Running Head: Aerobic Exercise for COPD
Funding Support: None
Date of Acceptance: October 23, 2025 | Published Online Date: October 29, 2025
Abbreviations: 6MWT=6-minute walk test; BMI=body mass index; CI=confidence interval; COPD=chronic obstructive pulmonary disease; FEV1=forced expiratory volume in 1 second; FVC=forced vital capacity; HADS=Hospital Anxiety and Depression Scale; mMRC=modified Medical Research Council; RoB2= risk-of-bias tool for randomized trials; SD=standard deviation; SGRQ=St George's Respiratory Questionnaire; SMD=standard mean difference; SpO2=peripheral capillary oxygen saturation; WMD=weighted mean difference
Citation: Zhang M, Khan MU. The effects of aerobic exercise on prognosis, quality of life, and psychological outcomes of patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. Chronic Obstr Pulm Dis. 2026; 13(2): 167-176. doi: http://doi.org/10.15326/jcopdf.2025.0657
Online Supplemental Material: Read Online Supplemental Material (1443KB)
Introduction
Chronic obstructive pulmonary disease (COPD) is a common cause of respiratory morbidity and mortality worldwide, and affects nearly 12.64% of adults aged more than 40 years, with a higher prevalence among men.1 COPD imposes a great burden on patients, families, and society and has been linked to higher clinical disease burden, lower socioeconomic status, and decreased health-related quality of life.2 COPD is strongly associated with muscle wasting and frailty, which can limit exercise tolerance and functional capacity and increase the risk of fatigue, anxiety, and depressive symptoms among patients with COPD.3,4 It has been reported that psychological comorbidities, like anxiety and depression, can significantly increase the clinical and economic burdens of COPD, highlighting the importance of the psychological aspect of care in COPD.5
Due to the importance of frailty and physical rehabilitation in patients with COPD, an increasing number of studies have investigated the effects of different modalities of exercise on different aspects of COPD management.6-8 In particular, aerobic exercise, defined as physical activities increasing the heart rate and the body's use of oxygen, was found to enhance the muscle mass and strength of patients with COPD, thereby contributing to pulmonary rehabilitation.9 Due to the beneficial effects of aerobic exercise on physical rehabilitation and strength of patients with COPD, this meta-analysis pools data from randomized controlled trials to assess the effects of aerobic exercise on functional recovery, prognosis, quality of life, and psychological outcome of patients with COPD, thereby providing a comprehensive overview of the effect of aerobic exercise on COPD. Although several meta-analyses assessed the effects of aerobic exercise on COPD,10-12 this meta-analysis analyzes different outcomes, compares the effect of aerobic exercise with a control group, unlike previous meta-analyses, and includes a markedly higher number of clinical trials. For instance, Ward et al measured the effects of aerobic exercise only on peak oxygen uptake and reported that aerobic exercise can moderately improve peak oxygen uptake; however, they combined the results of randomized and nonrandomized studies.10 By conducting a meta-analysis, Chen et al reported that aerobic exercise can significantly improve dyspnea levels and functional and endurance exercise capacity, but not lung function, in COPD.11 The results of previous meta-analyses remain inconsistent for some physical outcomes, and their data came from a smaller number of studies. Also, previous meta-analyses have rarely addressed the effects of aerobic exercise on psychological outcomes in COPD. In addition to addressing such points, this meta-analysis conducted several subgroup analyses to investigate in which subgroup of patients with COPD aerobic exercise can offer more benefits.
Method
Search Strategy
PubMed, the Cochrane Library, Scopus, and Web of Science were systematically searched from the inception to June, 20, 2025, based on the following search keywords (Supplementary Material 1 in the online supplement): ("chronic obstructive pulmonary disease" OR “COPD”) [title, abstract, keywords] AND ("aerobic" OR " aerobic exercise" OR "aerobic training") [title, abstract, keywords] AND (“clinical trial” OR "randomized controlled trial" OR "RCT").
