruginosa culture during the baseline period among those with CRS. This process was repeated for S aureus and H influenzae. All models were adjusted for the previously mentioned covariates. All analyses used a significance level of 0.05 (p<0.05). No corrections were made for multiplicity. SAS version 9.4 (SAS Institute; Cary, North Carolina) was used for all analyses.
Results
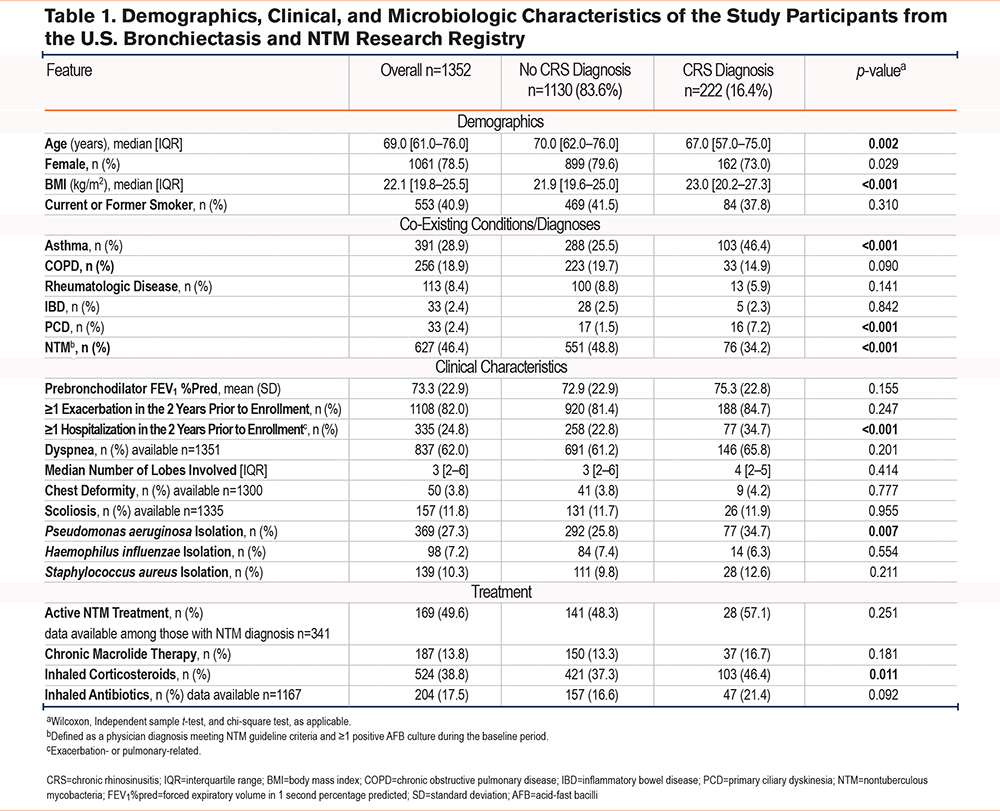
Study Population
A total of 1352 patients met the study eligibility criteria (Figure 1). Study participants’ demographic, clinical, and microbiologic characteristics are shown in Table 1. Of the study population, 16% (n=222) had physician-reported CRS. The CRS group was less likely to be female (73% CRS versus 80% non-CRS group; p=0.03) and to have a history of NTM (34% CRS versus 49% non-CRS group; p<0.001). Conversely, patients with CRS had higher BMIs (23.0kg/m2 in CRS versus 21.9kg/m2 in non-CRS group; p<0.001), were more likely to have a history of asthma (46% CRS versus 25% non-CRS group; p<0.001) and primary ciliary dyskinesia (7% CRS versus 2% non-CRS group; p<0.001), use inhaled corticosteroids (46% in CRS versus 37% in non-CRS group; p=0.01), and be hospitalized for pulmonary-related conditions within 2 years prior to enrollment in the BRR (35% CRS versus 23% non-CRS group, p<0.001) (Table 1). Screening our BRR cohort for positive bacterial sputum cultures demonstrated that P aeruginosa was the most common pathogen (n=369; 27.3% of the total cohort), followed by S aureus (n=139; 10.3% of the total cohort) and H influenzae (n=98; 7.2% of the total cohort) (data not shown). Interestingly, the CRS group was more likely to have positive sputum culture for P aeruginosa (35% CRS versus 26% non-CRS group; p=0.007; Table 1), but not for H influenzae (6% CRS versus 7% non-CRS group; p=0.55) or S aureus (13% CRS versus 10% non-CRS group; p=0.21) (Table 1).
