Running Head: Nebulizer Use in SPIROMICS Cohort
Funding Support: SPIROMICS was supported by contracts from the National Institutes of Health (NIH)/National Heart, Lung, and Blood Institute (NHLBI) (HHSN268200900013C, HHSN268200900014C, HHSN268200900015C, HHSN268200900016C, HHSN268200900017C, HHSN268200900018C, HHSN268200900019C, HHSN268200900020C), grants from the NIH/NHLBI (U01 HL137880, U24 HL141762, R01HL182622, and R01 HL144718), and supplemented by contributions made through the Foundation for the NIH and the COPD Foundation from Amgen; AstraZeneca/MedImmune; Bayer; Bellerophon Therapeutics; Boehringer-Ingelheim Pharmaceuticals, Inc.; Chiesi Farmaceutici S.p.A.; Forest Research Institute, Inc.; Genentech; GlaxoSmithKline; Grifols Therapeutics, Inc.; Ikaria, Inc.; MGC Diagnostics; Novartis Pharmaceuticals Corporation; Nycomed GmbH; Polarean; ProterixBio; Regeneron Pharmaceuticals, Inc.; Sanofi; Sunovion; Takeda Pharmaceutical Company; and Theravance Biopharma and Mylan/Viatris.
Dr. Buhr additionally received support from NIH/NCATS grant KL2TR001882 and NIH/NHLBI grant L30HL134025, related to this work, and the Solovy Award for Advancement in COPD from the Respiratory Health Foundation. Dr. Fazio additionally received support from NIH/NHLBI grant T32HL072752 and the UCLA Bruin Scholars Program. Dr. Gandhi additionally received support from NIH/NCATS grant 5KL2TR001870-05. The funders had no influence on the decision to publish nor the content of this manuscript.
Date of Acceptance: February 17, 2026 | Published Online Date: February 25, 2026
Abbreviations: 6MWD=6-minute walk distance; ADI=Area Deprivation Index; BMI=body mass index; CAT=COPD Assessment Test; CI=confidence interval; COPD=chronic obstructive pulmonary disease; DPIs=dry powder inhalers; FEV1=forced expiratory volume in 1 second; GOLD=Global initiative for chronic Obstructive Lung Disease; HR=hazard ratio; IC=inspiratory capacity; ICPH=interval-censored proportional hazards; MDIs=metered-dose inhalers; mMRC=modified Medical Research Council; OR=odds ratio; SD=standard deviation; SGRQ=St George’s Respiratory Questionnaire; SPIROMICS=SubPopulations and InteRmediate Outcome Measures In COPD Study; TEPS=tobacco-exposed persons with preserved spirometry; V=visit; VSAQ=Veterans Specific Activity Questionnaire
Citation: Fazio JC, Hong AW, Markovic D, et al. Exacerbations and decreased lung function predict nebulizer use and uptake in COPD and tobacco-exposed persons with preserved spirometry. Chronic Obstr Pulm Dis. 2026; 13(2): 111-124. doi: http://doi.org/10.15326/jcopdf.2025.0714
Online Supplemental Material: Read Online Supplemental Material (339KB)
Introduction
Inhaled pharmacotherapies are the staple of symptom management in chronic obstructive pulmonary disease (COPD) because they deliver a high concentration of drug directly to the lungs, reducing systemic side effects.1,2 The mainstay of inhaled treatments in COPD includes bronchodilators such as muscarinic antagonists, beta-sympathomimetic agonists, and inhaled corticosteroids for patients experiencing exacerbations and eosinophilia.3 These medications are delivered via either hand-held inhaler devices such as pressurized metered-dose inhalers (MDIs), dry powder inhalers (DPIs), or soft-mist inhalers, or via nebulizers.4
Nearly two-thirds of COPD patients use nebulizers at some point5; however, patterns and guidelines for nebulizer prescribing have not been adequately developed.6,7 Furthermore, one-third of inhaler users demonstrate poor technique with coordination, speed/depth of inhalation, and failure to fully exhale before inhalation or a postinhalation breath hold as the most common errors.8-11 Additional predictors of ineffective handheld inhaler use include older age, lower levels of education and socio-economic status, prescription of multiple devices, and inadequate peak inspiratory flow.6,12-14 Studies link incorrect inhaler technique to worse health-related quality of life and increased exacerbations in asthma and COPD.6 Nebulized delivery is a practical alternative for patients with poor respiratory effort, cognitive impairment, or impaired dexterity.15
Small observational studies have associated hypoxemia, increased exacerbation frequency, and worsening health status, as indicated by higher COPD Assessment Test (CAT) scores, with nebulizer use.5 Additionally, many tobacco-exposed persons with preserved spirometry (TEPS) report significant respiratory symptom burden and exacerbations, and have potential for disease progression.16,17 A randomized controlled trial of handheld bronchodilators among TEPS did not show benefit.18 However, randomized or longitudinal cohort studies have not evaluated nebulizer utilization in TEPS, which prompted their inclusion in our analysis. Additionally, assessment of the effectiveness of nebulizers versus handheld delivery devices are sparse in COPD and TEPS,19,20 yet observational studies suggest potential benefits in quality of life and symptom management in COPD with nebulized delivery.21,22 However, little is known about patterns of nebulizer uptake and utilization in relationship to clinical and epidemiologic features of patients with COPD.
Using a longitudinal cohort of individuals with spirometry-proven COPD and TEPS, we sought to understand the patterns of nebulizer use and identify and quantify factors associated with nebulizer uptake. We hypothesized that recent exacerbations, greater symptom burden, and more severe obstructive ventilatory defects independently predict baseline utilization and longitudinal uptake of nebulizers among TEPS and those with COPD. Although not a primary or secondary SubPopulations and InteRmediate Outcome Measures In COPD Study (SPIROMICS) objective, this analysis leverages rich longitudinal data to examine factors associated with nebulizer use among persons with and at risk for COPD, an area with limited prior longitudinal study.