Furthermore, we searched the reference list of the included original studies and recent reviews to find additional eligible studies. After transferring the retrieved studies to EndNote 7.0, duplicate articles were removed. This study strictly followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines,13 and the protocol of this meta-analysis was prospectively registered in The International Prospective Register of Systematic Reviews: PROSPERO (CRD420251090253). We included articles measuring the effects of aerobic exercise, defined as physical activities increasing the heart rate and the body's use of oxygen, on different aspects of care in COPD. The type or intensity of exercise was not limited in this meta-analysis.
Inclusion and Exclusion Criteria
We adopted the following inclusion criteria: (1) study design was a clinical trial; (2) all participants had COPD; (3) the study compared the effects of aerobic exercise with a control group not receiving aerobic exercise or other forms of exercises; and (4) the study reported data on at least 1 of the outcomes of interest, including 6-minute walk test (6MWT), forced expiratory volume in 1 second (FEV1), FEV1 to forced vital capacity (FVC), peripheral capillary oxygen saturation (SpO2), the St George's Respiratory Questionnaire (SGRQ) score, the modified Medical Research Council dyspnea scale (mMRC) score, and the Hospital Anxiety and Depression Scale (HADS) score.
Studies meeting the following criteria were excluded: (1) case reports, case series, observational studies, or review articles; (2) studies lacking a control group; (3) studies reporting none of the outcomes of interest; and (4) non-English studies.
Data Extraction and Outcome Measures
Two authors independently performed the literature search and evaluated the eligibility of the identified studies. All discrepancies were resolved through consultation. Based on the titles and abstracts, the 2 authors assessed the relevance of the articles and measured their compliance with the aforementioned inclusion and exclusion criteria. Thereafter, the full-text version of the remaining articles was assessed to determine whether they were eligible for inclusion in this meta-analysis.
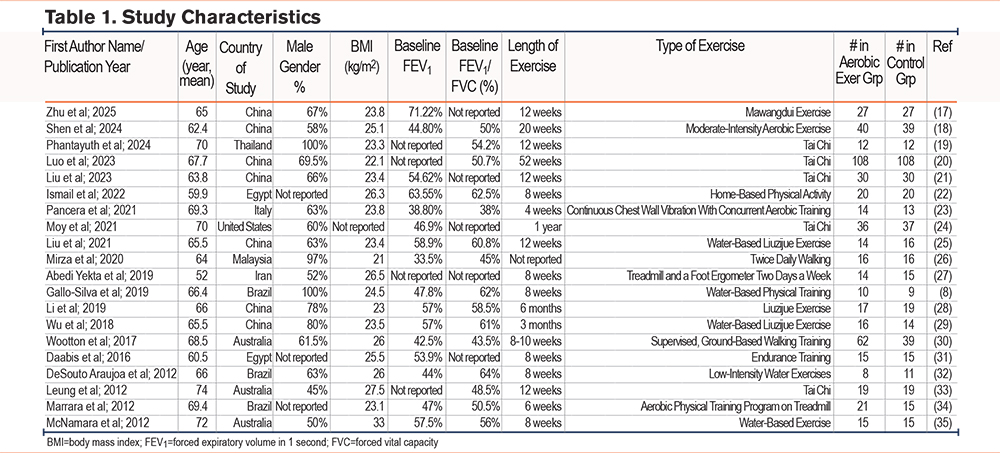
Data extracted from each article were as follows: study characteristics (the first author’s name, country of study, publication year, length of exercise, and type of exercise), population (participant number in each group, participants’ age, gender, and baseline body mass index [BMI], baseline FEV1, baseline FEV1/FVC), and the outcomes of interest.
