Running Head: COVID-19 Pandemic Impact on COPD Patients
Funding support: This research was funded by grants from the American Lung Association and the National Institutes of Health-National Heart, Lung and Blood Institute (U01HL128951 and U01HL128954). Clinical Trial Registration: NCT00720226
Date of Acceptance: August 17, 2022 | Published Online Date: August 23, 2022
Abbreviations: chronic obstructive pulmonary diseases, COPD;coronavirus disease 2019, COVID-19; Losartan Effects on Emphysema Progression, LEEP; forced expiratory volume in 1 second, FEV1; high-resolution computed tomography, HRCT; Airways Clinical Research Centers, ACRC; bronchodilator, BD; forced vital capacity, FVC; COPD Assessment Test, CAT; Generalized Anxiety Disorder-7 questionnaire, GAD-7; Patient Health Questionnaire, PHQ-8; Patient-Reported Outcome Measurement Information System, PROMIS; National Institutes of Health, NIH; interquartile range, IQR
Citation: Zhang WZ, LaBedz SL, Holbrook JT, et al; the American Lung Association Airways Clinical Research Centers. Impact of the coronavirus disease 2019 pandemic on physical and mental health of patients with COPD: results from a longitudinal cohort study conducted in the United States (2020-2021). Chronic Obstr Pulm Dis. 2022; 9(4): 510-519. doi: http://doi.org/10.15326/jcopdf.2022.0287
Online Supplemental Material: Read Online Supplemental Material (629KB)
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has had a devastating impact on health worldwide, resulting in excess morbidity and mortality, especially among individuals with comorbidities.1 Patients with chronic obstructive pulmonary disease (COPD) are at increased risk for more severe COVID-19 infection, including an elevated risk of intensive care unit admission, mechanical ventilation, and death2,3 from COVID-19. Additionally, there is concern that social restrictions and heightened awareness of health risks may lead to increased rates of depression or anxiety, both of which are associated with poor medication adherence,4 morbidity, and mortality in people with COPD.5-10 There are conflicting reports about the effects of the pandemic on those with chronic respiratory disease, in particular COPD. While some studies report negative consequences from social restrictions and lockdown,11-14 others describe a remarkable resilience in these high-risk populations.15,16
Quantitative information on the effects of the pandemic on COPD patients is required to determine the needs of this vulnerable population now and to prepare for public health emergencies in the future. In this study, we examined the impact of the COVID-19 pandemic on patients with moderate to severe COPD living in the United States from June 2020 to April 2021. Specifically, we examined their physical and mental health, their knowledge and beliefs about COVID-19, and their risk-reduction behaviors to better understand how patients were impacted by and coped with the pandemic during its first year.
Methods
Participants
This study was conducted as an ancillary study to the Losartan Effects on Emphysema Progression (LEEP) trial (NCT02696564), a randomized clinical trial designed to evaluate the effect of losartan on the progression of the emphysema based on high-resolution computed tomography (HRCT).17,18 LEEP was conducted by the American Lung Association Airways Clinical Research Centers (ACRC) in collaboration with the Pulmonary Trials Cooperative. Ongoing and completed participants were invited to participate between May 2020 and November 2020. LEEP eligibility criteria included post-bronchodilator (BD) forced expiratory volume in 1 second (FEV1) 20%-80% predicted, forced vital capacity (FVC) ratio ≤0.70, age 40 years or older, a history of 10 pack years or greater of cigarette smoking, and an HRCT scan with between 5% and 35% of voxels with density <-950 Hounsfield units. Twenty-one of the 26 ACRC sites participated in the ancillary study. All sites participating in the ancillary study received local institutional review board approval before contacting participants and all ancillary study participants provided informed consent.
At the genesis of this ancillary study in March 2020, the objective was to evaluate whether current treatment with losartan versus placebo affected COVID-19 infection rates in the participants under active follow-up in the LEEP trial. Secondary objectives were to evaluate overall infection rates and to describe effects of the pandemic on physical and mental health status, and social support networks in the entire group and perform exploratory analysis for potential risk modifiers in well-characterized COPD patients. Given the few cases of COVID-19 observed, our primary and some of our secondary objectives were not achievable. A protocol for the study is posted in the online supplement.