Methods
Study Population
SPIROMICS is a multicenter prospective cohort study that enrolled TEPS and nonsmoking controls across 12 U.S. institutions.23 This analysis was conducted posthoc using data collected prospectively. Participants completed in-person physiologic and laboratory assessments at 4 approximately annual in-person visits (V1–V4) and a fifth visit (V5) 4.2±0.8 years (mean±standard deviation [SD]) after V4. We analyzed data from V1 to V5 from years 2012 to 2021. We restricted this analysis to participants with tobacco exposure (≥20 pack years) with or without COPD at study entry and without missing data on baseline nebulizer use (Figure 1). The research protocol for SPIROMICS was approved by the institutional review board of all participating institutions, and written informed consent was obtained from all participants.
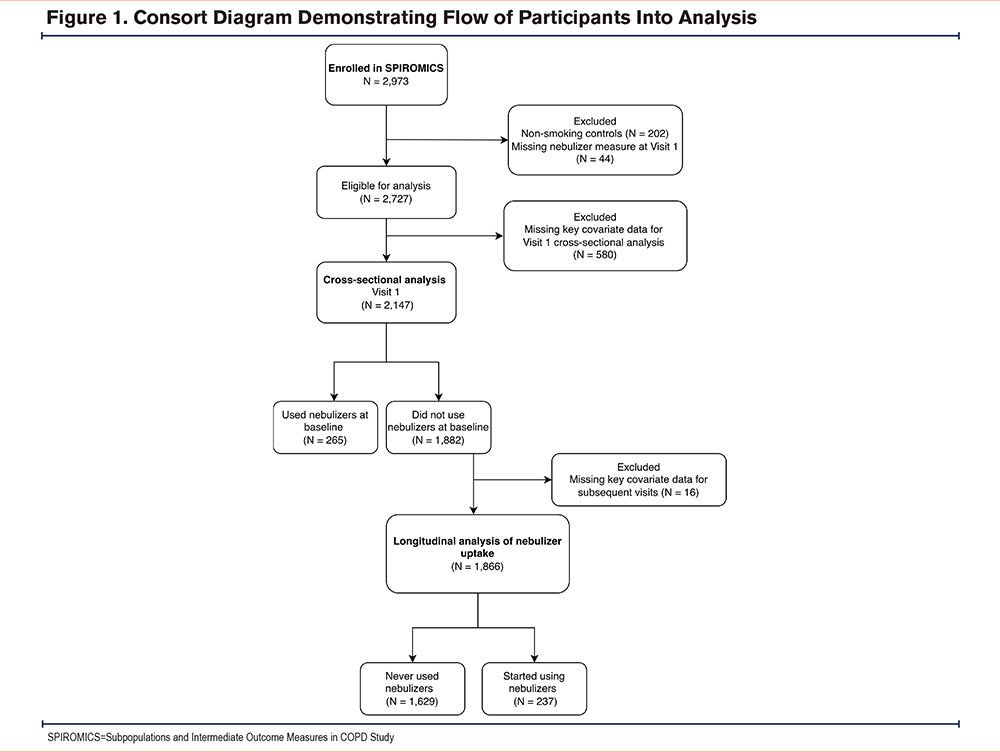
Outcome and Variable Definitions
We categorized participants into 2 outcome groups by baseline nebulizer use (yes/no). Nebulizer use was self-reported as any use of nebulizers within the last 3 months prior to study V1 through V4 (yes/no). At V5 the data collection instrument was changed to ask “do you regularly use nebulizers?,” (yes/no) and, if affirmative, frequency of use was collected (daily/as needed) (yes/no) (Supplemental Figure 1 in the online supplement). The V5 questionnaire also surveyed nebulized drug type, which was limited to approved drugs at the time of survey development.
We evaluated the following self-reported sociodemographic characteristics: age, sex, racial/ethnicity identity, household income, and tabulated Area Deprivation Index (ADI). The ADI is a neighborhood-level variable obtained by geocoding participants’ addresses to U.S. Census block groups and then matching them to the neighborhood-level ADI percentile, a composite measure of neighborhood disadvantage ranging from 1–100 (higher indicating more disadvantaged). ADI has been associated with worse outcomes in COPD patients.24-26
We assessed the following clinical features: body mass index (BMI), smoking status (current/former), history of asthma (yes/no), chronic bronchitis (yes/no, based on responses to the St George's Respiratory Questionnaire [SGRQ]),27 postbronchodilator forced expiratory volume in 1 second (FEV1),28 presence of COPD (defined by postbronchodilator FEV1 to forced vital capacity <0.70), and postbronchodilator inspiratory capacity (IC) derived from the slow vital capacity.
Exercise capacity was measured based on the 6-minute walk distance (6MWD)29 and the Veterans Specific Activity Questionnaire (VSAQ) (1–13, higher scores indicating greater self-reported exercise capacity).30 Respiratory symptom burden was assessed using: CAT score (0–40),31 modified Medical Research Council (mMRC) dyspnea score (0–4) with higher scores indicating greater symptom burden,32 and frequency of moderate to severe exacerbations (prescribed antibiotics or corticosteroids) in the 12 months preceding enrollment and between visit intervals. Respiratory-related quality of life was measured using the SGRQ.33 All data were collected at V1–V5, except exacerbations were reported during quarterly phone calls between each visit.
Statistical Analysis
To assess differences in sociodemographic and clinical characteristics between nebulizer use groups at baseline, we first performed a cross-sectional analysis at V1. Continuous variables were compared using the Wilcoxon rank-sum test and categorical variables were compared using Pearson's chi-squared test. We then examined the type of nebulized medications used between mutually exclusive categories of daily maintenance versus as-needed use (data only available for V5), using Fisher’s exact test due to low cell counts.