Risk of Bias Assessment
Two authors independently assessed the risk of bias for all included studies, and all differences were resolved via consultation. The revised Cochrane risk-of-bias tool for randomized trials (RoB2) was adopted to evaluate the risk of bias according to the randomization process, outcome measurement, missing outcome data, deviations from intended interventions, and selection of the reported results, thereby offering an overall risk of bias. Each domain received one of the following ranks: low risk of bias, some concerns, and high risk of bias.14
Statistical Analysis
Changes in mean and standard deviation were extracted for all outcomes of interest to calculate weighted mean difference (WMD) for nonscale outcomes and standardized mean difference (SMD) for scale outcomes and 95% confidence interval (CI). A random-effects model (DerSimonian-Laird) was employed to pool data due to high methodological heterogeneity. We performed this meta-analysis using Stata version 17.0 (StataCorp; College Station, Texas). The chi-square test and I2 statistic were utilized to evaluate heterogeneity between trials, and an I2 of more than 50% was deemed high heterogeneity.
Subgroup Analysis
Subgroup analysis was performed for the SGRQ score, which measures health impairment in patients with asthma and COPD, and the 6MWT based on sample size (less than 50 versus more than 50), length of exercise (less than 8 weeks versus more than 8 weeks), baseline FEV1 (less than 50% versus more than 50%), baseline FEV1/FVC (less than 50% versus more than 50%), participants’ age (less than 65 years versus more than 65 years), BMI (less than 25kg/m2 versus more than 25kg/m2), and country of study (China versus other countries). Regarding the threshold for grouping in subgroup analysis, we chose clinically relevant values based on the methodology of the included articles.
Sensitivity Analysis
Employing the leave-one-out test, sensitivity analysis was conducted for the SGRQ and the 6MWT, the only outcomes reported by more than 10 studies.15,16 Sensitivity analysis was not conducted for other outcomes because the leave-one-out test has been specifically recommended for meta-analysis of more than 10 studies.
Results
The Results of the Systematic Search
Initially, we found 52 records in PubMed, 221 articles in the Cochrane Library, 289 records in Scopus, and 137 records in Web of Science. After removing duplicates, 466 records were excluded through abstract and title screening, and 56 articles were excluded during full-text review. Finally, 20 randomized controlled trials with 1003 participants were included in this systematic review (Figure 1 and Table 1).8,17-35 Among the countries, China published the highest number of articles, followed by Australia and Brazil. Patients were mostly aged 60–70 years, and their BMI was mostly 22–27kg/m2. The length of exercise training ranged from 4 weeks to 1 year across studies, and most studies reported a baseline FEV1/FVC of around 50% (Table 1).
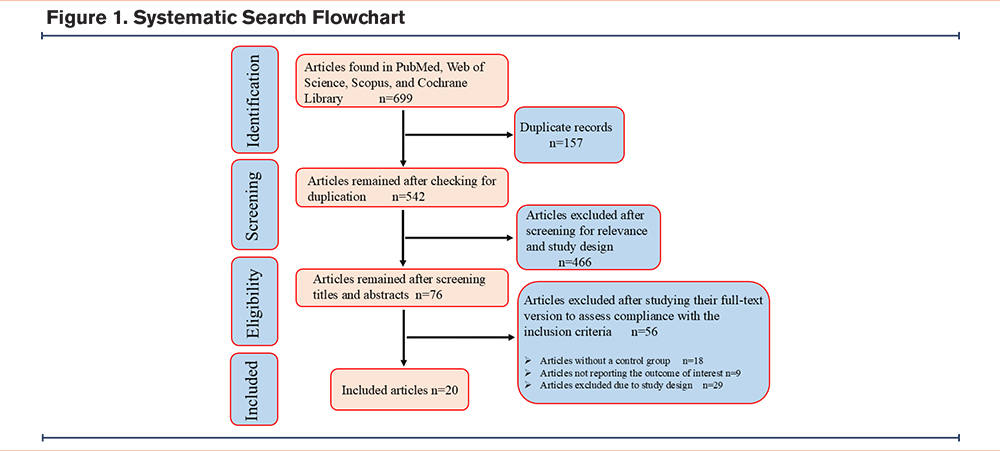
Risk of Bias Assessment
Using the RoB2 tool, the risk of bias for most of the included studies was determined to be high. Particularly, due to the nature of interventions and study design, blinding of participants and researchers was not feasible in most studies, which could potentially increase the risk of bias (Supplementary Material 2 in the online supplement).