Data Collection
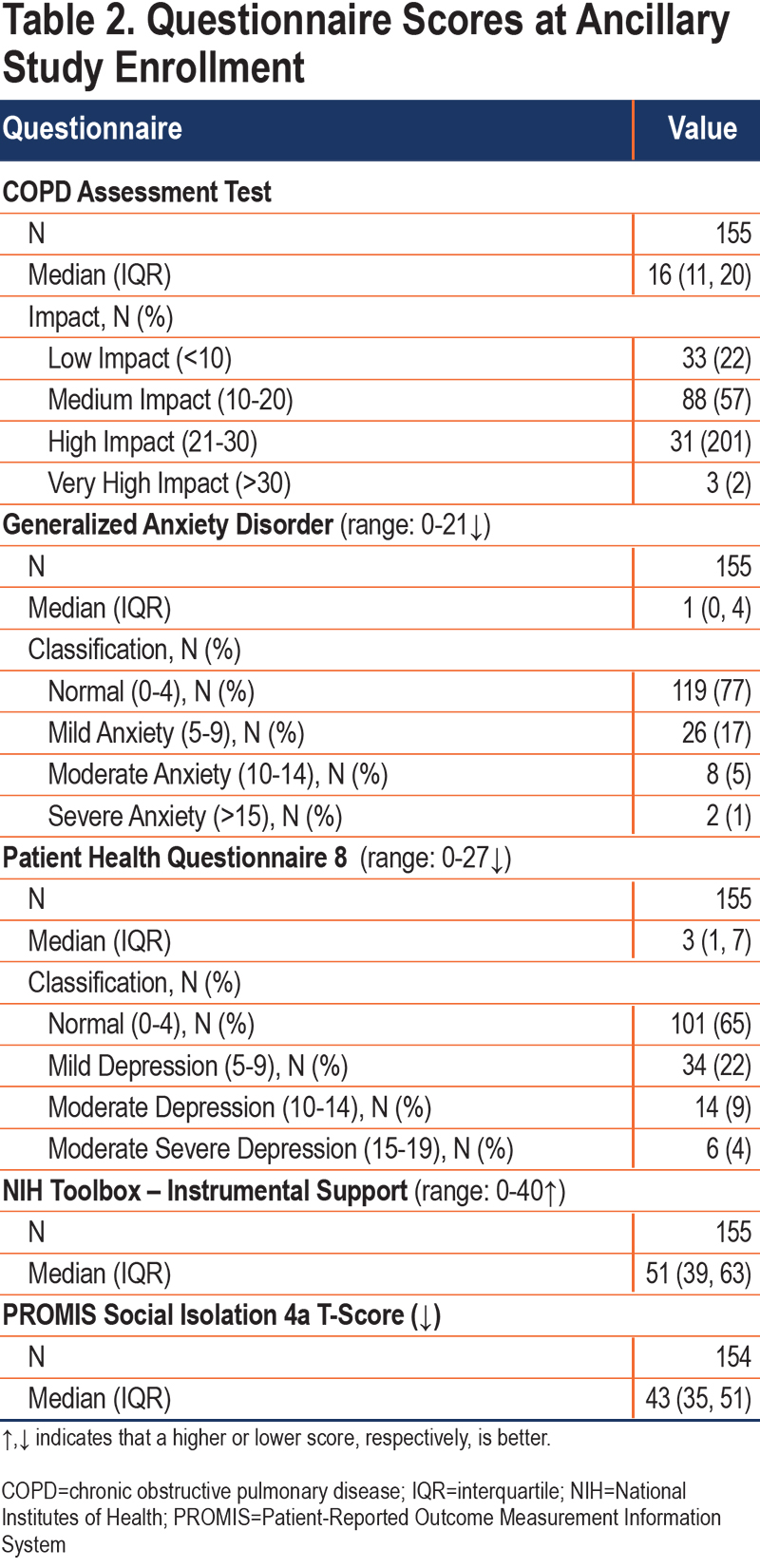
Study interviews were conducted by trained study staff at each study center by phone every 2 weeks to query participants about COVID-19 exposures, symptoms, testing, and health care utilization since the last interview. At the first interview and every 8 weeks thereafter, participants were also asked about their knowledge of and attitudes towards COVID-19 and administered the following 5 validated questionnaires (Supplementary Table 1 in the online supplement): the COPD Assessment Test (CAT), used to evaluate the impact of COPD symptoms on a patient’s life19; Generalized Anxiety Disorder-7 questionnaire (GAD-7), used to measure symptoms of general anxiety in the prior 2 weeks20; Patient Health Questionnaire (PHQ-8), used to evaluate symptoms of depression in the prior 2 weeks21; Patient-Reported Outcomes Measurement Information System (PROMIS) Social Isolation Short Form 4a, used to measure social isolation22; and the National Institutes of Health (NIH) Toolbox Items Bank v2.0 – Instrumental Support, used to evaluate instrumental support, a type of social support that is considered tangible and/or physical.23
All health outcomes, including COVID-19 diagnosis and test results, were based on self-reports. The COVID-19 case definition was a report of an infection confirmed by a positive test result, regardless of the type of test. Individuals at elevated risk of depression, anxiety, or social isolation based on their response to standard questionnaires were referred to local mental health and community support resources maintained by each site. Most data are based on information collected between May 2020 and May 2021. Vaccination rates are based on data collected as of December 30, 2021.
Statistical Analysis
Descriptive analyses were used to summarize participant characteristics at enrollment and during follow-up. Data from all interviews were evaluated to determine the incidence of COVID-19 infections and hospitalizations during the follow-up period. Data from the standard and COVID-19 specific questionnaires were evaluated over 6 approximately 2-month periods beginning in May 2020 and with the last period ending in May 2021. If 2 sets of standard or COVID-19 specific questionnaires were available for a participant in one 2-month period, the first one was selected for inclusion in the analysis.