We tested associations between participant factors and baseline nebulizer use with a cross-sectional multivariable logistic regression comparing baseline nebulizer users to nonusers, controlling for age, sex, race, ADI, BMI, COPD diagnosis at study entry, enrollment smoking status, history of asthma, chronic bronchitis, absolute FEV1 (liters), number of exacerbations during the year prior to enrollment, CAT score, and 6MWD (meters). We used a conceptual framework based on existing literature to determine the above listed covariates of interest, and further refined the model using tests of collinearity.
To evaluate factors associated with the uptake of nebulizers during the study period, we performed a longitudinal subgroup analysis of all those who did not use nebulizers at or within 3 months of baseline. We compared those who initiated nebulizer use during the study following V1 to those who never used nebulizers using time-to-event analysis via an interval-censored proportional hazards (ICPH) model controlling for the same covariates analyzed in the baseline logistic regression model, detailed above.34,35 Because event times were interval-censored, primary time-to-event analyses were conducted using ICPH models. Kaplan-Meier curves were included for descriptive visualization and were constructed by assigning events to the visit date at which they were first observed. These curves do not account for interval censoring and should be interpreted as approximate summaries of observed event patterns rather than formal survival estimates. Time invariant variables included race, ADI, income, COPD status, and history of asthma. Time-varying variables included current smoking status, chronic bronchitis using SGRQ criteria, CAT score, FEV1, 6MWD, and exacerbation frequency. Due to collinearity, age was modeled as time-invariant using V1 age rather than time-varying. The outcome of interest was time to initiation of nebulizer use at a subsequent visit. Subgroup analyses were also independently performed on the TEPS and COPD groups.
All analyses were completed using Stata version 18.0 (StataCorp; College Station, Texas) using a 2-tailed alpha of 0.05. Missing values for ADI were imputed using the mean ADI for that study site and accounted for 2.9% of the data. Complete case analysis was used for all statistical models.
Results
Characteristics of Participants at Study Enrollment
Of the 2147 participants included in the baseline cross-sectional analysis (Figure 1), 33.3% (717) were TEPS and 66.6% (1430) had COPD. At baseline, 12% (265) reported nebulizer use (5% of TEPS [35/717] compared to 16.1% of COPD participants [230/1,430]) (Supplemental Table 1 in the online supplement). Nearly half of those in both Global initiative for chronic Obstructive Lung Disease (GOLD)36 Stage 4 (42%,[48/115]) and formerly GOLD Group D (49%,[56/115]) (noting that a subsequent