Meta-Analysis
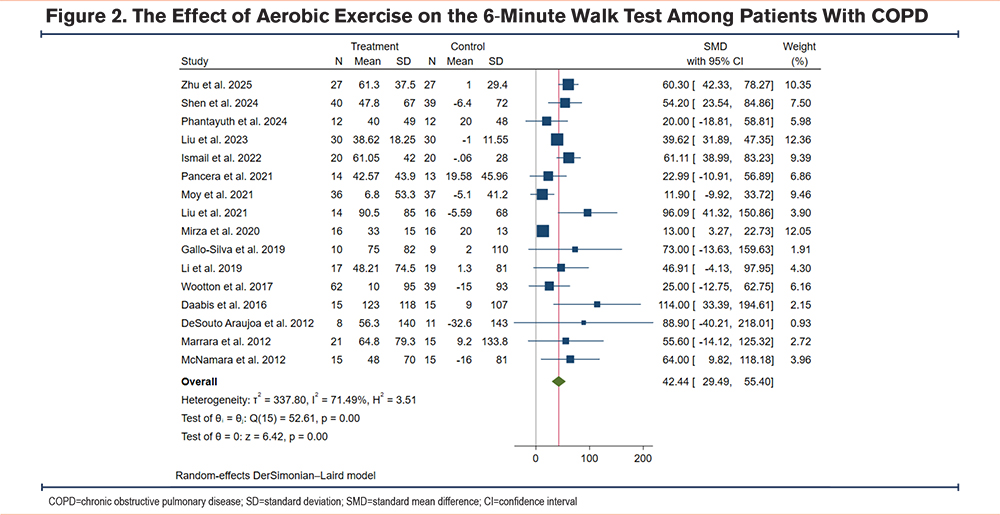
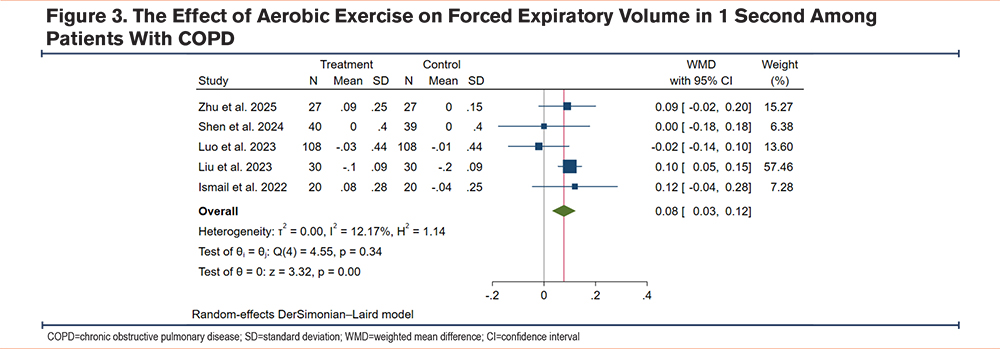
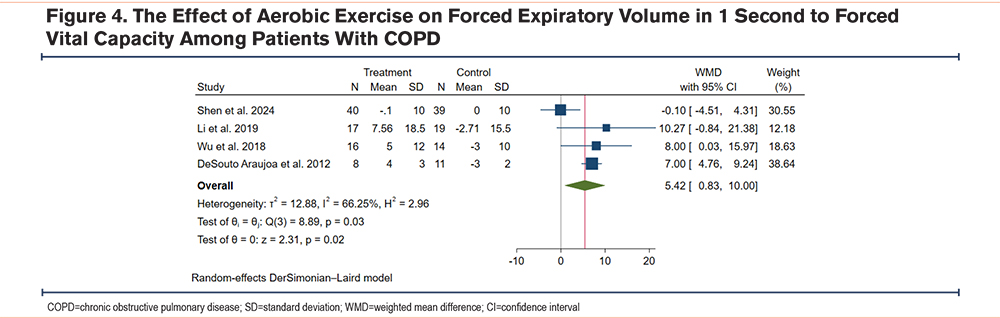
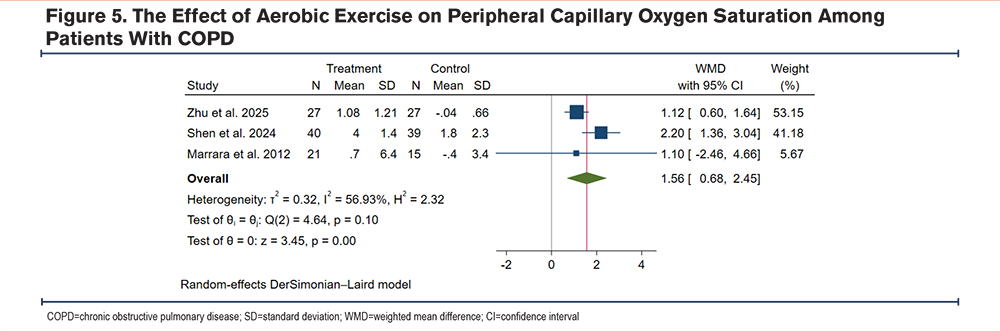
Due to high statistical and methodological heterogeneity, a random-effects model was employed to pool data. The result showed that compared to the control group, patients with COPD undergoing exercise training had a significantly increased 6MWT (WMD 42.44m, 95% CI [29.49, 55.40], I2=71.49%) (Figure 2), FEV1 (WMD 0.08L, 95% CI [0.03, 0.12], I2=12.17%) (Figure 3), FEV1/FVC (WMD 5.42%, 95% CI [0.83, 10.00], I2=66.25%) (Figure 4), and SpO2 (WMD 1.56%, 95% CI [0.68, 2.45], I2=56.93%) (Figure 5), which suggests that aerobic exercise can improve the functional capacity and respiratory reserve of patients with COPD.
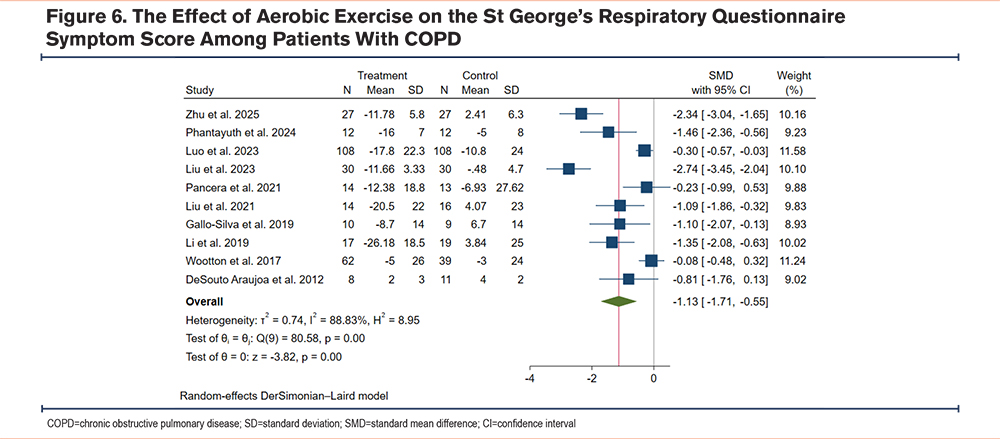
On the other hand, the results revealed that compared to the control group, aerobic exercise markedly decreased the SGRQ symptom score (SMD -1.13, 95% CI [-1.71, -0.55], I2=88.83%) (Figure 6) and the SGRQ total score (SMD -1.44, 95% CI [-2.09, -0.78], I2=92.37%) (Supplementary Material 3 in the online supplement), suggesting an improvement in health condition. Similarly, compared to the control group, aerobic exercise markedly decreased the mMRC score for dyspnea (SMD -0.81, 95% CI [-1.13, -0.48], I2=27.68%) (Supplementary Material 3 in the online supplement), and the HADS-anxiety score (SMD -1.17, 95% CI [-2.25, -0.09], I2=88.16%) (Supplementary Material 3 in the online supplement), but its effect on the HADS-depression score (SMD -0.25, 95% CI [-1.16, 0.66], I2=85.37%) (Supplementary Material 3 in the online supplement) did not meet the threshold of statistical significance. These findings suggest that aerobic exercise markedly improved the quality of life, dyspnea, symptom severity, and anxiety of patients with COPD.