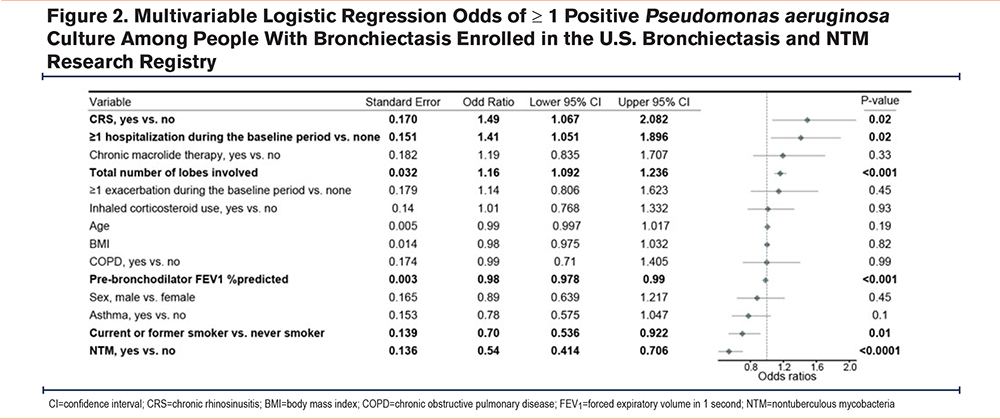
History of Chronic Rhinosinusitis Was Associated With Positive Sputum Culture for Pseudomonas aeruginosa in People With Bronchiectasis
To further investigate the clinical factors associated with P aeruginosa infection in people with bronchiectasis, we fit a multivariable logistic regression model using positive sputum culture as the dependent variable. This analysis revealed increased odds of P aeruginosa with CRS (odds ratio [OR]: 1.5; 95% confidence interval [CI]: 1.07 to 2.08) after adjusting for patient characteristics, including age, BMI, sex, smoking history, asthma, COPD, NTM, FEV1%pred, exacerbations, hospitalizations, total number of lobes involved, chronic macrolide treatment, and inhaled corticosteroid use (Figure 2). History of hospitalizations (OR: 1.4; 95% CI: 1.05 to 1.89), and total number of lobes involved (OR: 1.2; 95% CI: 1.09 to 1.24) were also independently and positively associated with positive sputum culture for P aeruginosa (Figure 2). In contrast, higher FEV1%pred (OR: 0.98; 95% CI: 0.98 to 0.99), current or former smoking status (OR: 0.7; 95% CI: 0.54 to 0.92), and history of NTM (OR: 0.5; 95% CI: 0.41 to 0.71) were negatively associated with the odds of a positive sputum culture for P aeruginosa (Figure 2).
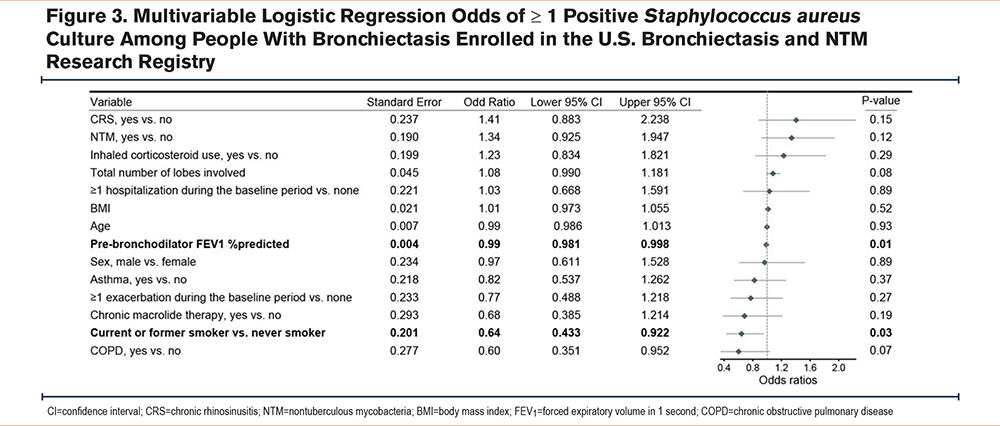
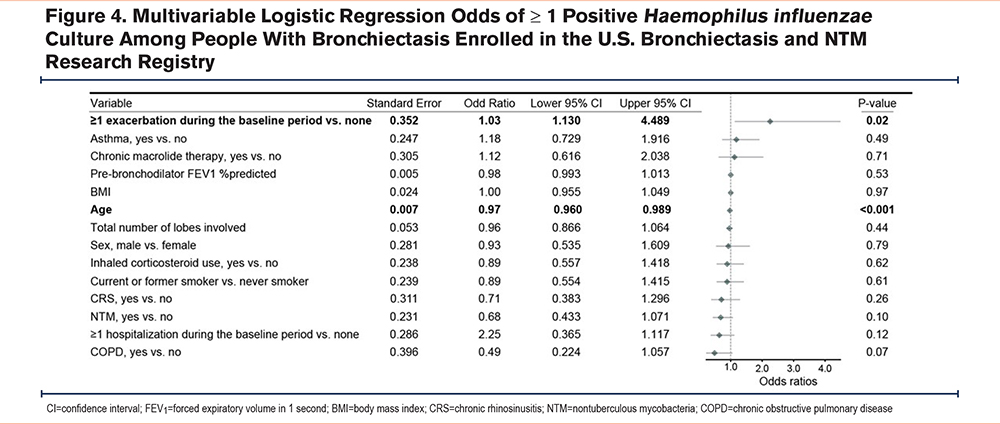
History of Chronic Rhinosinusitis Was Not Associated With Positive Sputum Culture for Staphylococcus aureus or Haemophilus influenzae
We fit similar multivariable logistic regression models, as described above, and found no significant association between CRS and positive sputum cultures for S aureus (OR: 1.4; 95% CI: 0.88–2.24; Figure 3) or H influenzae (OR: 0.7; 95% CI: 0.38–1.30; Figure 4) after adjusting for patient characteristics.
Discussion
Both CRS and bacterial airway infection are common in people with bronchiectasis, but whether CRS is associated with lower airway bacterial infection is not known.6,28 To address this knowledge gap, we used the BRR data to evaluate the association between history of CRS and the 3 most common bacterial pathogens cultured from sputum of people with bronchiectasis. While Staphylococci, H influenzae, and P aeruginosa are frequently cultured from the paranasal sinuses of patients with CRS,13,14,29,30 the only significant association we found between CRS and positive sputum culture was with P aeruginosa. This association persisted after adjusting for clinical and demographic characteristics.