guideline change collapsed groups C and D into group E) reported nebulizer use at baseline. Nebulizer users were slightly younger and more often female (Table 1). Nebulizer users also had significantly higher rates of overall neighborhood level disadvantage by ADI (45.2±28.7 versus 36.8 ±27.7 percentile, mean±SD, P<0.001)[. Additionally, household incomes were more often <$15,000 among nebulizer users (31% ]81/265[ versus 18% ]332/1882[, P<0.001). Nebulizer users were also more likely to identify as Black (27% versus 17%, P<0.001).
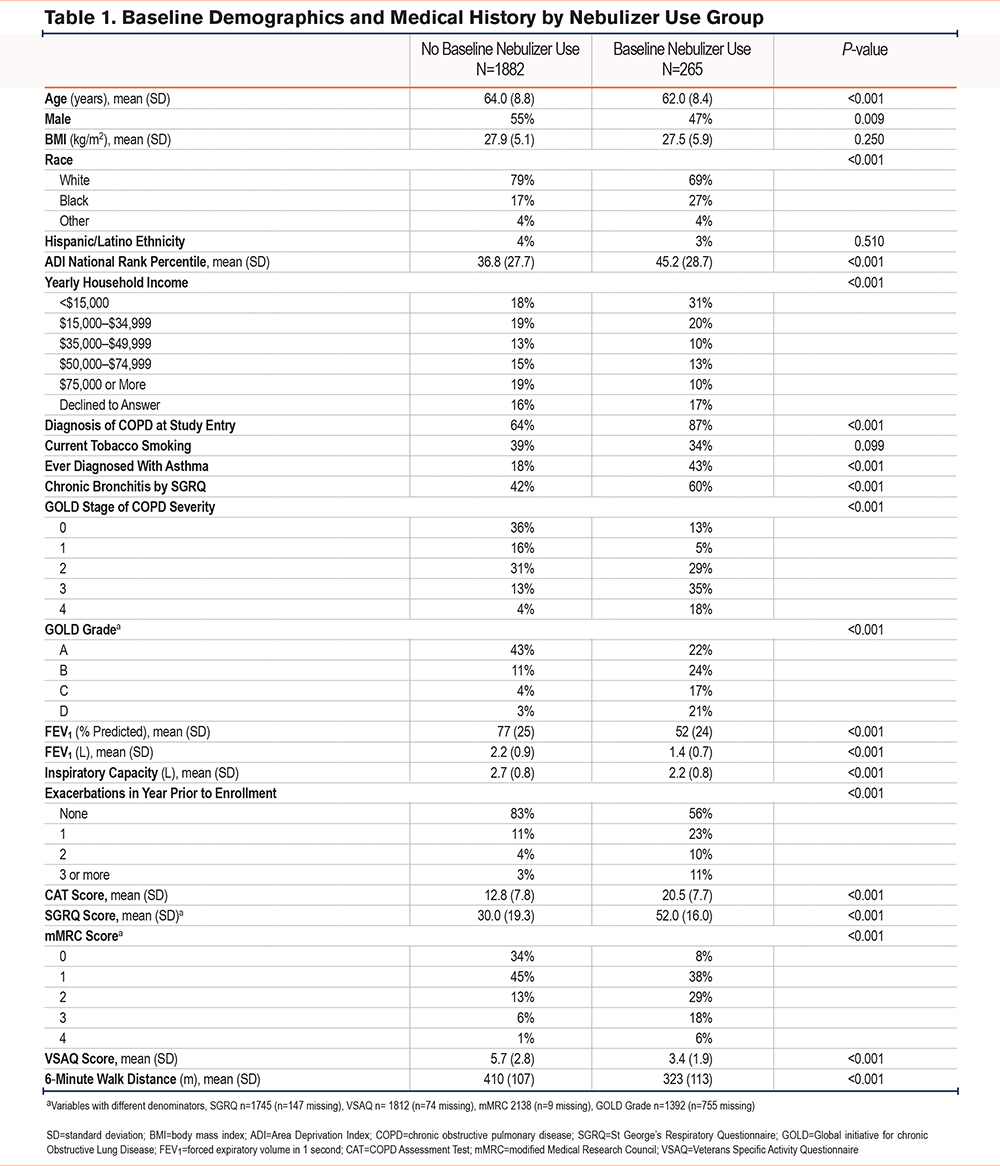
Nebulizer users more often reported a diagnosis of COPD (87% ]230/265[ versus 64% 1200/1882], P<0.001) or asthma (43% [114/265] versus 18% [333/1882], P<0.001) at baseline compared to nonusers. They also more frequently had chronic bronchitis (60% [158/265] versus 42% [792/1882], P<0.001), but the groups did not differ in current smoking status. Baseline pulmonary function was lower among nebulizer users (Table 1), both by postbronchodilator FEV1 (1.4±0.7L versus 2.1±0.9L, P<0.001) and inspiratory capacity (IC, 2.2±0.8L versus 2.7±0.8L, P<0.001). More severe symptoms by CAT and SGRQ and poorer exercise capacity by VSAQ and 6MWD were seen among nebulizer users (Table 1). Nebulizer users were also more likely to have more frequent exacerbations within the year before enrollment, (1, 23% [62/265] versus 11% [198/1882]; 2, 10% [27/265] versus 3% [69/1882]; or 3 or more, 11% [29/265] versus 3% [55/1882] compared to no exacerbations; P<0.001).
Among TEPS who used nebulizers, most were symptomatic by CAT, SGRQ and mMRC, 60% [21/35] had a history of asthma and 77% [27/35] and chronic bronchitis. Apart from lung function measures, characteristics of nebulizer users among participants with COPD and TEPS were similar (Supplemental Table 1 in the online supplement).
Prediction of Nebulizer Use at Study Enrollment
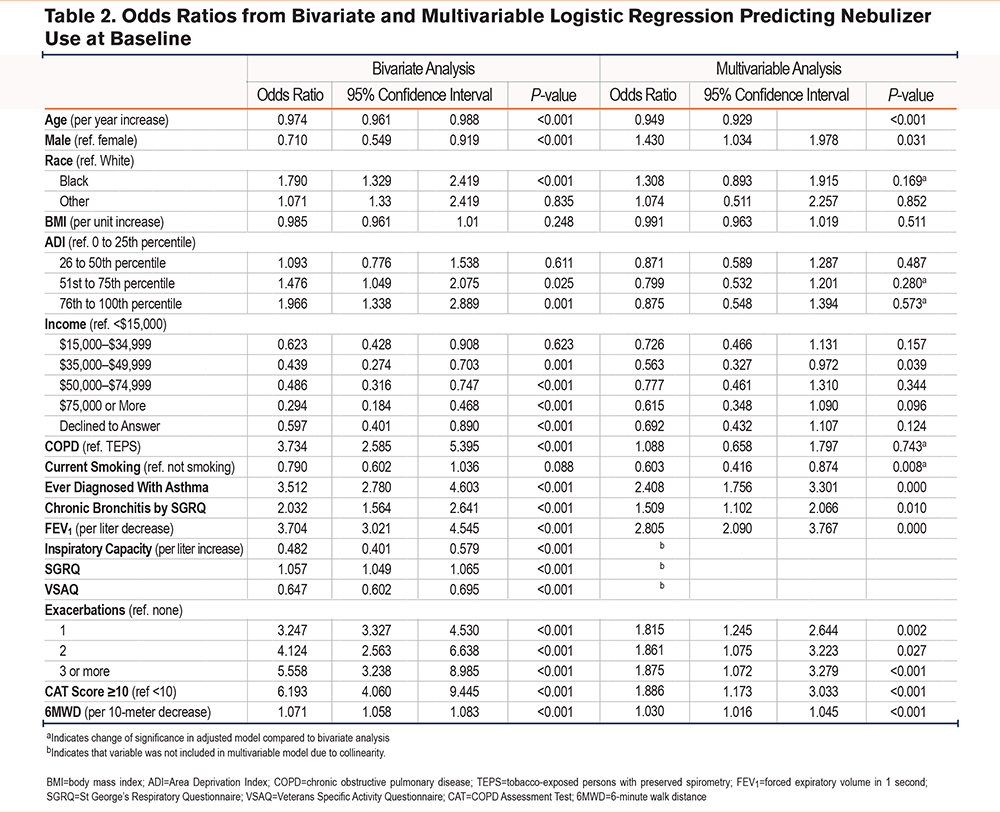
In the cross-sectional multivariable logistic regression analysis (Table 2 for both bivariate and adjusted estimates), significant factors associated with baseline nebulizer use included history of asthma (OR 2.41, 95%CI [1.76, 3.30]), more moderate to severe COPD exacerbations within the last year (1, OR 1.81, 95%CI [1.24, 2.64]; 2, OR 1.86, 95%CI [1.07, 3.22]; 3 or more, OR 1.87, 95%CI [1.07, 3.28]), FEV1 absolute (OR 2.81 per liter lower baseline FEV1, 95%CI [2.09, 3.77]), a CAT score ≥10 (OR 1.89, 95%CI [1.17, 3.03]), 6MWD distance (OR 1.03 per 10 meters lesser baseline 6MWD, 95%CI [1.02, 1.05]). These findings were robust to sensitivity analysis, with few covariates changing sign or statistical significance, and none had both a sign and significance change (Supplemental Table 2 in the online supplement). SGRQ, VSAQ, and IC were all dropped from the model due to collinearity with CAT or FEV1 (pearson correlation coefficient >0.7, P<0.001).