Subgroup Analysis
Subgroup analysis regarding the 6MWT indicated that the results were significant in all subgroups and there was no significant difference between the 2 subgroups of sample size (less than 50 versus more than 50), length of exercise (less than 8 weeks versus more than 8 weeks), participants’ age (less than 65 years versus more than 65 years), BMI (less than 25kg/m2 versus more than 25kg/m2), and country of study (China versus other countries), but aerobic exercise led to a greater improvement in 6MWT results when FEV1 and FEV1/FVC were more than 50% (Supplementary Material 4 in the online supplement).
Subgroup analysis for the SGRQ score unveiled that the results were significant in all subgroups of sample size (less than 50 versus more than 50), length of exercise (less than 8 weeks versus more than 8 weeks), participants’ age (less than 65 years versus more than 65 years), BMI (less than 25kg/m2 versus more than 25kg/m2), and country of study (China versus other countries), but the results of aerobic exercise on the SGRQ test result were significant only when FEV1 and FEV1/FVC were more than 50%. Furthermore, subgroup analysis indicated that aerobic exercise for more than 8 weeks had a significantly greater impact on the SGRQ score compared to aerobic exercise for less than 8 weeks (Supplementary Material 4 in the online supplement).
Collectively, subgroup analysis indicated that aerobic exercise can offer greater benefits in the long term, and those with an FEV1 and FEV1/FVC more than 50% can benefit more from aerobic exercise.
Sensitivity Analysis
The leave-one-out test was conducted for the 6MWT and the SGRQ score, 2 outcomes reported by more than 10 studies. The results indicated that removing individual studies did not change the overall results, suggesting the robustness of our results (Supplementary Material 5 online supplement).
Discussion
This meta-analysis with 20 clinical trials indicated that compared to the control group, aerobic exercise increased the 6MWT, FEV1, FEV1/FVC, and SpO2 and decreased the SGRQ symptom score, the SGRQ total score, the mMRC score, and the HADS-anxiety score. In addition, subgroup analysis revealed that aerobic exercise can offer greater benefits after 8 weeks, and those with FEV1 and FEV1/FVC more than 50% can benefit more from aerobic exercise.
Similar to our study, Chen et al conducted a meta-analysis and reported that compared to the control group, aerobic exercise improved the dyspnea and functional capacity of patients with COPD; however, they showed that aerobic exercise did not significantly change lung function.11
Consistent with our findings, Benzo-Iglesias et al pooled data from 4 studies and found that water-based aerobic exercise can significantly improve the functional capacity of patients with COPD as measured by the 6MWT.12 Qiao et al included 15 studies in their network meta-analysis and reported that high-intensity lower limb aerobic exercise is superior to medium-intensity and low-intensity lower limb aerobic exercise in strengthening functional capacity and promoting health-related quality of life.36 In patients with asthma who suffer from obstructive airway disease similar to those with COPD, aerobic exercise was shown to increase FEV1 and enhance the quality of life of patients.37
Physical training, particularly aerobic exercise, was found to improve oxidative phosphorylation, mitochondrial biogenesis, and muscle strength and enhance physiological adaptation in people with COPD, characterized by a significant increase in peak oxygen uptake.7,10,38 Importantly, we found that aerobic exercise is more effective when FEV1 and FEV1/FVC are more than 50%, offering a greater capacity for adaptation.