Differences in prevalence of characteristics among time periods were evaluated with logistic regression models with covariates for sex and race using generalized estimating equations and adjusting for repeated measurements24; an autoregressive covariance structure was used. The first period, June 2020, was the reference period and P-values reported for period differences are from contrasts of subsequent periods with June 2020. Rates of hospitalization from data collected in the ancillary study were compared to the rates in the LEEP cohort from May 2017 to March 1, 2020. Data were analyzed with SAS version 9.4 and Stata/IC 15.1 for comparison of rates.
Results
Characteristics of the Study Population
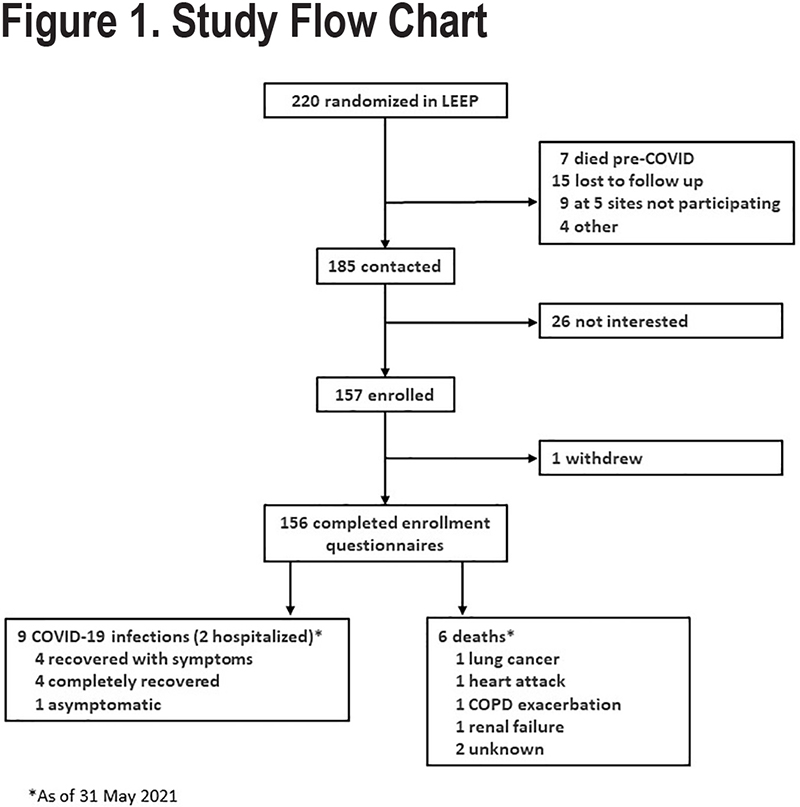
Of the 220 patients enrolled in the parent LEEP study, 185 were contacted about the COVID-19 ancillary study, 157 of whom enrolled (Figure 1). Contacted participants who elected not to enroll cited “time” as the main reason for not participating. Most participants, 104 (66%), had completed the LEEP trial, and overall, 50% had been randomly assigned to losartan (Table 1).
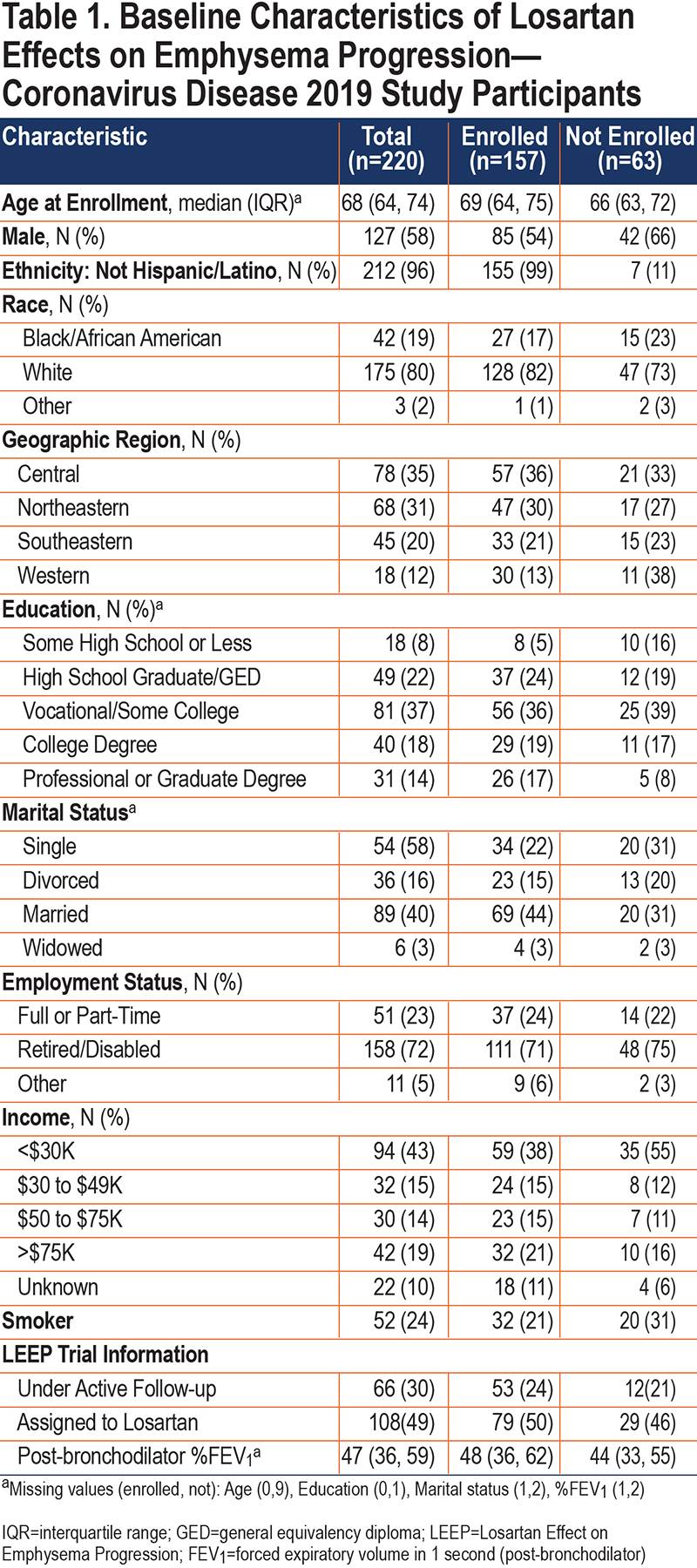
The median age of ancillary study participants was 66 years; most were White (82%) and male (56%) (Table 1). The median post-BD FEV1 % predicted at trial enrollment was 48% predicted, with over 75% of participants having CAT scores greater than 10 at enrollment, indicating that this was a cohort of severe, symptomatic COPD patients (Table 2). Most participants (73%) enrolled between May 2020 and July 2020 with 73 to 138 participants being interviewed in each 8-week period (Supplemental Figure 1 in the online supplement). The median length of follow-up was 38 weeks and the median number of interviews completed per participant was 19 (Supplemental Figure 2 in the online supplement).
Impact of Coronavirus Disease 2019 on Physical Health
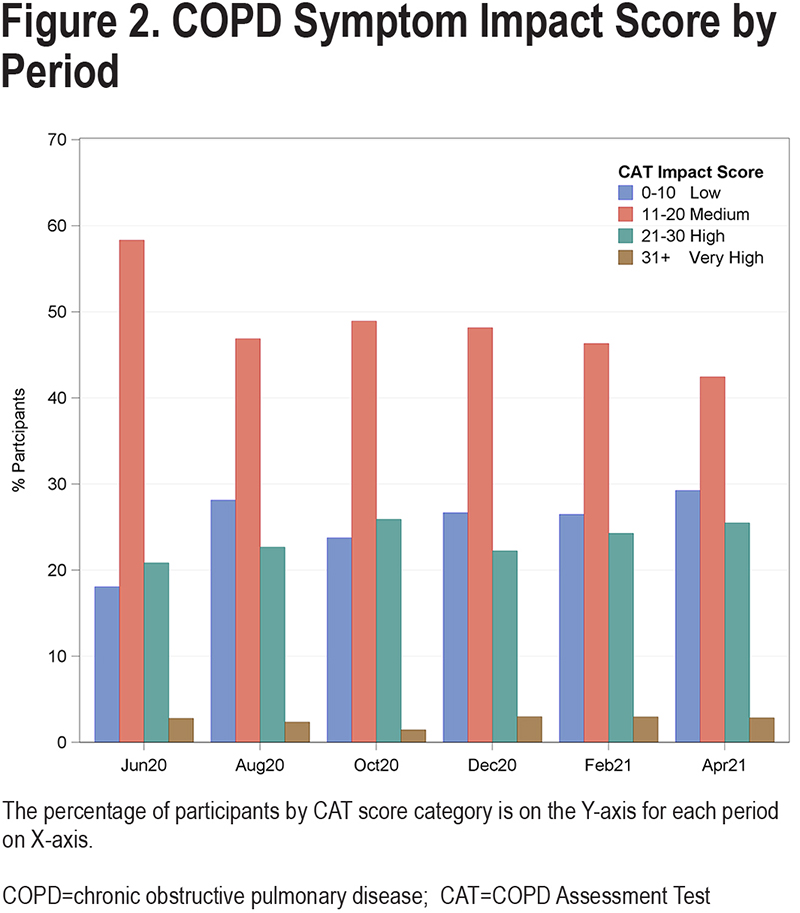
Between May 2020 and May 2021, 9 participants reported a diagnosis of COVID-19 that was confirmed by a positive test, 1 of whom was asymptomatic. Two reported being hospitalized for COVID-19 infection, including 1 before enrolling in the ancillary study (Figure 1). A total of 36 hospitalizations not-related to COVID-19 were reported by 30 participants, including 7 for COPD exacerbations and 9 for other respiratory problems. The rate of all-cause hospitalizations in this ancillary study was similar to the rate in the entire LEEP cohort prior to March 1, 2020, 0.31 and 0.35 per person year, respectively (P=0.63). However, hospitalization rates for COPD exacerbations were reduced in this ancillary study compared to the pre-pandemic rate observed in the LEEP trial (0.06 versus 0.14 per person-year, P=0.05). We recorded 6 deaths during follow-up, none of which were attributed to COVID-19. Patients remained fairly symptomatic during the follow-up period, with most patients having medium impact of symptoms (CAT scores 10-20) and nearly 30% having high or very high symptom burden (CAT scores 21-30) (Figure 2).