Nebulizer Uptake During Study Follow-Up
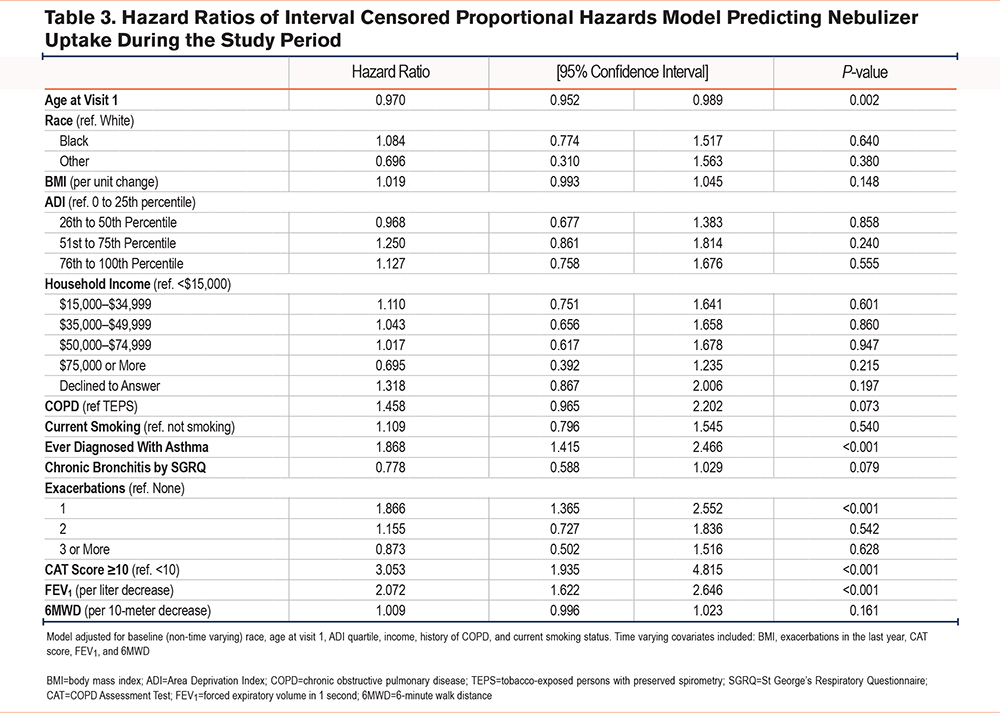
Of the 1866 participants who indicated no nebulizer use at baseline, 237 subsequently reported uptake at a later visit. In the ICPH model, nebulizer uptake was significantly associated with: a history of asthma (hazard rato [HR] 1.87, 95%CI [1.42, 2.47]); at least one COPD exacerbation in the year prior to any postbaseline study visit (HR 1.87, 95%CI [1.37, 2.55]); a CAT score ≥10 (HR 3.05, 95%CI [1.94, 4.82]); and per liter decrease in FEV1 (HR 2.07, 95%CI [1.62, 2.64]) (Table 3). These findings were robust to independent sensitivity analyses in TEPS and COPD participants (Supplemental Table 3 in the online supplement).
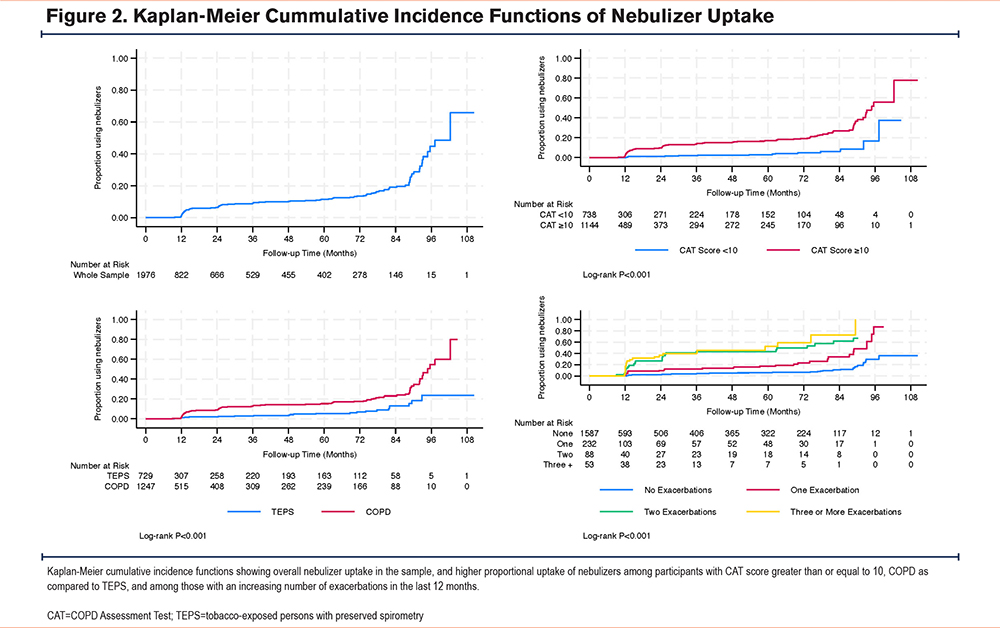
The overall median time to nebulizer uptake was 102 months among those not using nebulizers at study entry (Figure 2). The Kaplan-Meier cumulative incidence function (Figure 2) shows a significantly earlier initiation of nebulizers among those with more exacerbations, a CAT score ≥10, and among COPD versus TEPS (log-rank P<0.001 for all). For those not using nebulizers at V1, the median time to nebulizer uptake varied by exacerbations (93, 85, or 74 months for 1, 2, or 3 or more exacerbations, respectively). Among those without exacerbations in the year prior to V1, 37% had started using nebulizers by V5. Among those not using nebulizers at V1 with a CAT score <10, 26% had started using nebulizers by V5, whereas among those with a CAT score ≥10 at V1, the median time to nebulizer uptake was 96 months. Among TEPS not using nebulizers at V1, 31% had started using nebulizers by V5, and among participants with COPD not using nebulizers at V1 the median time to nebulizer use was 97 months.