A significant concordance between upper and lower airway bacterial cultures in people with bronchiectasis has been previously reported.15,31 In lung transplant recipients with CF, culturing P aeruginosa from the sinuses before transplantation correlated highly with the growth of the same pathogen in post-transplant bronchoalveolar-lavage fluid.32 Furthermore, in people with CF and occasional P aeruginosa in sputum, functional endoscopic sinus surgery helped clear the infection from sputum in 31 out of 50 patients.33 These observations suggest that one potential explanation for our observed association between P aeruginosa and CRS in people with bronchiectasis is that chronic sino-nasal inflammation allows the sinuses to act as a reservoir of infection, from which P aeruginosa can descend into the lower airways.
Another potential explanation for the association of CRS and lower respiratory tract infection with P aeruginosa may involve nitric oxide (NO). The paranasal sinuses are a major source and reservoir of NO in the human respiratory tract.34 In addition to its well described signaling and vasogenic properties, NO also plays a role in host defense.35 For example, it has been implicated in P aeruginosa biofilm dispersal.36 In CRS, however, nasal NO levels are reduced.37 Therefore, reduced nasal NO in CRS may promote biofilm formation and microbial persistence in the lower airways.38
While S aureus and H influenzae were the next most frequently cultured pathogens in the sputum of our BRR cohort, no significant association was observed between history of CRS and positive sputum culture for either pathogen. There are several possible explanations for this. Firstly, the association between CRS and lower airway infection may truly be specific to P aeruginosa. This microorganism possesses unique biologic characteristics making it highly suitable for infecting low-oxygen compartments such as poorly ventilated sinuses or mucous-plugged airways.39-43 Consistently, P aeruginosa has been shown to establish chronic lower airway infections more often than S aureus or H influenzae in people with bronchiectasis.9 Secondly, the number of participants with S aureus or H influenzae infections was relatively small (i.e., 28 and 14 out of 222 patients with CRS, respectively), limiting our power to detect the difference between the CRS-positive and -negative patient samples.
In addition to CRS, our study also found that higher frequency of bronchiectasis-related hospitalizations, a greater total number of lobes involved, and lower FEV1%pred were independently associated with positive sputum culture for P aeruginosa. These findings are in line with prior studies suggesting that P aeruginosa serves as a marker of more severe bronchiectasis44,45 and highlight the importance of P aeruginosa-focused clinical management in people with bronchiectasis.