Patients with COPD, particularly those suffering from frequent exacerbations and several comorbidities, have worse health-related quality of life.39 In addition, physical activity and better functional capacity in the 6MWT are believed to improve health-related quality of life among patients with COPD.39 In addition, it was found that the SGRQ total score and component scores significantly increase with the worsening of COPD grade and aging.40 Using the SGRQ and mMRC scores, we found that aerobic exercise can significantly improve the symptom score and health-related quality of life among patients with COPD.
Nearly half of patients with COPD suffer from anxiety and depression, which highlights the importance of psychological care for these patients.41 Furthermore, previous studies have indicated that anxiety and depression increase the risk of acute exacerbation, rehospitalization, and mortality among patients with COPD.42 The meta-analysis conducted by Selzler et al showed a weak but negative correlation between physical activity and anxiety or depression in patients with COPD.43 Our meta-analysis showed that physical activity significantly improved anxiety scores among patients with COPD, but its effect on depression did not reach the threshold of statistical significance.
The findings of this meta-analysis collectively suggest that aerobic exercise not only improves the prognosis and functional capacity of patients with COPD but also enhances their psychological outcome and quality of life, offering benefits for all aspects of care.
Limitations
There are some limitations to this meta-analysis. First, the clinical trials included in this meta-analysis had a small sample size. Second, due to the nature of the included trials, blinding of participants and researchers was not feasible in all studies, which may have increased the risk of bias, necessitating more caution when interpreting the results. Third, methodological differences, such as differences in the length of intervention and baseline characteristics of participants, were observed across studies; however, degrees of such differences are inevitable in a meta-analysis. Fourth, some of the included studies incompletely reported the baseline characteristics of participants. Future large-scale clinical trials with more detailed data are needed to validate the findings of this meta-analysis.
Strengths
This meta-analysis also had several strengths. First, it included a markedly higher number of studies compared to previous meta-analyses in this regard. Second, it included more outcomes and covered both the physical and psychological aspects of care, thereby providing a holistic view. Third, several subgroup and sensitivity analyses were conducted to assess the factors associated with the protective effects of aerobic exercise on COPD and determine the robustness of our findings.
Conclusion and Clinical Implications
This meta-analysis with 20 clinical trials and 1003 participants showed that compared to the control group, aerobic exercise significantly increased the 6MWT, FEV1, FEV1/FVC, and SpO2 and decreased the SGRQ symptom score, the SGRQ total score, the mMRC score, and the HADS-anxiety score. In addition, subgroup analysis revealed that aerobic exercise may offer greater benefits after 8 weeks, and those with FEV1 and FEV1/FVC more than 50% may benefit more from aerobic exercise. These findings suggest that aerobic exercise may markedly benefit the prognosis, functional capacity, respiratory reserve, symptoms, quality of life, and anxiety of patients with COPD. Given the various benefits of aerobic exercise for patients with COPD and its greater impact in those with milder disease, future guidelines should recommend routine aerobic exercise from the early stages of COPD.
Acknowledgments
Author contributions: MZ was responsible for the conceptualization of the manuscript. MUK and MZ searched the literature and contributed to data extraction and data analysis. MUK was responsible for writing the draft, and MZ was responsible for reviewing the draft. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials: Data will be available by the corresponding author on a reasonable request.
Declaration of Interest
The authors declare no conflict of interest.