Description of Nebulized Medication Type
Of the 17.5% (194/1136) participants who reported nebulizer use at V5, 73% (142/194) indicated only as-needed use (Supplemental Table 4 in the online supplement). A total of 87% (170/194) used short-acting bronchodilators, and 3% (6/194) used long-acting bronchodilators or corticosteroids. Use of long-acting bronchodilators or other nebulized treatments, including hypertonic saline and “other” not defined, accounted for 7% (14/194). Additionally, among baseline nebulizer users, 92% used bronchodilators and 71% used inhaled steroids in the prior 3 months (Supplemental Table 4 in the online supplement).
Discussion
This posthoc analysis of a multicenter, prospective, observational cohort study reports the prevalence and patterns of nebulized drug utilization in individuals with smoking histories with or without spirometrically defined COPD. This study adds to the existing literature by evaluating nebulizer uptake over time in both COPD and TEPS populations. In contrast to prior large studies of nebulizer use, which have primarily been cross-sectional, our longitudinal design allowed for assessment of changes in utilization patterns and provides insight into real-world prescribing beyond single time-point estimates. Our data reveal that the factors most strongly associated with nebulizer use at baseline were increased frequency of respiratory exacerbations, decreased exercise capacity, low lung function, and greater respiratory symptom burden among TEPS and patients with COPD. In longitudinal analysis, moderate to severe respiratory exacerbations in the year before each visit had the largest effect size on new nebulizer use, and those with more frequent exacerbations showed earlier uptake. Similar to the cross-sectional analysis, greater symptom burden was associated with uptake, however, decreased exercise capacity did not remain an independent predictor in the longidutinal analysis. Overall, these results support prior studies and should motivate specific investigation into the efficacy of nebulizer use in the management of both COPD and TEPS.
Among participants with advanced COPD, defined as either GOLD spirometric Stage 4 or GOLD Group D, nearly half used nebulizers. Although current professional society guidelines do not preferentially recommend nebulizers to manage chronic COPD symptoms, efficacy is generally considered similar to handheld devices.13,36,37 This assumption is influenced by similar short-term improvement in lung function in comparisons of hand-held inhalers versus nebulizers in patients who demonstrated proper technique of inhaler use.19
Whether significant clinical benefits were derived in our cohort from nebulizer use is outside the scope of this analysis. Nevertheless, according to previous studies, COPD patients with severe disease burden may perceive greater symptom relief from nebulized medications, due to their ease of use relative to hand-held inhalers (i.e., minimal need for breath coordination or optimal inspiratory effort).5,38-41 Asthma patients similarly show increased nebulizer use with severe symptoms.42 Additionally, clinicians may hold similar beliefs and experience difficulty teaching proper inhaler technique, especially in those with advanced COPD, older age, and impaired cognition.38 Both patient and clinician factors likely influence nebulizer utilization. Furthermore, our study highlights the association between nebulizer use and exacerbations, which often lead to emergency department visits and hospitalizations where patients may receive nebulized therapies in the hospital and upon discharge.43,44
Additionally, we made the novel finding that a substantial number of TEPS reported nebulizer use, a finding associated with respiratory exacerbations in the longitudinal analysis. TEPS represent a distinct subset, analogous to COPD Stage 0 or “pre-COPD,” and are at an elevated risk of disease progression.45 An investigation conducted by McKleroy et al in the SPIROMICS cohort found that while rates of FEV1 decline or incidence of COPD were not increased when comparing symptomatic to asymptomatic TEPS, symptomatic TEPS had significantly more exacerbations.17 Nevertheless, the use of maintenance inhaled bronchodilators may not significantly improve symptom control in TEPS.18 However, we showed that nebulizer uptake among TEPS was associated with greater respiratory symptom burden. Our findings are noteworthy in showing that real-world nebulizer utilization is not always associated with advanced COPD or age; but that it does represent a relevant drug delivery option even among those with early disease; as technology continues to evolve, patterns of usage may also change.46
Participants with COPD and TEPS predominantly used nebulizers to deliver short-acting bronchodilators. This suggests use during acute dyspnea events, akin to nebulizer utilization for acute exacerbations in hospital settings.3 By contrast, few used nebulized long-acting bronchodilators or corticosteroids. This finding is unsurprising, as the landscape of nebulized medications is evolving rapidly, with only recent approval of nebulized long-acting muscarinic antagonists, (glycopyrrolate and revefenacin), and the phosphodiesterase-3/-4 inhibitor ensifentrine.3,47 These marked a significant milestone in novel treatment options for advanced COPD. Future studies should evaluate the benefits of nebulized long-acting therapies to optimize outcomes in COPD.