Our study has several strengths, including the use of a large, well-characterized cohort of patients recruited from multiple clinical sites for enrollment in the BRR. Furthermore, clinical and laboratory characteristics were physician reported, improving data reliability.
Some limitations deserve mention. Reported prevalence of CRS among individuals with bronchiectasis varies widely across studies. Interestingly, larger registry-based studies, such as the Korean Multicenter Bronchiectasis Audit and Research Collaboration,5 as well as multicenter studies conducted across 4 European centers,46 have reported relatively low CRS prevalence estimates (7.1% and 13.1%, respectively). In contrast, smaller single-center studies have reported substantially higher prevalence estimates, with a pooled point estimate of 62% reported in a meta-analysis.6 These findings suggest that CRS may be underreported in large registry-based datasets or, alternatively, that smaller studies may be subject to selection bias that enriches for patients with CRS. Therefore, it is difficult to assess whether the 16% prevalence of CRS reported in our study population, based on physician-reported CRS history, is an accurate estimate. Despite this caveat, even if underdiagnosis or under-reporting were present, they were likely nondifferential which would consequently bias our estimates toward the null. We do acknowledge, however, that if for some reason CRS was preferentially under-reported in P aeruginosa-colonized patients, a resulting differential misclassification bias could exaggerate the observed association between CRS and P aeruginosa.
Another limitation is that incomplete data on history of CRS, sputum microbiology or clinical characteristics resulted in exclusion of a significant number of BRR participants, potentially introducing a selection bias. While we adjusted the model for several important variables, including individual markers of disease severity such as history of exacerbations, residual confounding cannot be excluded. The cross-sectional nature of this study did not permit the evaluation of a temporal relationship between CRS and sputum cultures.
Interpretation
Our study demonstrates an association between history of chronic rhinosinusitis and positive sputum culture for P aeruginosa, the most commonly isolated pathogen in people with bronchiectasis enrolled in the BRR. Further research is needed to determine the temporal relationship between CRS and bacterial colonization of the upper and lower airways, and whether treatment of CRS can reduce the burden of bacterial infection and clinical outcomes in people with bronchiectasis.
Acknowledgements
Author contributions: TG conceptualized the work. TG, AB, MM, and AG contributed to the design of the work. TG, AB, MM, and AG acquired, analyzed, and interpreted the data. TG drafted and edited the manuscript. AB, MM, and AG drafted the methods portion of the manuscript and edited the manuscript. All authors reviewed, edited, and approved the final version of the manuscript. TG accounted for the accuracy and integrity of the manuscript.
Ethics statement: The Bronchiectasis and NTM Research Registry is a centralized database of patients with bronchiectasis identified at 13 clinical sites throughout the United States. The institutional review board of each participating site approved the study, as did an administrative institutional review board for the data collecting center. For further information, please refer to: Aksamit TR, O’Donnell AE, Barker A, et al. Adult patients with bronchiectasis. Chest. 2017;151(5):982-992. doi: https://10.1016/j.chest.2016.10.055
Other acknowledgements: The authors would like to acknowledge the Bronchiectasis and NTM Association, who manages the Bronchiectasis and NTM Research Registry, a 501(c)(3) nonprofit organization. The authors would like to acknowledge that the Bronchiectasis and NTM Research Registry is supported in part by corporate donors, all of which can be found on the Supporters page of the Association website. It should also be noted that this work would not have been possible without the comprehensive chart reviews and recording of data by the dedicated research coordinators and PIs at each of the participating Bronchiectasis and NTM Research Registry sites.
Declaration of Interest
MLM has received consulting fees from Boehringer Ingelheim, Insmed Incorporated, Tactile Inc., and Zambon; served on data safety monitoring boards for AN2 Therapeutics, Renovion, and Verona; and received clinical trial support to the University of Connecticut from Insmed, Armata, and Sanofi.
GMS has received grant funding from the National Institutes of Health, the CF Foundation, Electromed, Insmed, Verona, Genentech, Sanofi, 4DMT, and Vertex; is a member of the advisory board at Electromed and Insmed; and consults for Electromed, Astra Zeneca, and Genentech.
NCL has been an educational speaker and has served on the advisory board of Insmed.
MKS has participated in the asthma registry with Astra Zeneca.
KW has received grant support and consulting fees from Insmed, Paratek, AN2, Renovion, Spero, and Mannkind.
TG, AB, KJ, and AG have no disclosures to report.
Role of sponsors: The statements and conclusions in this manuscript are solely the responsibility of the authors and do not necessarily represent the views of the U.S. Bronchiectasis and NTM Research Registry, its Board of Governors, or its Methodology Committee.