The Impact of Coronavirus Disease 2019 on Social Support and Mental Health
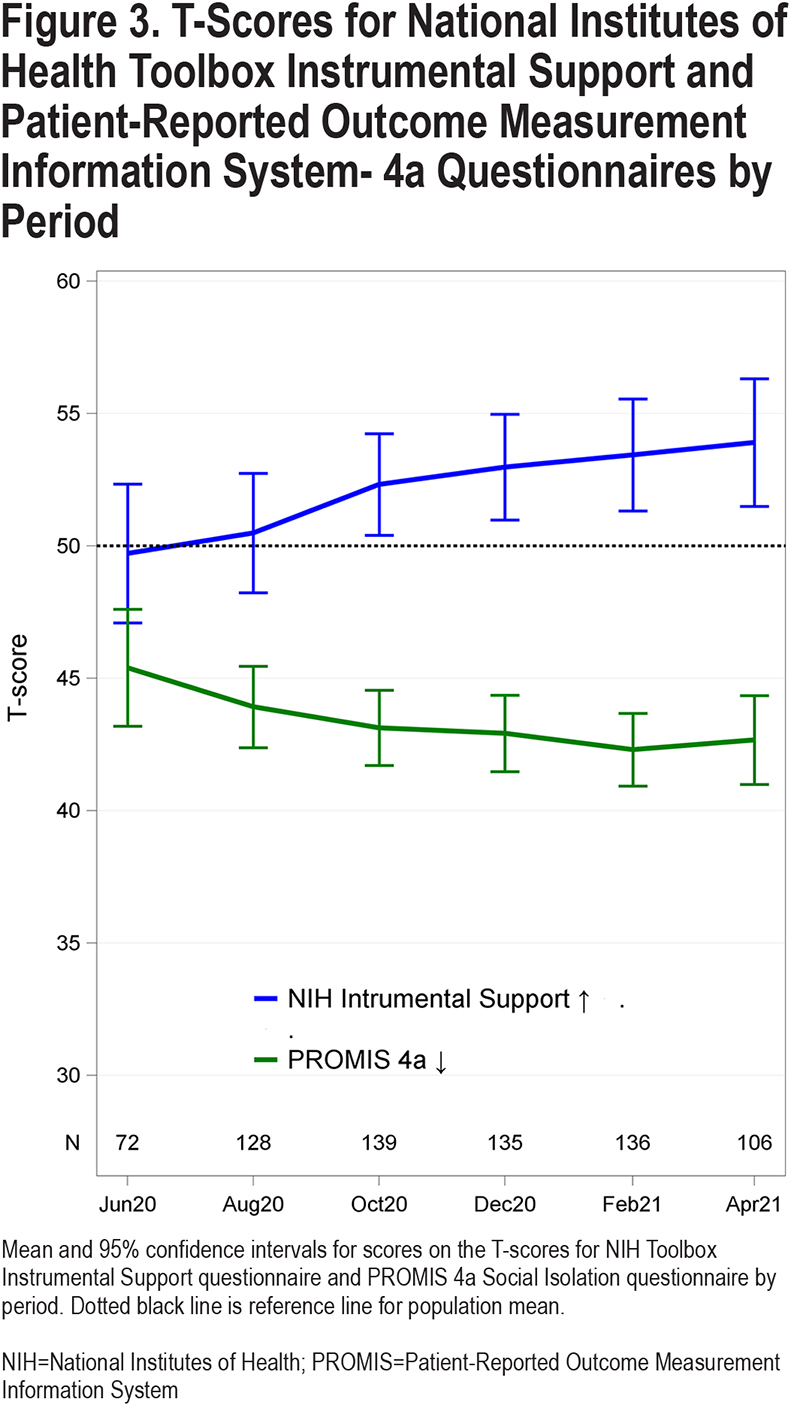
At enrollment, the median (interquartile range [IQR]) scores on the NIH Toolbox measure of Instrumental Support and the PROMIS Social Isolation 4a questionnaires T-scores were 51 (39, 63) and 43 (35, 51), indicating that most participants had assistance, if needed, with activities of daily living and had social support networks (Table 2). These scores remained stable throughout follow-up (Figure 3).