Another novel finding is that nebulizer users were on average younger than nonusers (62 versus 64 years), although after adjusting for other factors in cross-sectional multivariable analysis, age was not a significant predictor. This finding contrasts with studies showing more frequent nebulizer use among older COPD patients.48 Other previously demonstrated associations were with lower FEV1 percentage and with greater air trapping, which causes lower IC, but these have not been rigorously analyzed in multivariable models.48 Reduced IC is linked to poor peak inspiratory flow rates, a known factor limiting the efficacy of MDIs and DPIs.48 Given this, we can hypothesize that reduced IC may drive nebulizer use. However, more investigation into the patterns and efficacy of nebulizer use would clarify this relationship. In agreement, nebulizers users in our study on average had a statistically significantly lower IC, and in bivariate analysis, lower IC was predictive of nebulizer use.48 However, we were unable to evaluate IC in the multivariable model due to collinearity. Future studies using plethysmography to measure subdivisions of lung volume are needed to explore the roles of air trapping and hyperinflation.
Socioeconomic and racial disparities were apparent in the baseline unadjusted analyses but attenuated in multivariable models, suggesting that clinical disease characteristics may underlie the observed demographic variation. The association between socioeconomic disadvantage and nebulizer use is consistent with prior findings linking lower socioeconomic status and education attainment to worse COPD symptom control and outcomes, as well as poorer inhaler technique and adherence.21,26,49,50 We found more frequent nebulizer use among Black participants, those with lower incomes, and those with greater neighborhood disadvantage. One potential explanation is the relatively low cost of nebulized short-acting bronchodilators relative to MDIs (approximately 10 times greater cost per dose for albuterol-ipratropium).22 Future investigation should assess the effect of health insurance on nebulizer use, which was out of the scope of this analysis. Additionally, higher odds of nebulizer use among women suggest sex-specific differences in disease presentation or health care utilization that merit further investigation.
Limitations and Strengths
Our study has several limitations. First, as an observational nonrandomized study, we cannot assume causality of the demonstrated associations. Second, we recruited participants from large academic medical centers, which may impact generalizability. Third, the frequency of nebulizer use and the distinction between maintenance versus rescue medications were collected only at V5. Although V5 was included in the time-to-event analysis to preserve longitudinal follow-up, the altered exposure definition at this visit may introduce misclassification and should be considered when interpreting hazard estimates. Fourth, nebulizer use was self-reported, potentially introducing recall bias. Fifth, the time-to-event analysis ignored participants whose nebulizer use varied from between visits, as we censored participants at the first visit reporting nebulizer use; this approach misses granularity in subsequently variable nebulizer usage. Sixth, because few participants used long-acting nebulized medications, our results primarily represent users of short-acting nebulized medications and should be interpreted in this context. Seventh, because the milieu of both long-acting hand-held and nebulized therapies and the GOLD treatment guidelines evolved substantially over the course of the study period, we did not control for concurrent handheld inhaled medication use in our analyses, as they would have complicated our models substantially. However, strengths of our design include its longitudinal, prospective nature, which, by taking into account time-variant characteristics of disease progression, identified temporal patterns in nebulizer uptake. Additionally, our use of an interval-censored modeling approach accounts for uncertainty in the exact time between study visits at which nebulizer use began. This approach provides highly reliable estimates for hypothesis generation to design future trials.
Conclusion
Nebulized medication delivery systems are widely used among SPIROMICS participants with smoking histories, regardless of the presence of spirometrically defined COPD. Nearly half of individuals in GOLD Group D reported nebulizer use at study enrollment, and many participants in all GOLD groups initiated nebulizer use over the study period, yet long-acting maintenance therapies were infrequently utilized. Nebulizer use was associated with previous respiratory exacerbations, symptoms, and diminished exercise capacity. Although our observational study design precludes assessment of causality, these findings argue for randomized controlled trials to compare directly the efficacy of nebulized versus hand-held devices, particularly in patients with advanced COPD with high symptom burden and frequent exacerbations.
Acknowledgements
Author contributions: All authors contributed to enrolling participants, conducting study procedures, and generating data for analysis. JCF, DM, RGBuhr, and IB conceived the design of the analysis. JCF conducted the analyses with guidance from DM and RGBuhr and attests to their validity. JCF, AH, RGBuhr, DPT, and IB drafted the manuscript with critical editing from all co-authors. All coauthors reviewed the final manuscript and approved it for publication.
Other Acknowledgements: The authors thank the SPIROMICS participants and participating physicians, investigators, study coordinators, and staff for making this research possible. More information about the study and how to access SPIROMICS data is available at www.spiromics.org. The authors would like to acknowledge the University of North Carolina at Chapel Hill BioSpecimen Processing Facility (http://bsp.web.unc.edu/) and Alexis Lab (https://www.med.unc.edu/cemalb/facultyresearch/alexislab/) for sample processing, storage, and sample disbursements.
We would like to acknowledge the following current and former investigators of the SPIROMICS sites and reading centers: Neil E. Alexis, MD; Wayne H. Anderson, PhD; Mehrdad Arjomandi, MD; Igor Barjaktarevic, MD, PhD; R. Graham Barr, MD, DrPH; Patricia Basta, PhD; Lori A. Bateman, MS; Christina Bellinger, MD; Surya P. Bhatt, MD; Eugene R. Bleecker, MD; Richard C. Boucher, MD; Russell P. Bowler, MD, PhD; Russell G. Buhr, MD, PhD; Stephanie A. Christenson, MD; Alejandro P. Comellas, MD; Christopher B. Cooper, MD, PhD; David J. Couper, PhD; Gerard J. Criner, MD; Ronald G. Crystal, MD; Jeffrey L. Curtis, MD; Claire M. Doerschuk, MD; Mark T. Dransfield, MD; M. Bradley Drummond, MD; Christine M. Freeman, PhD; Craig Galban, PhD; Katherine Gershner, DO; MeiLan K. Han, MD, MS; Nadia N. Hansel, MD, MPH; Annette T. Hastie, PhD; Eric A. Hoffman, PhD; Yvonne J. Huang, MD; Robert J. Kaner, MD; Richard E. Kanner, MD; Mehmet Kesimer, PhD; Eric C. Kleerup, MD; Jerry A. Krishnan, MD, PhD; Wassim W. Labaki, MD; Lisa M. LaVange, PhD; Stephen C. Lazarus, MD; Fernando J. Martinez, MD, MS; Merry-Lynn McDonald, PhD; Deborah A. Meyers, PhD; Wendy C. Moore, MD; John D. Newell Jr, MD; Elizabeth C. Oelsner, MD, MPH; Jill Ohar, MD; Wanda K. O’Neal, PhD; Victor E. Ortega, MD, PhD; Robert Paine, III, MD; Laura Paulin, MD, MHS; Stephen P. Peters, MD, PhD; Cheryl Pirozzi, MD; Nirupama Putcha, MD, MHS; Sanjeev Raman, MBBS, MD; Stephen I. Rennard, MD; Donald P. Tashkin, MD; J. Michael Wells, MD; Robert A. Wise, MD; and Prescott G. Woodruff, MD, MPH. The project officers from the Lung Division of the National Heart, Lung, and Blood Institute were Lisa Postow, PhD, and Lisa Viviano, BSN;
Declaration of Interest
Dr. Barr received grant funding from the National Institutes of Health (NIH), the Foundation for the NIH, the COPD Foundation and the American Lung Association. Dr. Drummond reports grants paid to his institution from the NIH-National Heart, Lung, and Blood Institute (NHLBI) related to his work, reports consulting fees from AstraZeneca, Verona, GSK, and Sanofi unrelated to this work, and is an associate editor for the Journal of the COPD Foundation (in this role he was not involved in editorial decisions related to this manuscript during peer review). Dr. Jeffrey Curtis reports research support from the COPD Foundation to his institution, personal fees from Genentech, and payment for his role as associate editor for the American Thoracic Society. Dr. Christopher Cooper reports grants from the NIH/NHLBI, the NIH Foundation, and the COPD Foundation, during the conduct of the study; he also reports personal fees from AstraZeneca, GSK, Chiesi, NUVAIRA, MGC Diagnostics, Horizon Therapeutics, Respiree, Herbalife, Verona, RS BioTherapeutics, Genentech, and Cambridge University Press, outside the submitted work. Dr. Couper reports grants and contracts to his institution from the NHLBI and the COPD Foundation. Dr. Curtis reports grants from the NIH/NHLBI and the COPD Foundation for the submitted work; and grants from the NHLBI, the Department of Veterans Affairs, and the Department of Defense, and personal fees from the European Respiratory Society and the American Thoracic Society, outside the scope of this project. Dr. Fortis has received grants from the Office of Rural Health, served as a consultant for the Society of Hospital Medicine (SHM), and has stock options with ROMTech. Dr. Meilan Han reports receiving consulting fees from AstraZeneca, Boehringer Ingelheim, GSK, Novartis, Pulmonx, Teva, Verona, Merck, Mylan, Sanofi, DevPro, Aerogen, Polarian, Regeneron, Altesa, BioPharma, Amgen, Roche, RS Biotherapeutics, Apreo Health, Genentech, Owkin, Zymeworks, Bristol Myers, and Squibb. Dr. Martinez has been a member of COPD study steering committees for work sponsored by AstraZeneca, Chiesi, DevPro, GSK, and Sanori/Regenoron. He has advised on COPD-related work for AstraZeneca, Chiesi, DevPro, GSK, and Sanofi/Regeneron. He has received travel support but no honoraria. Dr. Jill Ohar has received grant funding from Chiesi, Viatris, and the COPD Foundation and personal funds from Theravance, Astra Zeneca, Chiesi, Aerogen, and Viatris. Dr. Ortega declares receiving compensation from Regeneron and Sanofi for work related to an independent data monitoring committee, unrelated to present work. Dr. Robert Paine reports receiving grant support for this work from the NHLBI and the COPD Foundation, support for unrelated work from the Department of Veterans Affairs, the NHLBI, and Partner Therapeutics. Dr. Putcha has no relevant interests to the work. She is an associate editor for the Journal of the COPD Foundation (in this role she was not involved in editorial decisions related to this manuscript during peer review). Outside of the work she has participated in advisory boards for Sanofi, Astra Zeneca, and Verona (Merck). Dr. Wells reports receiving grants to his institution from the NIH supporting the current work and grants and contracts from the Department of Veterans Affairs, the Alpha-1 Foundation, the American Lung Association, Verona Pharma, and Sanofi for projects unrelated to the current work. Dr. Wells has received personal payments for work on advisory boards from Verona and Sanofi for work outside the scope of the manuscript. Dr. Woodruff received funding for SPIROMICS from the NIH and the COPD Foundation and has consulted outside the present work for AnaptysBio, Abbvie, and Roche. Dr. Buhr received grants from the NHLBI, National Center for Advancing Translational Sciences, the COPD Foundation, and the NIH Foundation related to this work, and from the VA Office of Research and Development outside the submitted work; he received personal consulting fees from Chiesi and Optum outside the submitted work; he is an employee of the Department of Veterans Affairs. The statements and positions in this manuscript do not necessarily reflect those of the U.S. Government. Dr. Barjaktarevic reports consulting fees from Astra Zeneca, Theravance, Viatris, Verona Pharma, Merck, Insmed, Takeda, Grifols, Aerogen, Sanofi and Regeneron, and Genentech. Drs. Fazio, Hong, Bleecker, Bowler, Gandhi, and Kim and Ms. Marcovic have nothing to disclose.
Disclaimers: Drs. Buhr, Curtis, Fortis, and Paine are employees of the Veterans Health Administration. The findings and positions in this manuscript do not necessarily reflect those of the Department of Veterans Affairs or the United States Government.