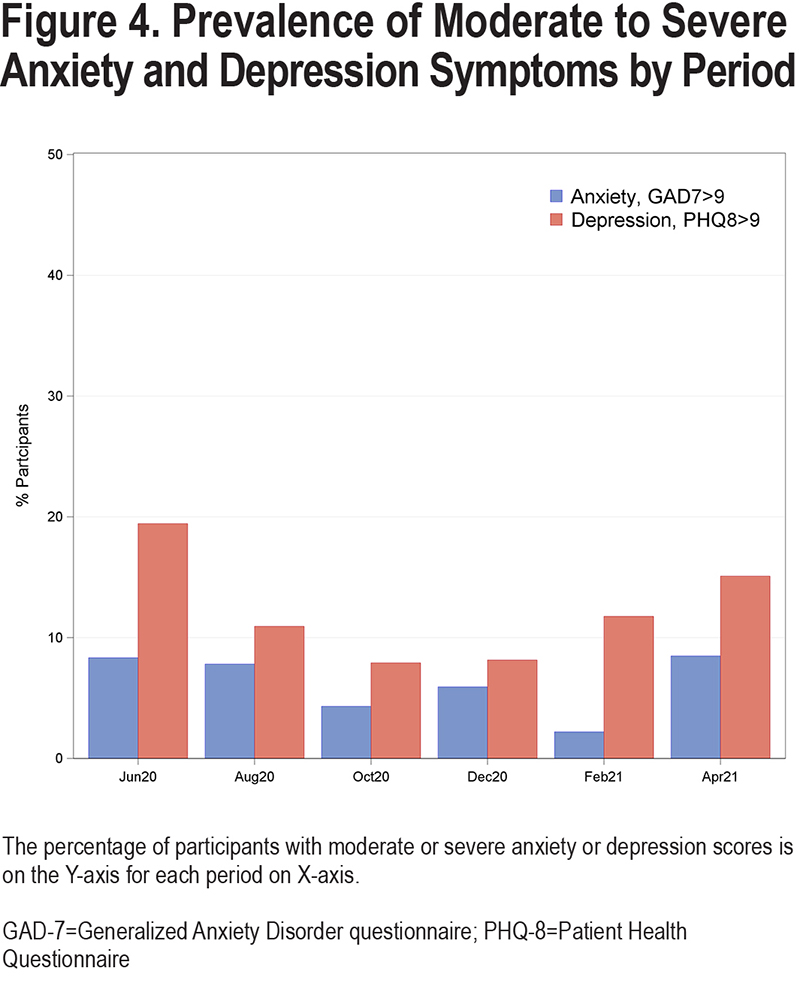
The prevalence of moderate to severe symptoms of anxiety (GAD-7≥10) or depression (PHQ-8≥10) was 8% (6 participants) and 19% (14 participants), respectively, in June 2020 (Figure 4). The prevalence of moderate to severe anxiety remained low throughout the study period, dropping to 2% by February 2021, a rate which was significantly lower than the rate at study onset (P=0.05). The prevalence of moderate to severe depression also decreased to 8% in October and December 2020, which were significantly lower as compared to June 2020 (P=0.03 for both periods) but rebounded to higher rates in February (12%) and April 2021 (15%).
Knowledge, Perceptions, and Attitudes About the Coronavirus Disease 2019 Pandemic
Study participants relied on a variety of sources for gathering information on COVID-19. The most trusted sources of information and guidance about COVID-19 were state and local government (76%), local news (76%), the Centers for Disease Control and Prevention (76%), and health care providers (69%) (Supplemental Figure 3 in the online supplement). Only 22% endorsed social media as trustworthy sources of information about COVID-19. Participants were well-informed about how COVID-19 was transmitted, with 97% knowing that asymptomatic people can be infectious. Over 75% knew that loss of smell or taste were distinctive symptoms of infection in June 2020, which increased to 90% by August 2020.
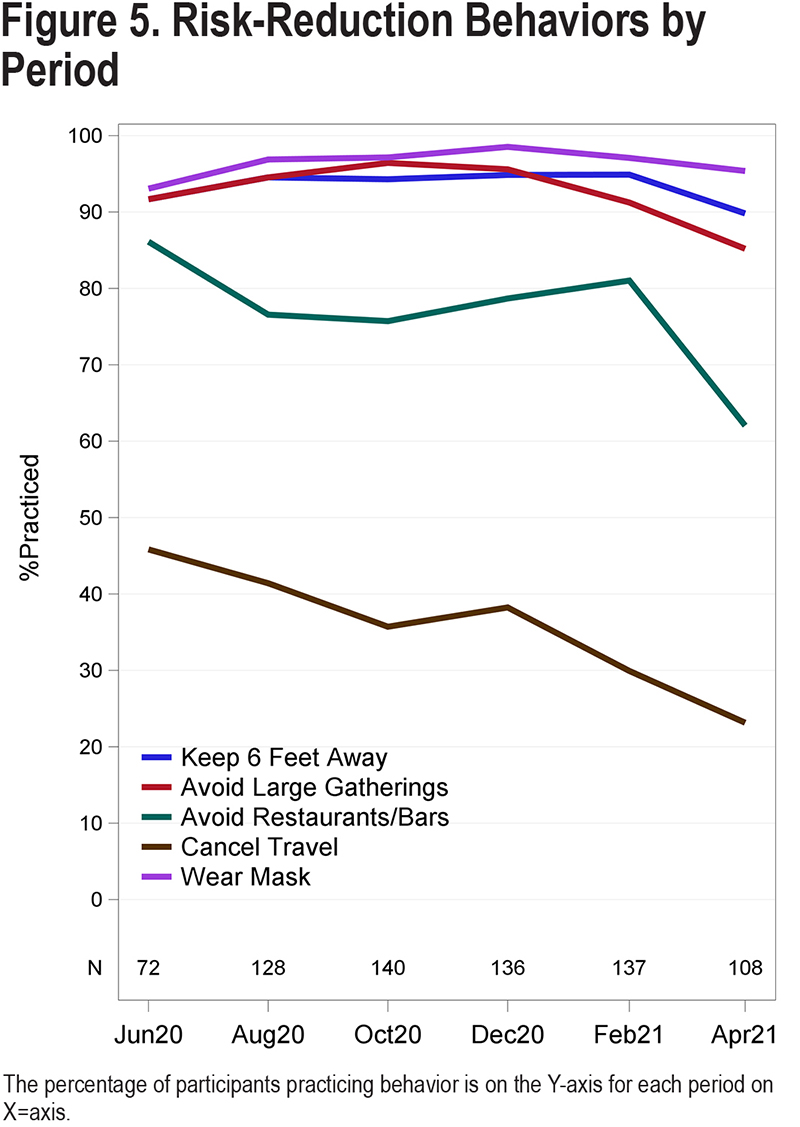
Most participants believed that personal actions such as wearing masks, social-distancing (maintaining 6 feet distance), and avoiding public places were effective in preventing COVID-19 transmission and practiced those behaviors (Figure 5). There was a decline in participants avoiding restaurants and bars as the pandemic continued, dropping from 84% in June 2020 to 62% in April 2021 (P<0.001). The percentage of participants canceling travel plans also declined, decreasing to 23% in April 2021 from 46% in June 2020 (P<0.0001).
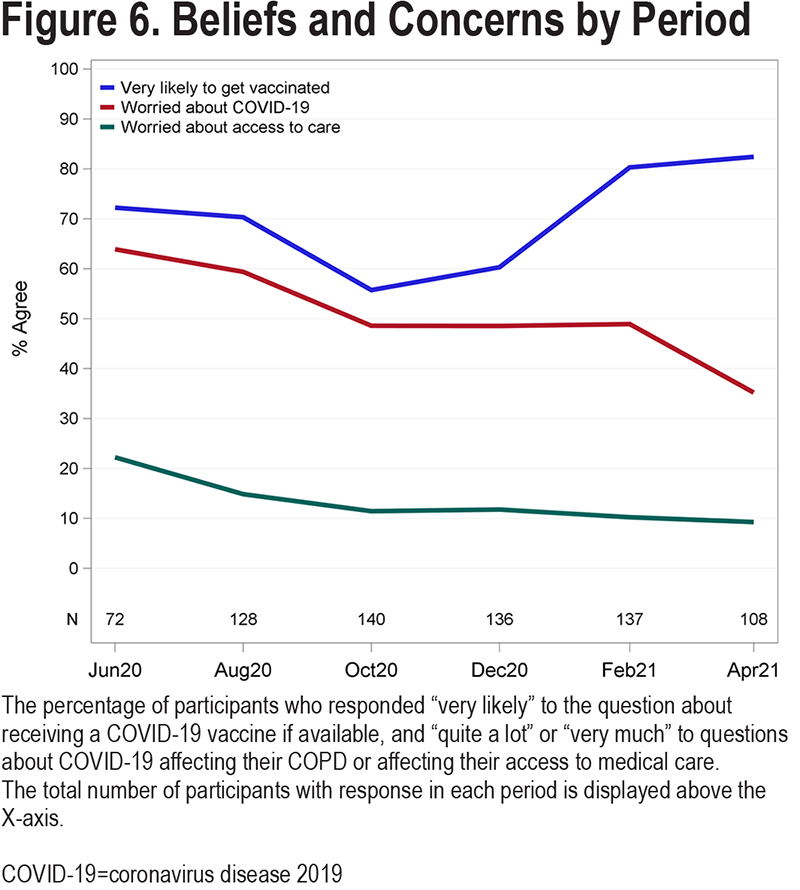
Concern about the pandemic affecting their COPD declined from 64% in June 2020 to 35% in June 2021 (P<0.001) (Figure 6), which corresponds to the decline in concerns about accessing medical care from 22% in June 2020 to 9% in April 2021. Willingness to receive a COVID-19 vaccine fluctuated during the study period (Figure 6), dropping initially from 72% in June 2020 to 56% in October 2020 (p=0.08) but subsequently rebounding to 83% by April 2021 (P=0.003). As of December 31, 2021, 91% of 138 participants had received 1 or more doses of a COVID-19 vaccine; the vaccination status of 18 participants is unknown.
Discussion
In this study, we examined the impact of the COVID-19 pandemic during the first year on the physical and mental health of a well-characterized cohort of patients with moderate to severe COPD with evidence of emphysema on CT enrolled in a clinical trial.18 We found similar rates of both hospitalization and of moderate to severe depression and anxiety compared to rates prior to the pandemic, but hospitalizations for COPD exacerbations were reduced in the pandemic. Our results indicate that our cohort of moderate to severe, symptomatic COPD individuals stayed strongly connected to health care systems and coped well during the pandemic. It should be noted that as participants in a clinical trial, these patients were a select group with strong ties to health care providers and high levels of health literacy, which limits the generalizability of our results.
We observed 9 COVID-19 infections in almost 100 person years but only 2 of the 9 participants were hospitalized for COVID-19, which was much lower than the rate of 62% reported for COPD patients diagnosed with COVID-19 infections in the National COVID Cohort Collaborative.25 None of the 9 died from COVID-19. We also observed lower rates of hospitalization for COPD exacerbations in our cohort than those observed in the clinical trial during the first 2 years of the study (2017-2019), which is consistent with reductions reported in a systematic review of hospitalization for COPD exacerbations.26 We speculate that the rapid adoption of COVID-19 risk reduction behaviors led to fewer respiratory viral infections which often trigger severe COPD exacerbations.27 Because of the few COVID-19 cases observed in our study we were unable to conduct planned analyses about potential factors affecting risk or severity of COVID-19 in patients with COPD.
While risk reduction behaviors were protective from COVID infection, there was concern that these behaviors could cut patients off from their support systems. In turn, this deprivation of support systems could lead to increased stress, anxiety, feelings of isolation or depression, all of which have been shown to be associated with higher risk of exacerbation and other poor outcomes in COPD patients.5,6,10 The rates of depression observed in our study (19%) were similar to the prevalence of elevated depression scores in a group of over 200 patients with less severe COPD enrolled in a cohort study in 2016 that used the same depression screening questionnaire.28,29 The observed rates are also similar to rates of 15% and 19% reported in a nationally representative sample of U.S. adults age 60 or older surveyed in the COVID-19 and Life Stressors Impact on Mental Health and Well-being study in 2020 and 2021, respectively.30 Therefore, while a rate of close to 20% may be alarming, the rates of depression in people with moderate to severe COPD are higher than those of the general population.31 The observed prevalence of moderate to severe symptoms of anxiety of 8% was similar to what we reported in the aforementioned cohort of COPD patients study in 2016, which was 13% using the same instrument.29 Some studies that asked participants about changes in mental health specifically due to the pandemic reported increased anxiety.13,14 Our study, and others15,16,32 that used validated measures of anxiety, such as the GAD-7, which do not reference COVID-19, reported no increase in anxiety.
Our participants reported high levels of social support and low levels of isolation which likely contributed to the relatively low rates of depression and anxiety. Anecdotally, many participants reported that they enjoyed the study interviews and looked forward to them, and it is possible that being in the study may have affected the very rates of depression and anxiety that we were trying to measure. Taken together, these findings suggest that for this select group of COPD patients who were already coping with physical limitations, the impact of pandemic restrictions on the prevalence of depression and anxiety may be less than in healthier, more active groups.
A major strength of our study is that we leveraged an ongoing clinical trial to collect longitudinal information about the impact of the pandemic on a well-characterized cohort of participants with moderate to severe COPD who were socioeconomically diverse and from different regions of the United States. However, generalizability of our findings is limited to a select group of patients with significant morbidity who are well connected to the health care community and not representative of COPD patients at large. Additionally, we did not have pre-pandemic measurements of depression and anxiety from the same population and, therefore, rely on rates from other studies for comparison. Finally, it should be noted that risk avoidance behaviors were self-reported, but it would be extremely difficult to objectively assess compliance of behaviors such as mask wearing and hand hygiene for a large, geographically diverse population.
Nevertheless, our study participants demonstrated remarkable resilience in coping with the risks and restrictions associated with the COVID-19 pandemic as there was no uptick in symptoms of depression or anxiety. They acquired knowledge about COVID-19 from reputable sources, quickly adopted risk avoidance behaviors, and voiced acceptance with vaccine recommendations, all of which likely contributed to the few cases of COVID-19 and the reduction in severe COPD exacerbations. These results emphasize the need to examine the impact of the COVID-19 pandemic on vulnerable populations across multiple domains of health.
Acknowledgements
Author contributions: Study conception and design provided by Janet Holbrook and Robert Wise. Data collection provided by Kaharu Sumino. Analysis and interpretation of results provided by William Zhang, Stephanie LaBedz, Janet Holbrook, Andrew Gangemi, Ramasubramanian Baalachandran, Michelle Eakin, Robert Wise, and Kaharu Sumino. Draft manuscript preparation provided by William Zhang, Stephanie LaBedz, Janet Holbrook, Andrew Gangemi, and Kaharu Sumino. All authors reviewed the results and approved the final version of the manuscript.
Declaration of Interest
The authors have no conflicts of interest to report